Signalment:
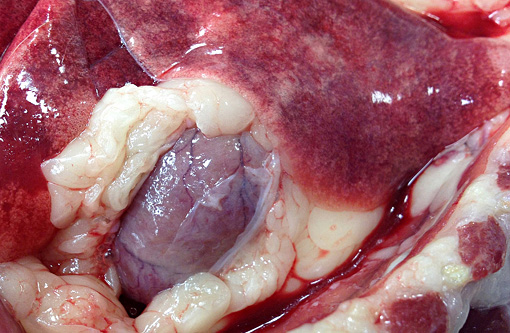
Gross Description:
Incidental findings included a unilateral ectopic ureter, hematoma within the urinary bladder wall and abdominal wall musculature caused by cystocentesis, and bilateral polydactyly of the thoracic limbs.
Histopathologic Description:
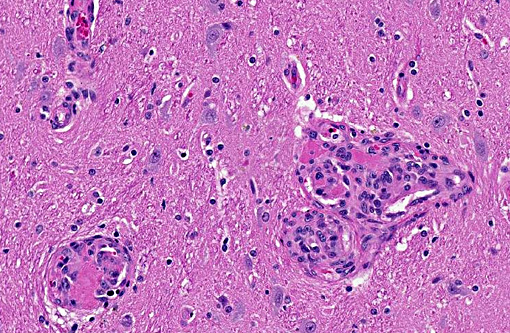
Scattered throughout the entire section, but most numerous in the ventral thalamus and adjacent leptomeningeal blood vessels, are several dozen variably-sized hypercellular foci up to 300 microns in diameter. Most are centered about small to medium-caliber arterioles and consist of mural and luminal proliferations of spindle cells. These form concentric whorls or haphazard tufts that frequently narrow or obliterate the lumen of affected vessels. Multifocally, luminal spindle cells form capillary clefts within which low numbers of red blood cells or fibrin thrombi are present. Spindle cells are bland, with a moderate amount of poorly demarcated smudged to very finely fibrillar eosinophilic cytoplasm. Nuclei are ovoid and pale with vesiculate to stippled chromatin and a single inconspicuous nucleolus. Mitotic figures are very rare. Multifocally, the perivascular (Virchow-Robin) space around both affected and unaffected parenchymal vessels is mildly expanded by clear space or lacy eosinophilic fluid (edema).Â
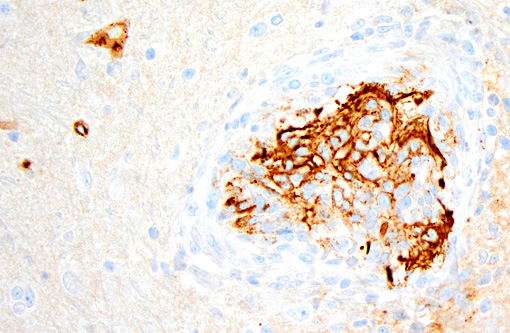
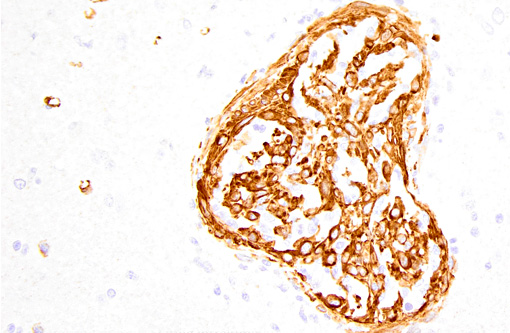
Immunohistochemistry was performed on this section. Approximately 50% of proliferating spindle cells showed strong immunoreactivity for factor VIII-related antigen and the remainder were strongly immunoreactive for smooth muscle actin. IHC for feline coronavirus was also performed. There was no immunoreactivity for feline coronavirus.Â
Morphologic Diagnosis:
Condition:
Contributor Comment:
In FSRA, the heart is the most consistently affected organ and myocardial dysfunction is the cause of illness reported in most cats.(5) The myocardial lesions of FSRA were described in an earlier case submitted to the Wednesday Slide Conference (Conference 03-2008; Case 2). In this current case, however, central nervous system disease caused the most important clinical signs, and the case was submitted as a reminder that FSRA is a multisystemic disease with a variety of clinical presentations. As such, FSRA should be considered in the differential diagnosis for cats with neurologic disease.Â
In this particular case, a specific cause for neurologic signs was not found in the brain sections examined. There was no evidence of obstructive hydrocephalus, severe edema or malacia in any section examined. However, myocardial dysfunction in FSRA is secondary to thrombosis and infarction(5) and it is possible that areas of infarction were present in the brain of this cat but not in sections examined histologically.Â
In addition to the myocardium and brain, other commonly affected organs in FSRA are the kidneys, spleen, lymph nodes, meninges, eyes and pancreas.(5) In the current cat, the most severely affected organs were the brain and heart, but milder vascular lesions were also present in the kidneys and the tunica muscularis of the gastrointestinal tract. Vascular lesions were not present in the urinary bladder and the mural hemorrhage noted at necropsy was attributed to cystocentesis.
The proliferating cells in FSRA are a mixture of endothelial cells and (presumed) pericytes, as identified by IHC for factor VIII-related antigen(5,10,11,13) and smooth muscle actin,(5,13) respectively. Because of the dual cell population and the lack of cellular atypia, lesions of FSRA are thought to represent an aberrant reactive process rather than neoplasia. (This is discussed in Fuji et al.s 2005 review,(5) and by the previous submitter of a myocardial FSRA case to the 2008 Wednesday Slide Conference. Since their reviews of the literature, only one further case report on FSRA has been published.(13))
The cause of FSRA is unknown. In humans, certain vasoproliferative disorders are associated with infection by various Bartonella species,(6) and several mechanisms by which Bartonella spp. might induce endothelial proliferation have been shown.(7,8) Since 2010 a similar link has been investigated in certain vasoproliferative disorders of domestic animals, including FSRA in cats, hemangiopericytoma, hemangiosarcoma and bacillary angiomatosis in dogs and SRA in a steer.(1,2,12) The results of these studies are not conclusive but do suggest that further investigation into Bartonella infection and vasoproliferative disorders in domestic animals is justified.
JPC Diagnosis:
Conference Comment:
While it consistently affects the heart, FSRA is usually observed in multiple organs of which the kidney, spleen, lymph node, gastrointestinal tract, brain, eye, and pancreas seem to be most common.(5) Regardless of affected organs, the disease is often fatal as the vasoproliferative lesions can induce thrombosis, hemorrhage, and necrosis of the myocardium causing acute myocardial failure.(5) Thrombotic thrombocytopenic purpura (TTP) has also been associated with FSRA, and consists of accumulations of large multidimers of vWF, possibly due to an acquired or genetic defect in vWF metalloprotease.(3) TTP may also be associated with certain drugs, viral infections, pregnancy, and systemic lupus erythematosus and may occur a single episode, relapsing intermittent episodes, or chronic unremitting forms.(3)
References:
1. Beerlage C, Varanat M, Linder K, Maggi RG, Cooley J, Kempf VAJ, Breitschwerdt EB: Bartonella vinsonii subsp berkhoffii and Bartonella henselae as potential causes of proliferative vascular diseases in animals. Medical Microbiology and Immunology 201: 319-326, 2012
2. Breitschwerdt EB, Linder KL, Day MJ, Maggi RG, Chomel BB, Kempf VAJ: Koch's Postulates and the Pathogenesis of Comparative Infectious Disease Causation Associated with Bartonella species. Journal of Comparative Pathology 148: 115-125, 2013
3. Breshears MA, Johnson BJ: Systemic reactive angioendotheliomatosis-like syndrome in a steer presumed to be persistently infected with bovine viral diarrhea virus. Veterinary Pathology 45: 645-649, 2008
4. Dunn KA, Smith KC, Blunden AS: Fatal multisystemic intravascular lesions in a cat. Veterinary Record 140: 128-129, 1997
5. Fuji RN, Patton KM, Steinbach TJ, Schulman FY, Bradley GA, Brown TT, Wilson EA, Summers BA: Feline systemic reactive angioendotheliomatosis: Eight cases and literature review. Veterinary Pathology 42: 608-617, 2005
6. Kaiser PO, Riess T, O'Rourke F, Linke D, Kempf VAJ: Bartonella spp.: Throwing light on uncommon human infections. International Journal of Medical Microbiology 301: 7-15, 2011
7. Kempf VAJ, Volkmann B, Schaller M, Sander CA, Alitalo K, Riess T, Autenrieth IB: Evidence of a leading role for VEGF in Bartonella henselae-induced endothelial cell proliferations. Cellular Microbiology 3: 623-632, 2001
8. Riess T, Andersson SGE, Lupas A, Schaller M, Schafer A, Kyme P, Martin J, Walzlein JH, Ehehalt U, Lindroos H, Schirle M, Nordheim A, Autenrieth IB, Kempf VAJ: Bartonella adhesin A mediates a proangiogenic host cell response. Journal of Experimental Medicine 200: 1267-1278, 2004
9. Rongioletti F, Rebora A: Cutaneous reactive angiomatoses: Patterns and classification of reactive vascular proliferation. Journal of the American Academy of Dermatology 49: 887-896, 2003
10. Rothwell TLW, Xu FN, Wills EJ, Middleton DJ, Bow JL, Smith JS, Davies JS: Unusual multisystemic vascular lesions in a cat. Veterinary Pathology 22: 510-512, 1985
11. Straumann Kunz US, Ossent P, Lottstolz G: Generalized Intravascular Proliferation in Two Cats: Endotheliosis or Intravascular Pseudoangiosarcoma? Journal of Comparative Pathology 109: 99-102, 1993
12. Yager JA, Best SJ, Maggi RG, Varanat M, Znajda N, Breitschwerdt EB: Bacillary angiomatosis in an immunosuppressed dog. Veterinary Dermatology 21: 420-428, 2010
13. Zielschot N, Kleinschmidt S, Wohlsein P: Feline systemic reactive angioendotheliomatosis in a cat. Kleintierpraxis 56: 131-134, 2011