Signalment:
Six-month-old,
male castrated, cross breed ox, (
Bos taurus).An outbreak of
neurological disease occurred in a herd of 99, 6 to 12-month-old, mixed breed,
beef cattle. The herd was grazing pasture with a daily supplementary ration of
2 kg/head of sprouted barley. Fifty-seven animals were affected and
eighteen animals were euthanized after becoming recumbent and unable to rise.
The cattle had been fed hydroponically sprouted barley for seven months before
the outbreak. Over the last few weeks of feeding, a green-blue downy mold had
grown on the barley, its development had coincided with some unseasonably warm
and humid spring weather. The mold did not have any effect on the appetite of
the cattle which were being fed approximately 2 kg/head daily when adverse
signs were first noticed. The first indication of toxicity was noticed by the
owners when a 12-month-old cross-bred heifer showed signs of what was thought
to be colic, based on her hunched posture and reluctance to walk. The next day
another two cattle were noticed sick and three more the following day. At this
stage the feeding of the sprouted barley was discontinued and the cattle were
moved to another paddock. New cases continued to develop over the next 18 days.
By six weeks, only a few of the affected cattle had not recovered, though all
had lost considerable weight. A range of clinical signs was observed. Mildly
sick cattle were instantly recognizable by the arching of their backs. Unless
disturbed, most appeared to graze normally. Ataxia, knuckling of the hind
fetlocks and hypermetria of hind limbs were also obvious in affected cattle.
Some cattle developed progressively
worsening ataxia with generalized muscle tremors. The cattle that were
euthanized had progressed to posterior paralysis, recumbency and were often
polypneic. One sternally recumbent animal appeared to be blind. Another was
found in lateral recumbency displaying opisthotonos.
The
mold on the barley sprouts was identified as
Aspergillus clavatus (Agrifood
Technology, Princes Hwy Werribee, Victoria 3030).
A. clavatus is
identifiable by its long, smooth conidiophore and huge club-shaped vesicle to
which are attached uniseriate phialides.
3 The mould was confirmed as
A. clavatus and a specimen is held at the DPI, Plant Diseases Herbarium,
Knoxfield Victoria.
Gross Description:
There
were no remarkable gross changes seen in the internal organs of the necropsied animals.
Histopathologic Description:
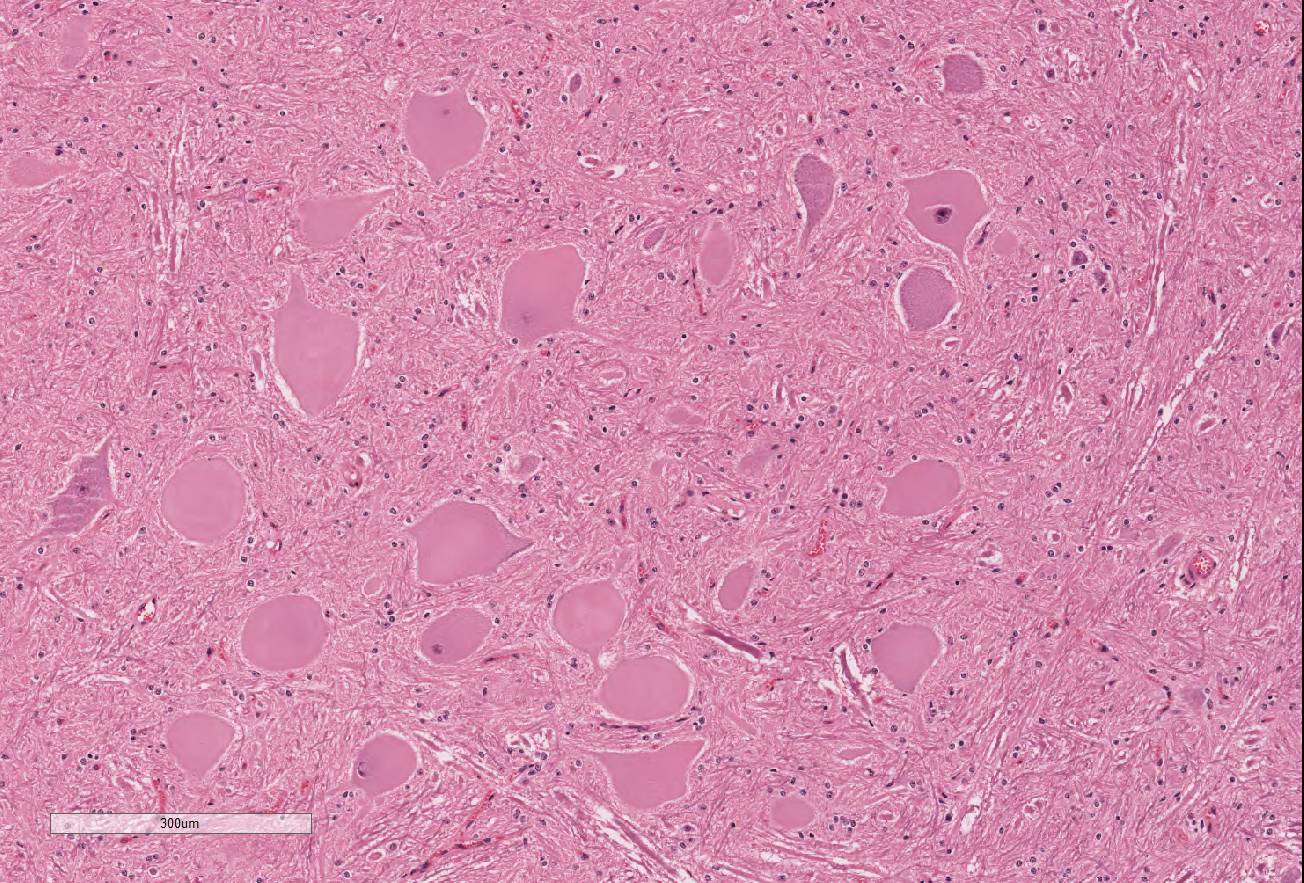
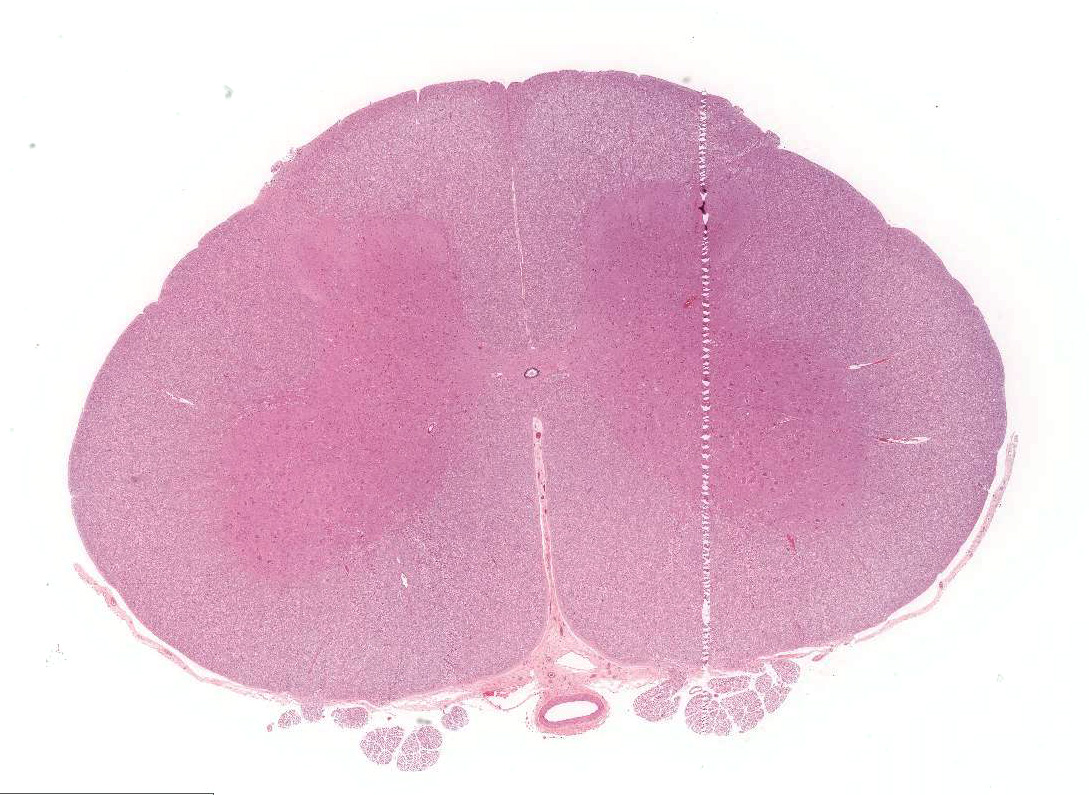
Significant histological abnormalities are limited
to the central nervous system. Neuronal changes are apparent in the brain and
spinal cord (spinal cord sections submitted for WSC). Affected neurons are
swollen and rounded, completely chromatolytic with pale or acidophilic
cytoplasm, and the few nuclei captured in section are peripheral, shrunken and
often pressed against the cell membrane. Many nuclei are affected, especially the
red nuclei, dorsal motor nucleus of the vagus, reticular nucleus of the medulla
and alpha motor neurons among the lateral and medial motor neurons of the
cervical and lumbar intumescences. Small gamma efferent neurons are not
involved. There is no apparent cell loss or glial reaction. Within the brain
and spinal cord, there is minor axonal swelling and myelin vacuolation with
astrocytic swelling. Otherwise, the integrity of myelin and axons is preserved.
The peripheral nervous system, including autonomic ganglia, peripheral nerves,
and craniospinal nerve roots, shows no abnormality.
Morphologic Diagnosis:
Brain and spinal cord: chromatolytic neuronal
degeneration, severe multifocal acute, with mild edema of myelin.
Lab Results:
Biochemical
analysis of sera from the two recently recumbent cattle found mild to moderate
elevations in GLDH (428 and 30 U/L; reference range < 20 U/L) and LDH (2318
and 2395 U/L; reference range 50-400 U/L). CK was mildly elevated in one
animal, as was AST in the other.
Condition:
Aspergillus clavatus
Contributor Comment:
The
epi-demiology, clinical signs, histological findings in the brain and spinal
cord and mycological examination in this case
3 are consistent with
those previously described and caused by
Aspergillus clavatus tremor-genic
neurotoxicosis.
5,6
A clavatus is present in
soil and commonly isolated from cereal grains and their germinated seeds and
other feedstuffs. It also occurs commonly in pigeon droppings.
6
Tremorgenic syndromes have been experimentally reproduced in ruminants by
feeding
A clavatus pure cultures and
A clavatus contaminated
grains. A number of toxins have been isolated from A clavatus including
cytochalasin E, tryptoquivaline, tryptoquivalone, patulin, but none have been
tested in ruminants to determine responsibility for the tremorgenic syndrome.
Toxic extracts from sorghum beer residue capable of reproducing the tremogenic
syndrome in sheep dosed orally did not contain patulin, trypotquivalone or nortryptoquivalone
so the identity of the toxin(s) remains undetermined.
6
The
case is submitted to the WSC with the kind permission of the surviving author,
Dr C El-Hage, University of Melbourne.
JPC Diagnosis:
Spinal cord,
grey matter: Neuronal degeneration, multifocal, severe, with chromatolysis and
vacuolar de-generation, cross breed ox,
Bos taurus.
Conference Comment:
The contributor provides a striking example of severe toxin-induced swelling
and central chromatolysis of neuronal cell bodies (soma). In this case, the
pathogenic process is most severe in the grey matter of the ventral horns. The
ventral horn of the spinal cord contains nuclei for lower motor neurons that
supply motor input to somatic muscle via axons in the white matter of the
ventral funiculus. Typically, lesions in lower motor neurons produce flaccid
paralysis, rather than the muscular tremors and hyperesthesia present in this
case; however, as mentioned by the contributor, the pathogenic mechanisms of
neuromycotoxicosis in
Aspergillus clavatus have not yet been determined.
Some authors
consider the mycotoxin patulin, produced by
Aspergillus sp.
and
Penicillium sp, to be the major contributor to the neurotoxicity induced by
A. clavatus.
1,4 Patulin has been implicated in previously
reported cases of neurotoxicity in animals. In the brain and spinal cord,
patulin inhibits acetylcholinesterase and the Na+/K+-ATPase resulting in a
buildup of the stimulatory neurotransmitter ace-tylcholine.
1,4
Decreased breakdown of acetylcholine at the neuromuscular junction results in
convulsions, tremors, stiffness, impaired locomotion, and hyperesthesia, seen
clinically in this case.
4 Additionally, inhibition of the
Na+/K+-ATPase may have devastating effects on the cell, including ionic
depolarization of the cell membrane resulting in neuronal signal transduction
deficiencies and disruption of the cells concentration gradient causing
increased osmolarity and cellular swelling.
4
Conference
participants discussed the degenerative changes associated with chromatolysis
in the central nervous system. Chromatolysis represents a change in the
histomorphologic appearance of the soma due to the central or peripheral
dispersal pattern of the Nissl substance.
2 The chromatolyic pattern
in this case is consistent with central chromatolysis. Central chromatolysis
occurs in the large neurons of the brainstem, spinal motor neurons, and
peripheral ganglia and is characterized by central clearance of Nissl granules
and marked cellular swelling with pale, eosinophilic, homogenous, ground-glass
appearance to the cytoplasm.
2 Soma nuclei are often peripheralized
and possess prominent nucleoli. Typically, central chromatolysis occurs
secondary to axonal injury and represents a reparative response in the soma,
incorporating increased free ribosomes for protein synthesis, lysosomes, and
mitochondria. This anabolic response required for axon
regeneration is referred to as the axon
reaction Central chromatolysis occurs in a number of neurodegenerative diseases,
including copper deficiency in sheep and goats, grass sickness in horses, avian
encephalomyelitis virus in chickens, and feline dysautonomia, known as
Key-Gaskell syndrome, in cats. Peripheral chromatolysis is generally associated
with cell body shrinkage rather than swelling. It is characterized by Nissl
granules surrounding the nucleus and is a relatively nonspecific degenerative
lesion.
2
References:
1. Brotha
CJ, Legg MJ, Truter M, Sulyok M. Multitoxin analysis of
Aspergillus clavatus-infected
feed samples implicated in two outbreaks of neuromycotoxicosis in South Africa.
Ondersepoort J Vet Res. 2014; 12:
doi:10.4102/ojvr.v81i1.848.
2. Cantile C, Youssef S. Nervous system. In: In: Maxie MG, ed. Jubb
Kennedy and Palmer's Pathology of Domestic Animals. Vol 1. 6th ed.
Philadelphia, PA: Elsevier Saunders; 2016:252-255,327.
3. Hage C M; Lancaster M J: Mycotoxic nervous disease in cattle fed
sprouted barley contaminated with Aspergillus clavatus. Aus Vet J.
2004; 82:639-641.
4. Loretti AP, Colodel EM, Driemeier D, et al. Neurological disorder in
dairy cattle associated with consumption of beer residues contaminated with Aspergillus
clavatus. J Vet Diagn Invest. 2003; 15:123-132.
5. Maxie MG, Youssef S. Nervous system. In: Jubb, Kennedy and Palmers
Pathology of Domestic Animals. Maxie MG, ed. 5th ed, Edinburgh
UK: Elsevier Saunders; 2007:369
6. McKenzie RA, Kelly MA, Shivas RG, et al. Aspergillus clavatus
tremorgenic neurotoxicosis in cattle fed sprouted grains. Aus Vet J.
2004; 82:635-638.