Signalment:
Gross Description:
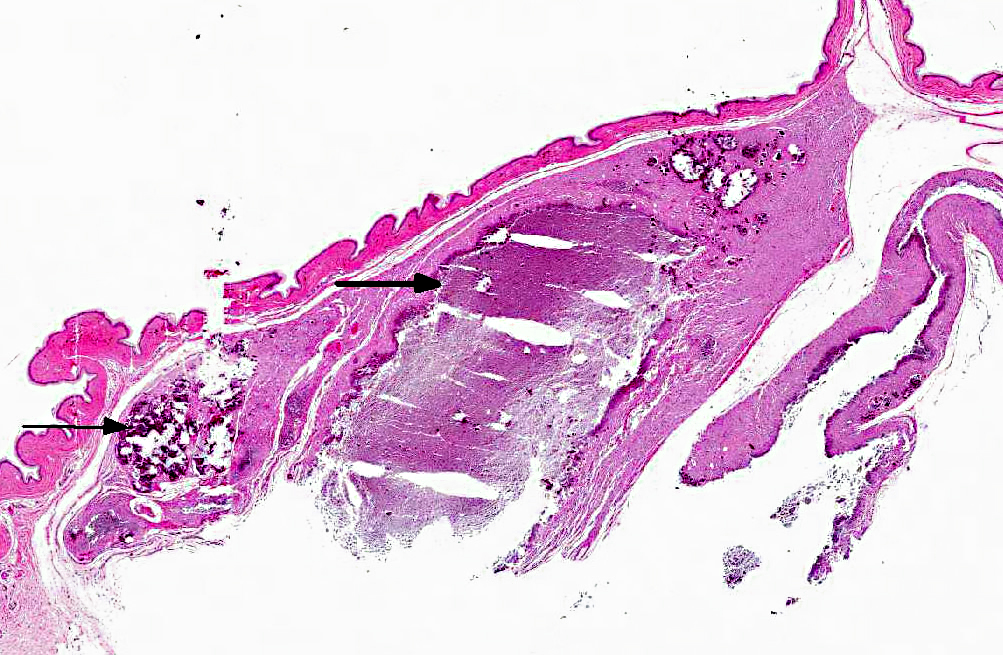
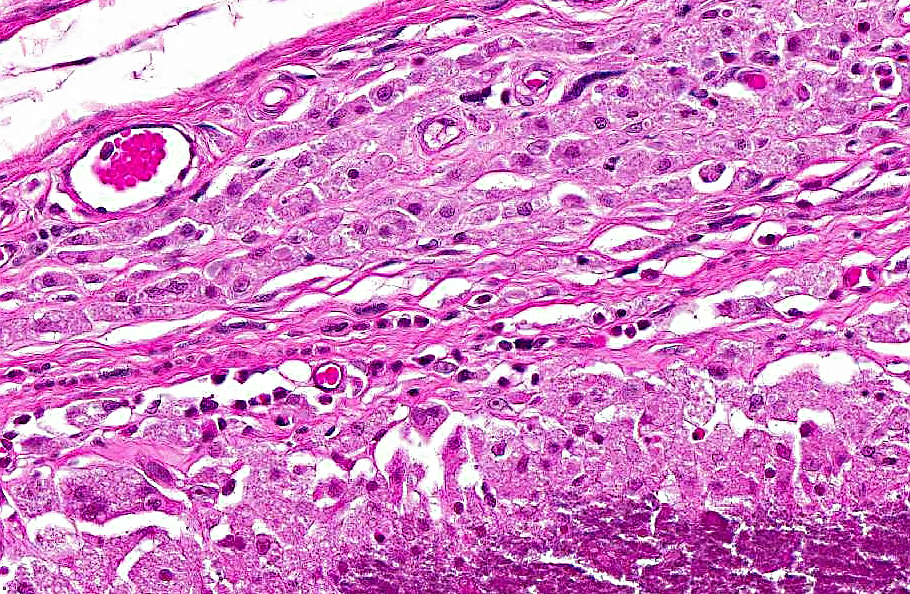
Histopathologic Description:
In each of the excisional biopsy samples there is a large dermal granuloma with central mineral as described in the preceding paragraph.
Morphologic Diagnosis:
Haired skin, ventral body wall: Moderate to severe, multifocal dermal mineralization (calcinosis cutis) with granulomatous dermatitis, epidermal necrosis, ulceration, mild orthokeratotic hyperkeratosis and serocellular surface crusts.
Condition:
Contributor Comment:
Calcinosis cutis and calcinosis circumscripta are characterized by ectopic cutaneous and subcutaneous deposition of calcium salts and may be part of a metastatic or dystrophic process, though in domestic mammals the lesions are often idiopathic.(10) Calcinosis cutis refers to lesions in which there is more diffuse involvement of collagen and adnexa while the diagnosis of calcinosis circumscripta is reserved for circumscribed mass-like lesions (also called tumoral calcinosis).(5) Interestingly, the lesions in these naked mole rats appear to have features of both. The larger mass at the neck and shoulder is a nodular accumulation of mineral with secondary granulomatous inflammation, consistent with calcinosis circumscripta. Dermal lesions along the body wall are more diffuse and characteristic of calcinosis cutis.
In domestic animals, calcinosis cutis is most often associated with either iatrogenic or Cushings disease-related hyperglucocorticoidism. In domestic dogs, calcinosis circumscripta has been reported in the footpads in association with renal failure. Lesions also occur in the tongue, possibly secondary to local trauma. Additionally there is an uncommon form of idiopathic cutaneous calcinosis circumscripta that seems to occur mainly in large breed dogs, particularly of the German Shepherd dogs. In these cases, dogs often have normal serum calcium and phosphorus levels, a lack of identifiable nutritional deficiency or excess and lack of known preexisting trauma to the affected areas.(10)
As is frequently the case in domestic mammals, the cause of the lesions in these naked mole rats is poorly understood. There is a strong temporal and spatial association between rabies vaccination and the tumoral calcinosis, but this may not explain the calcinosis cutislike lesions along the body wall. Skin trauma in naked mole rats does not typically lead to calcinosis; perhaps mineral deposition in these cases was exacerbated by the injection. The vaccine given (IMRAB-� 3, Merial) makes use of an aluminum hydroxide adjuvant to promote macrophage antigen presentation and contains gentamicin as a preservative. Naked mole rats are not typically vaccinated for rabies at this institution so it is unknown if the type of vaccine and/or adjuvant had anything to do with the development of the lesions. In those animals treated via surgical excision, tumoral lesions did not recur. There did not appear to be calcinosis cutis-like lesions in the biopsied animals, though sampling of skin in places other than at tumoral lesions was not attempted. Injection site calcinosis circumscripta has been previously reported in a dog that had received medroxyprogesterone acetate contraception.(4) No reports were found in which calcinosis circumscripta was associated with vaccination.
Given that multiple members of the colony were affected, a review of commonalities among these animals (such as diet, genetics and clinical history) and possibly serum chemistry analyses may be helpful. Adrenal and pituitary glands in the necropsied cases were grossly and histologically within normal limits, so hyperadrenocorticism was considered less likely. Given their subterranean lifestyle and lack of normal exposure to vitamin D, differences in calcium/phosphorus metabolism could make this animal more susceptible to cutaneous calcinosis. Abnormal skin calcification has been reported previously in naked mole rats. In this study, despite massive dietary vitamin D overload, mineralization was only evident in the skin and in increased tooth density. It was speculated that in naked mole rats, skin and teeth may be used for deposition and subsequent sloughing of excess calcium, perhaps to help prevent metastatic calcification in other organs.(3)
Naked mole rats are a fascinating rodent species of relatively recent interest as research animals. Naked mole rats are native to Africa where they live deep underground in a complex system of burrows. They are a eusocial species, meaning they have a complex social structure whereby many non-reproductive -�-�workers care for a single dominant -�-�queen and a few other reproductively active individuals.(8) They are extraordinarily long-lived for a rodent species (in excess of 20 years). Naked mole rats appear to lack pain response to certain irritants (ammonia, capsaicin and acids) and have a high tolerance for hypoxic conditions, adaptations that are likely courtesy of the extreme environment in which they live. Neoplastic disease has yet to be reported in this species.(1,2,6) For these reasons, naked mole rats are being utilized in aging, pain and cancer studies.
JPC Diagnosis:
1. Haired skin, neck: Dermal granulomas, calcareous, multiple, coalescing (calcinosis circumscripta), with mild epidermal pigmentary incontinence.
2. Haired skin, ventral body wall: Dermatitis, granulomatous, diffuse, marked, with multifocal dermal mineralization (calcinosis cutis) and fibrosis, moderate acanthosis, orthokeratotic hyperkeratosis and serocellular surface crusts.
Conference Comment:
In addition to the naked mole rats described by the contributor, other animals affected by calcinosis circumscripta include dogs, cats, horses, cows, buffalo, rabbits, turtles, nonhuman primates and a captive antelope. Reports of calcinosis circumscripta in nonhuman primates include two female rhesus macaques and a male common marmoset, in which the lesion was associated with microchip implantation. Additionally, idiopathic calcinosis circumscripta has been reported in two cynamologus macaques and a case of bilateral dystrophic calcinosis circumscripta was recently reported in a six-year-old female cynomolgus macaque with a history of bilateral foot trauma.(9)
There was moderate slide variation; the slide used for the JPC morphologic diagnosis did not exhibit the necrosis and ulceration associated with the calcinosis cutis as described by the contributor.
References:
2. Borges RM. Of pungency, pain, and naked mole rats: chili peppers revisited. J Biosci. 2009;34:349-351.
3. Buffenstein R, Laundy MT, Pitcher T, Pettifor JM. Vitamin D3 intoxication in naked mole-rats (Heterocephalus glaber) leads to hypercalcaemia and increased calcium deposition in teeth with evidence of abnormal skin calcification. Gen Comp Endocrinol. 1995;99:35-40.
4. Ginel PJ, Lopez R, Rivas R, Perez J, Mozos E. A further case of medroxyprogesterone acetate associated with calcinosis circumscripta in the dog. Vet Rec. 1995;136:44-45.
5. Gross TL, Ihrke PJ, Walder EJ, Affolter VK. Degenerative, dysplastic and depositional diseases of dermal connective tissue. In: Skin Diseases of the Dog and Cat. 2nd ed. Ames, IA; 2005:373-380.
6. Larson J, Park TJ. Extreme hypoxia tolerance of naked mole rat brain. Neuroreport. 2009:20;1634-1637.
7. Kumar V, Abbas AK, Fausto N. Cellular adaptations, cell injury and cell death. In: Pathologic Basis of Disease. 7th ed. Philadelphia, PA: Elsevier Saunders; 2005:41-42.
8. Nowak RM, Paradiso JL. Rodentia; Bathyergidae; genus Heterocephalus. In: Walkers Mammals of the World. 4th ed. Baltimore, MD: Johns Hopkins University Press; 1983:855-857.
9. Radi ZA, Sato K. Bilateral dystrophic calcinosis crcumscripta in a cynomolgus macaque (Macaca fascicularis). Toxicol Pathol. 2010;38:637.
10. Scott DW, Buerger RG. Idiopathic calcinosis circumscripta in the dog: a retrospective analysis of 130 cases. J Am Anim Hosp Assoc. 1988;24:651-658.