Signalment:
50-day-old piglet (
Sus scrofa)The piglet was euthanized because of severe emaciation.
Gross Description:
Consolidation with multiple microabscesses was found in the cranial-ventral portions of the lung. Multiple petechiae and microabscesses were present throughout the liver. The inguinal lymph node was swollen and edematous.
Histopathologic Description:
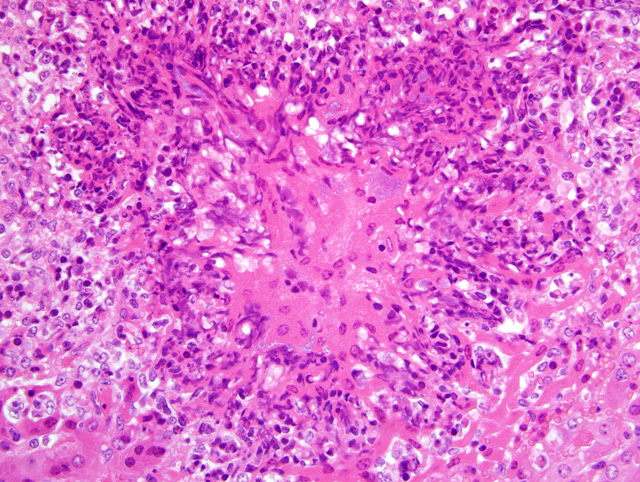
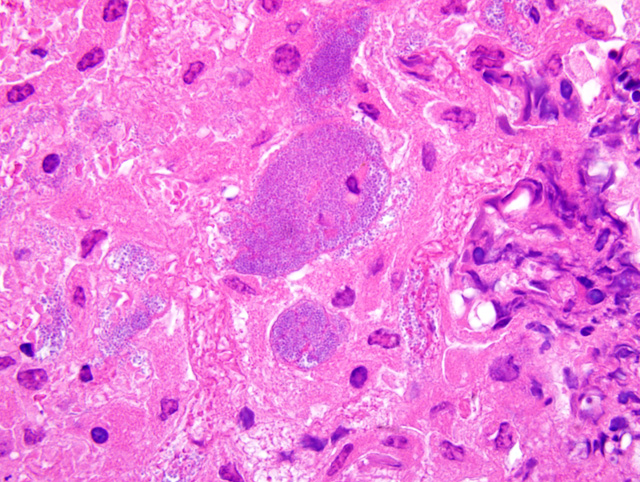
Multiple microabscesses with bacterial colonies at the center of the lesion were found in the liver, lung, spleen, lymph nodes and the serosa of the urinary bladder. The infiltrating inflammatory cells surrounding the bacterial colonies had a spindle-shaped nucleus and cytoplasm which were morphologically different from neutrophils usually seen in a typical abscess. In addition, thrombi often containing bacteria were observed in hepatic portal veins. In the lung, similar lesions of the microabscesses with bacterial colonies were formed in the alveolar space and bronchiolar lumen. Immunohistochemical examination with rabbit hyperimmune serum against
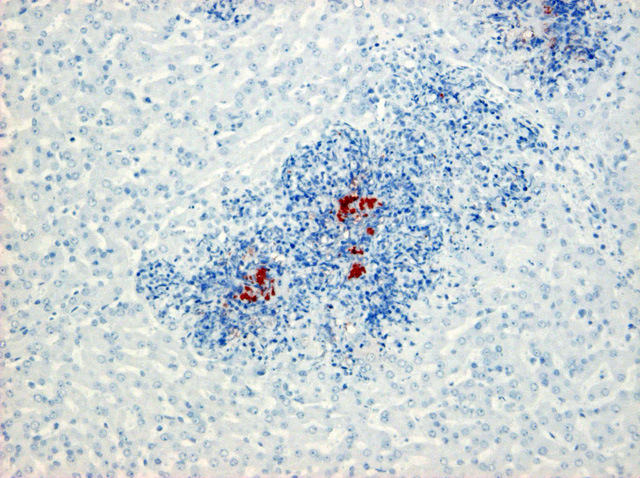
A. pleuropneumoniae type 2 revealed a positive reaction of the bacteria in the liver and lung. Other than the lesions caused by
A. pleuropneumoniae, lymphocytic depletion was found in the various lymphoid tissues. In the tonsil, porcine circovirus type 2 antigens were detected by immunohistochemistry. Intense plasma cell infiltration was observed in the interstitial tissue of the kidney.Â
Balantidium spp. infested in the colon. No lesions were found in the heart, stomach, duodenum, pancreas, ileum and brain.
Morphologic Diagnosis:
Liver: Multiple microabscess formation with bacterial colonies and thrombi.
Lab Results:
Actinobacillus pleuropneumoniae (A. pleuropneumoniae) and
Pasteurella spp. were isolated from the lung.Â
Pasteurella spp. and
Haemophilus parasuis were isolated from the pulmonary lymph node.
Condition:
Actinobacillus pleuropneumoniae
Contributor Comment:
A. pleuropneumoniae is a pathogenic agent of the respiratory disease called porcine pleuropneumonia. In a typical case of actinobacillosis, the histopathological changes are characterized by necrosis, hemorrhage, neutrophil infiltration and widespread edema with fibrinous exudates. However, the present case is characteristic in that multiple microabscesses were observed in various organs. Ohba et al (2) reported that
A. pleuropneumoniae produced granulomatous lesions with asteroid bodies in the liver, showing a possibility of
A. pleuropneumoniae as a pathogenic agent affecting not only the lung but also the liver and other organs. In the present case, asteroid bodies were not observed. However, bacterial colonies positive for
A. pleuropneumoniae type 2 were found at the center of microabscess, indicating that these microabscesses could lead to granulomatous lesions later in the course of the disease.
JPC Diagnosis:
Liver: Hepatitis, random, necrotizing and histiocytic, acute, multifocal, moderate, with fibrin thrombi and colonies of coccobacilli.
Conference Comment:
This very interesting case underscores the importance of
A. pleuropneumoniae not only as a cause of pleuropneumonia in pigs, but as a potential cause of necrotizing lesions in other organs as well. Less common lesions associated with
A. pleuropneumoniae in pigs include meningitis, nephritis, osteomyelitis, arthritis, tenosynovitis, endocarditis, and pericarditis. In a recent case report of 11 pigs with granulomatous hepatitis caused by
A. pleuropneumoniae, 7 had concurrent pleuropneumonia, which the authors concluded was likely the primary site of infection, with secondary dissemination to the liver; a similar pathogenesis is plausible in the present case.(2)
While conference participants generally concurred with the contributors histopathologic description, most could not definitively identify neutrophils either within or adjacent to the lesions, and thus preferred the above morphologic diagnosis over the use of the term microabscess. Participants interpreted the necrotic cells with streaming nuclei as predominantly Kupffer cells and hepatocytes. Despite the rather unusual anatomic location, most participants suspected
A. pleuropneumoniae as the etiology based on 1) the characteristic nuclear streaming in foci of necrosis, 2) the presence of small to occasionally large colonies of bacilli, and 3) the abundance of fibrin within affected areas, particularly in blood vessels and sinusoids.Â
Nuclear streaming of leukocytes is typical in pleuropneumonia caused by
A. pleuropneumoniae, and is attributed to secreted cytotoxins belonging to the repeats in toxin (RTX) family, namely Apx I, II, and III. These potent virulence factors are critical to the development of disease, and mutants lacking the Apx toxin have reduced virulence. Other virulence factors include a capsule that impedes phagocytosis by macrophages, lipopolysaccharide which induces macrophage activation, enzymes (e.g. superoxide dismutase, catalase, and hydroperoxide reductase) that protect against oxidative killing by leukocytes, metalloproteinase, urease, outer membrane proteins, and iron-binding proteins.(1)
References:
1. Caswell JL, Williams KJ: Respiratory system.Â
In: Jubb, Kennedy, and Palmers Pathology of Domestic Animals, ed. Maxie MG, 5th ed., vol. 2, pp. 587-589. Elsevier Saunders, Philadelphia, PA, 2007
2. Ohba, T, Shibahara T, Kobayashi H, Takashima A, Nagoshi M, Osanai R, Kubo M: Multifocal granulomatous hepatitis caused by
Actinobacillus pleuropneumoniae serotype 2 in slaughter pigs. J Comp Pathol
139:61-66, 2008