Signalment:
Gross Description:
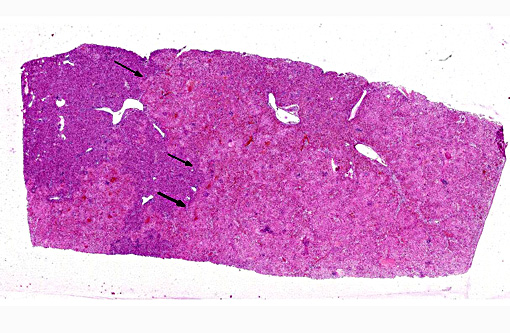
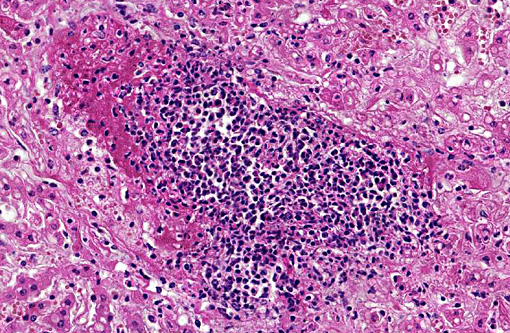
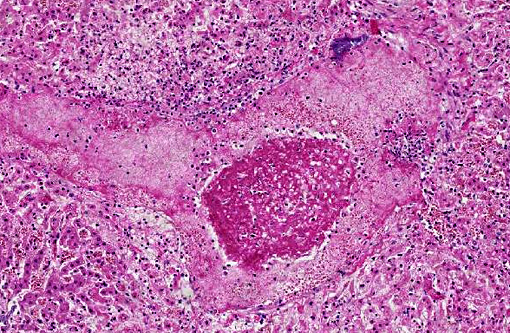
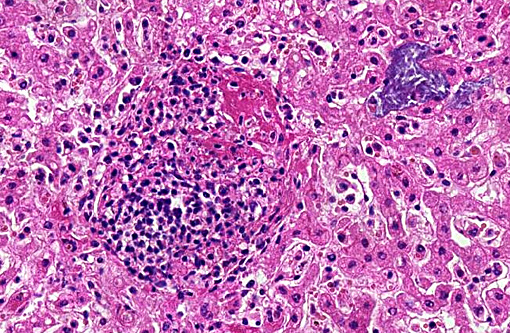
Histopathologic Description:
In the spleen (section not submitted) there were numerous hemosiderin-laden macrophages and increased erythrophagocytosis, associated with moderate lymphoid depletion.
In the lungs (sections not submitted), there was an interstitial pneumonia characterized by marked thickening of the alveolar wall with interstitial infiltration of macrophages, lymphocytes and a few neutrophils. There was multifocal intra-alveolar accumulation of fibrin and a few neutrophils.
Morphologic Diagnosis:
Lab Results:
Condition:
Contributor Comment:
Salmonella is highly invasive, crossing the intestinal epithelial barrier inducing ruffling of the apical surface of the epithelial layer, which results in internalization of the organism within a cytosolic membrane bound vacuole. This process is dependent on the expression of several effector bacterial proteins that are translocated into the host cells through a type III secretion system encoded by the Salmonella pathogenicity island 1 (SPI-1).(9-12) Once the organism crosses the epithelial layer, it quickly localizes in the lamina propria, where it is found mostly within macrophages. Salmonella processes another type III secretion system encoded by SPI-2 that is activated as soon as the bacteria localizes in the phagosome. This SPI-2-encoded type III secretion system is essential for intracellular survival of the organism in macrophages and, therefore, systemic dissemination of the infection.(8)
In our empirical experience, Salmonella is usually not observed in HE-stained section, both in enteric and systemic sites of infection. Interestingly, in the present case there were abundant intralesional bacterial colonies, which likely represent postmortem overgrowth of the organism.
JPC Diagnosis:
Conference Comment:
Typhoidal serotypes of Salmonella spp. are less common than the nontyphoidal forms in most species, including humans with the exception being in mice. Mice regularly develop septicemia with oral inoculation of Salmonella isolates, often without exhibiting gross and microscopic changes in the alimentary tract.(2,7)
The nontyphoidal or enteric form of salmonellosis, characterized by self-limiting enterocolitis or diarrhea in the absence of systemic disease, is a major cause of morbidity and mortality in calves and often over one billion human cases per year. While both are incriminated in cattle, Salmonella enterica serovar Typhimurium infects all species and is the most common isolate in people, while S. enterica serovar Dublin is more specific to cattle.(1,6) The similarity in enteric disease manifestation between cattle and humans and has led to their use as an experimental model for human infections.(6,11) Characteristic histopathology of nontyphoidal salmonellosis is the massive influx of neutrophils, which is also suggested as the major triggering event of gastrointestinal necrosis and diarrhea.(6) Vasculitis and thrombosis is also prominent, and in severe cases can lead to rectal strictures in pigs who have poor collateral circulation.(4) Fibrinous cholecystitis is pathognomonic for acute enteric salmonellosis in calves.(4) Chronic enteric salmonellosis is characterized by discrete foci of necrosis and ulceration called button ulcers and seen most often in pigs but also cattle and horses.(4)
The colonies of coccobacilli and large numbers of rods found in some areas of the slide are both gram-positive, leading conference participants to conclude these are postmortem bacterial overgrowth not related to the Salmonella infection in this case.
References:
1. Brown CC, Baker DC, Barker IK. Alimentary system. In: Maxie MG, ed. Jubb, Kennedy, and Palmers Pathology of Domestic Animals. 5th ed. Vol 2. Philadelphia, PA: Elsevier Saunders; 2007:200-202.
2. Brown DE, Libby SJ, Moreland SM, et al. Salmonella enterica causes more severe inflammatory disease in C57/BL6 NrampIG169 mice than SV129S6 mice. Vet Pathol. 2013;50(5):867-876.
3. Cullen, JM, Brown DL. Hepatobiliary system and exocrine pancreas. In: Zachary JF, McGavin MD, eds. Pathologic Basis of Veterinary Disease. 5th ed. St. Louis, MO: Elsevier Mosby; 2012:433.
4. Gelberg HB. Alimentary system and the peritoneum, omentum, mesentery, and peritoneal cavity. In: Zachary JF, McGavin MD, eds. Pathologic Basis of Veterinary Disease. 5th ed. St. Louis, MO: Elsevier Mosby; 2012:376-377.
5. Morter RL, Armstrong CH, Amstutz HE, Thacker HL: Systemic salmonellosis in mature beef cows. J Vet Diagn Invest 1:22-24, 1989
6. Nunes JS, Lawhon SD, Rossetti CA, et al. Morphologic and cytokine profile characterization of Salmonella enteric serovar typhimurium infection in calves with bovine leukocyte adhesion deficiency. Vet Pathol. 2010;47(2):322-333.
7. Percy DH, Barthold SW. Pathology of Laboratory Rodents and Rabbits 3rd ed. Ames, IA: Blackwell Publishing; 2007:61-63.
8. Santos RL, B+â-ñumler AJ: Cell tropism of Salmonella enterica. Int J Med Microbiol 294:225-233, 2004
9. Santos RL, Tsolis RM, B+â-ñumler AJ, Adams LG: Pathogenesis of Salmonella-induced enteritis: a review. Braz J Med Biol Sci 36:3-12, 2003
10. Santos RL, Zhang S, Tsolis RM, Kingsley RA, Adams LG, Baumler AJ: Animal models of Salmonella infections: gastroenteritis vs. typhoid fever. Microbes Infect 3:1335-1344, 2001
11. Santos RL, Zhang S, Tsolis RM, B+â-ñumler AJ, Adams LG: Morphologic and molecular characterization of Salmonella typhimurium infection in neonatal calves. Vet Pathol 39:200-215, 2002
12. Zhang S, Santos RL, Tsolis RM, Stender S, Hardt WD, B+â-ñumler AJ, Adams LG: The Salmonella enterica serotype Typhimurium effector proteins SipA, SopA, SopB, SopD and SopE2 act in concert to induce diarrhea in calves. Infect Immun 70:3843-3855, 2002