Signalment:
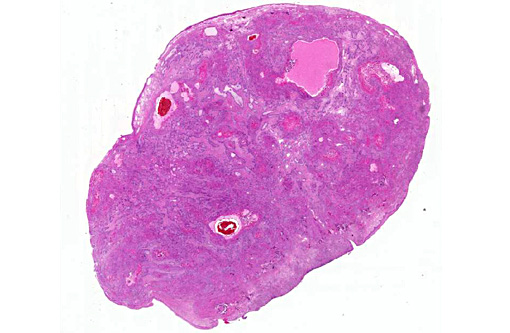
Gross Description:
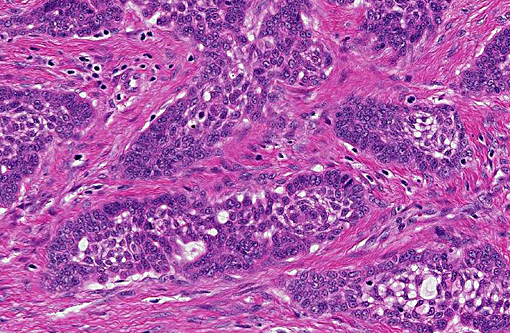
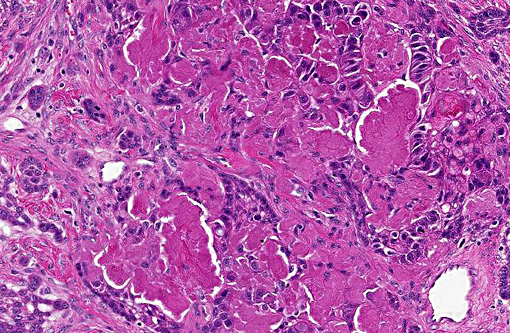
Histopathologic Description:
Morphologic Diagnosis:
Condition:
Contributor Comment:
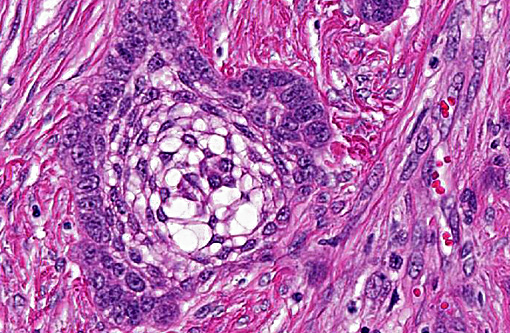
In this case the overlying mucosa is ulcerated, with associated neutrophilic infiltration. Additionally, there is accumulation of smudged, pale eosinophilic hyaline material, both within the islands and cords as well as between them. This material is consistent with amyloid, as it stains strongly with Congo red, and is pale green under polarized light.(10)
Amyloid-producing odontogenic tumors (APOT) have been described in dogs, cats and a Bengali tiger.(8-10) They have been reported as focally extensive, infiltrating, firm to friable lesions, frequently involving the entire maxilla.(2,8) These tumors are slowly progressive, locally invasive and can recur locally after excision.(11) There are no reports of metastasis of APOT in the literature.(4)
The histological feature that sets APOTs apart from ameloblastoma and acanthomatous ameloblastoma is the accumulation of amyloid between neoplastic epithelial cells. Other features are shared between these tumors.(5) Recently, the amyloid found in a single case of feline APOT was typed as odontogenic ameloblast-associated protein (formerly termed APin).(2)
Ultrastructurally in APOTs, cords and islands are delimited by a basement membrane, to which cuboidal to polygonal cells attach by hemidesmosomes. There are also numerous desmosomes between adjacent cells (consistent with epithelial cells). All these cells contain numerous tonofilaments, and pseudoinclusions containing 10nm thick filaments are occasionally noted. The ultrastructural presentation of the amyloid noted on light microscopy consists of intercellular accumulations of haphazardly arranged, non-branching, 10nm diameter filaments, which are frequently in direct contact with the epithelial cells.(3)
With regards to terminology, APOTs have also been designated calcifying epithelial odontogenic tumors in the past.(4) This designation has fallen out of favor, as this term is used to name an odontogenic tumor in humans that has a different histological presentation. Calcifying epithelial odontogenic tumors in humans frequently contain sheets of polyhedral cells, with extracellular mineralization of amyloid deposits and intracellular mineralization. Although these features are present in APOTs of dogs and cats, they are rare.(5)
JPC Diagnosis:
Conference Comment:
The majority of, if not all, odontogenic tumors can fit into one of three categories: epithelial, mixed or inductive, and mesenchymal. All of the various manifestations of ameloblastoma, including canine acanthomatous ameloblastoma, solid/multicystic ameloblastoma and the tumors discussed above are epithelial derived. Mixed or inductive tumors consist of ameloblastic fibroma/fibro-odontoma and feline inductive odontogenic tumors. These are composed of proliferative odontogenic epithelium and odontogenic ectomesenchyme, often with inductive change, and may all occur along a single continuum. Finally, mesenchymal proliferations are perhaps most common, and consist of peripheral odontogenic fibroma (POF) and focal fibrous hyperplasia. The term POF has replaced the use of fibromatous epulis of periodontal ligament origin (FEPLO) among many veterinary surgical pathologists because epulis is a nonspecific term, and the lesion is derived from periosteal stroma rather than periodontal ligament. Both POF and FFH are recognized as separate entities, but also share overlapping clinical and pathologic features.(1)
References:
1. Bell CM, Soukup JW. Nomenclature and classification of odontogenic tumors part II: clarification of specific nomenclature. J Vet Dent. 2014;31(4):234-243.
2. Bock P, Hach V, Baumg+â-ñrtner W: Oral Masses in Two Cats. Vet Pathol (ahead of print, 2011).
3. Breuer W, Geisel O, Linke RP, Hermanns W: Light microscopic, ultrastructural, and immunohistochemical examinations of 2 calcifying epithelial odontogenic tumors (CEOT) in a dog and a cat. Vet Pathol 31:415420, 1994.
4. Gardner, DG, Dubielzig, RR: The so-called calcifying epithelial odontogenic tumour in dogs and cats (amyloid-producing odontogenic tumor) J Comp Pathol 111:221230. 1994.
5. Head KW, Cullen JM, Dubielzig RR, Else RW, Misdorp W, Patnaik AK, Tateyama S, van der Gaag I: Histological Classification of Tumors of the Alimentary System of Domestic Animals, 2nd ed., Vol. X. pp. 46-57. Armed Forces Institute of Pathology, Washington DC, 2003.
6. Head KW, Else RW, Dubielzig RR: Tumors of the Alimentary Tract. In: Tumors in Domestic Animals, ed. Meuten DJ, 4th ed., pp. 402-410. Iowa State Press, Iowa, 2002.
7. Hirayama K, Miyasho T, Ohmachi T, et al. Biochemical and immunohistochemical characterization of the amyloid in canine amyloid-producing odontogenic tumor. Vet Pathol. 2010;47:915-922.
8. Kang MS, Park MS, Kwon SW, Ma SA, Cho DY, Kim DY, Kim Y: Amyloid-producing odontogenic tumour (calcifying epithelial odontogenic tumour) in the mandible of a Bengal tiger (Panthera tigris tigris). J Comp Pathol 134:236240, 2006.
9. Langham RF, Bennett R, Koestner A: Amyloidosis associated with a calcifying ameloblastoma (calcifying epithelial odontoma) in a cat. Vet Pathol 21:549550. 1984.
10. Myers RK and McGavin MD: Cellular and Tissue responses to Injury. In: Pathological Basis of Veterinary Disease, ed. McGavin MD and Zachary JF, 4th ed., pp.45-46. Mosby, Inc., Washington DC, 2003.
11. Poulet FM, Valentine, BA, Summers, BA: A survey of epithelial odontogenic tumors and cysts in dogs and cats. Vet Pathol 29:369380. 1992.