CASE III: 31712 (JPC 4153553)
Signalment:
Male, howler monkey, Alouatta spp. (age unknown)
History:
Multiple free ranging neotropical primates were found dead in the state of Rio de Janeiro, Brazil during the last outbreak of yellow fever (YF). All primates found dead were submitted to necropsy and YF diagnosis as a part of the YF epizootic surveillance program of the Brazilian Ministry of Health. From 1,304 primates, 56 were considered positive for YF by real time RT-PCR from the official diagnostic laboratory. Several non-human primate species tested positive for YF, including Alouatta sp., Callithrix sp., Sapajus sp., Callicebus sp. and Leonthopitecus sp.
The slide presented here is from a howler monkey (Alouatta sp.) found dead in a rural area in the State of Rio de Janeiro, Brazil.
Gross Pathology:
No data available.
Laboratory results:
Yellow Fever RNA was detected in tissue samples by Real Time RT-PCR performed by the official diagnostic laboratory.
Microscopic Description:
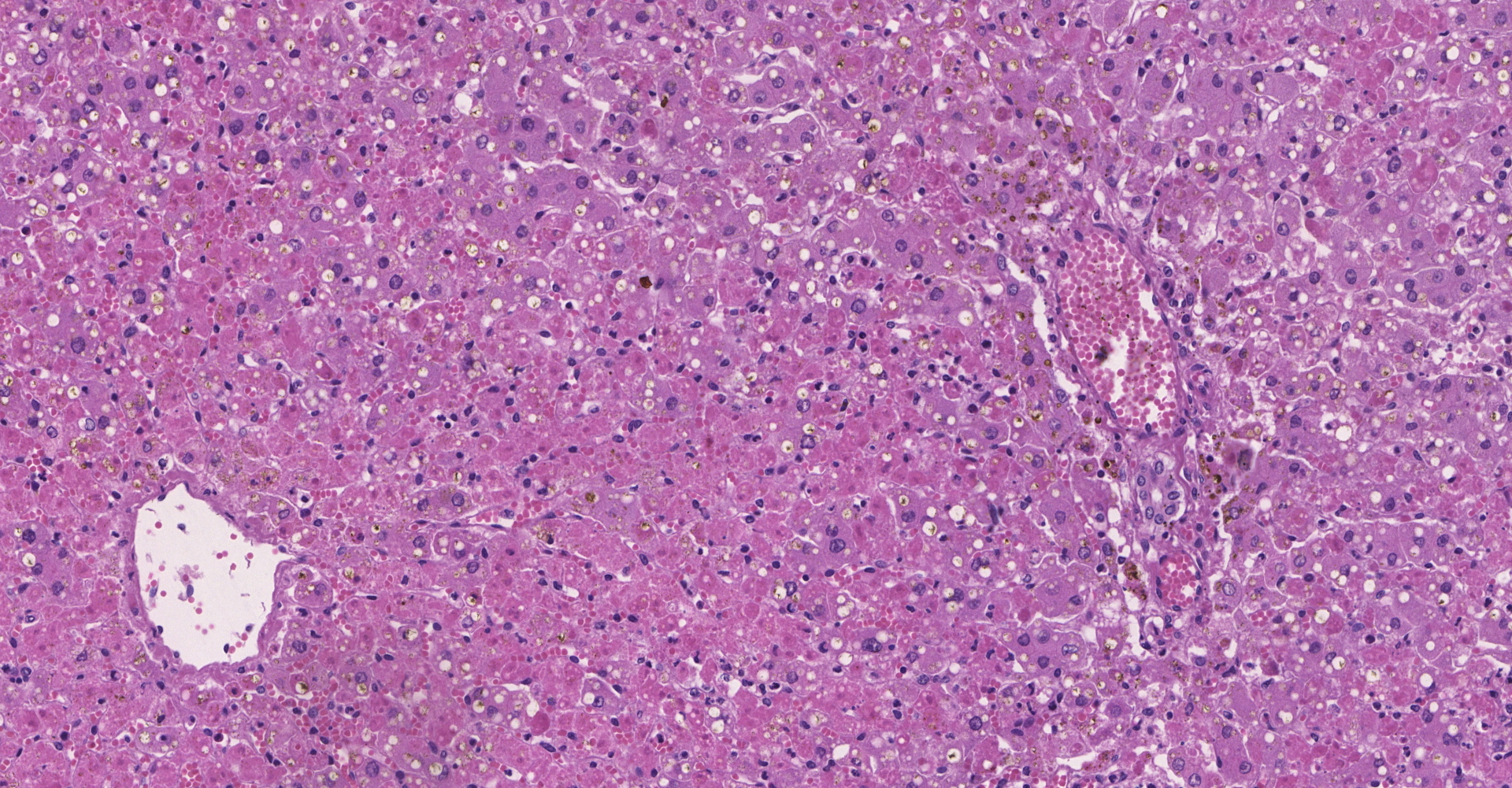
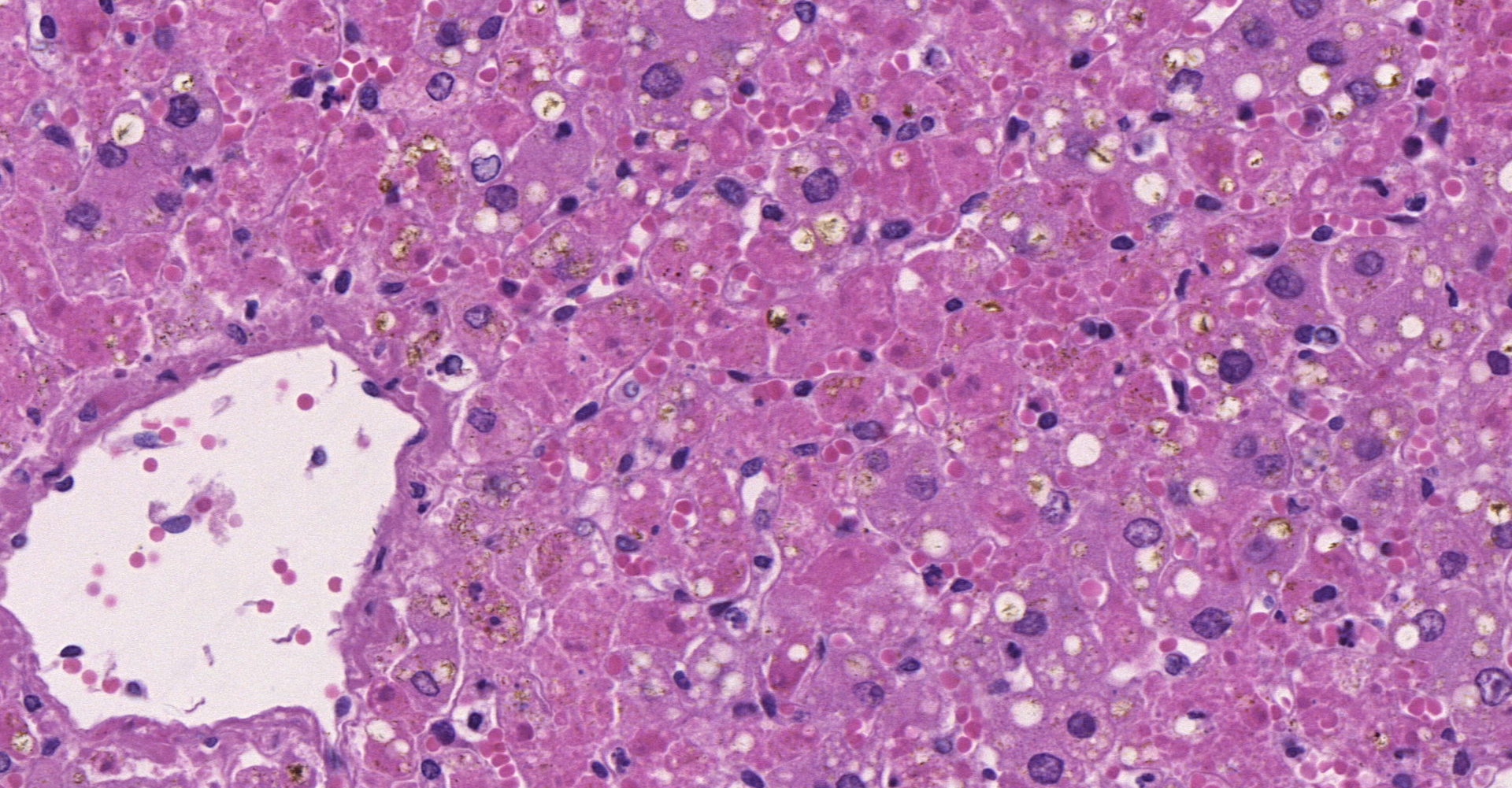
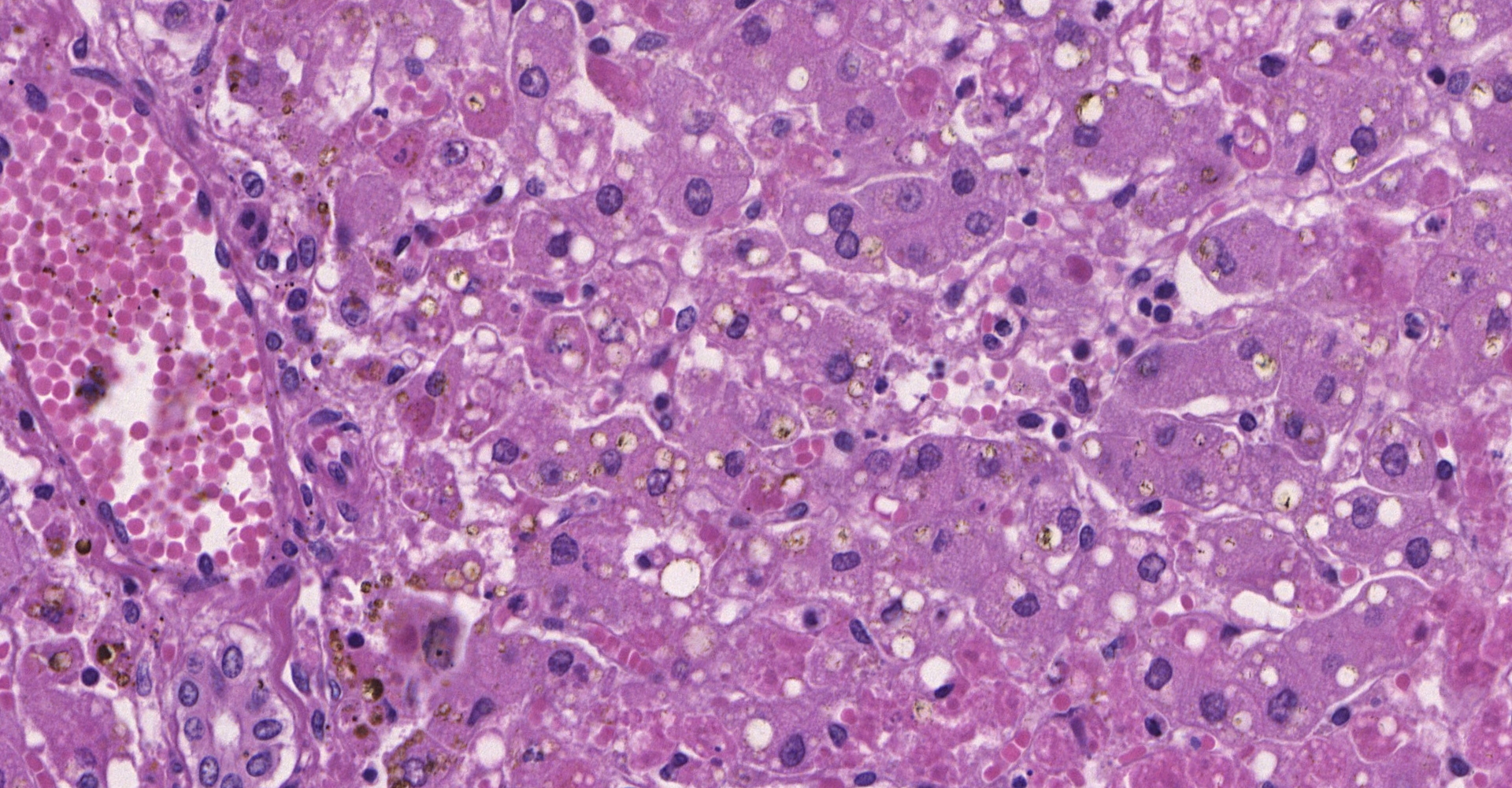
Liver: diffuse severe hepatocyte coagulative necrosis with a few remaining hepatocytes in the portal and central zones with mild intracytoplasmatic vacuoles (compatible with lipidosis). Multifocal mild lymphohistioplasmacytic portal infiltrate with a few neutrophils.
Lungs: mild multifocal areas of alveolar septum thickening with a lymphohistiocytic infiltrate. Mild multifocal edema and hemorrhage within the alveolar lumen. Mild multifocal antracosis.
Kidney: mild multifocal lymphohistioplasmacytic interstitial infiltrate. Few glomeruli with mesangial thickening and mesangial cells proliferation.
Heart: mild focal lymphohistiocytic infiltrate in the myocardium.
Spleen and brain with no significant histological lesion.
Contributor's Morphologic Diagnoses: Liver: diffuse severe acute necrotizing hepatitis with mild lipidosis.
Lungs: mild multifocal lymphohistiocytic interstitial pneumonia with mild multifocal edema and hemorrhage.
Kidney: mild multifocal lymphohistioplasmacytic interstitial nephritis with mild multifocal membranoproliferative glomerulopathy.
Heart:
mild focal lymphohistiocytic myocarditis.
Contributor's Comment:
Yellow fever is an important zoonotic disease related to death of both human and non-human primates. YF is a mosquito-borne hemorrhagic disease transmitted to a susceptible host by Haemagogus sp. and Sabethes sp., or Aedes aegypti, in the sylvatic or urban environments, respectively.9 The disease is considered endemic in the Brazilian Amazon region; however, outbreaks occur in non-endemic areas, as the most recent one which happened mainly in the southeastern region from 2016 to 2018, with more than one thousand human cases and 35.1% case fatality rate.4,6
Neotropical primates are susceptible to YF infection and this disease became an important concern for conservation of free-ranging populations.1,5,20 Monitoring the occurrence of YF in non-human primates is part of the Epizootic Surveillance Program of the Brazilian Ministry of Health acting as an early warning to viral circulation because death of non-human primates usually precedes YV human cases.3,12,17,20 The official diagnosis is made by reference laboratories and it is based on real time RT-PCR and/or immunohistochemistry from samples of liver, spleen, kidney, heart, lungs, and brain.17
Information about microscopic changes associated with YF in non-human primates are limited, including both natural and experimental infection.7,8,12 Even with scarce information, there is evidence of differences in susceptibility for different neotropical primates. Briefly, Alouatta sp. and Callithrix sp. are considered susceptible to YF while Sapajus sp. are resistant.1,9,12,20 Recently, these differences of susceptibility were associated with histopathological patterns of changes, demonstrating that Alouatta sp. develop a severe necrotizing hepatitis while Callithrix sp., a reported susceptible specie, showed minimal hepatic injuries.16 Another recent study demonstrated that Callithrix sp. had less viral load than other primates, such as Alouatta sp. and Sapajus sp. 4
Yellow fever virus affects mainly the liver of infected hosts, causing necrosis/apoptosis of hepatocytes as the most relevant lesion typically affecting primarily midzonal hepatocytes.15 This pattern of distribution is associated with viral tropism and was described in human and non-human primates, including free-ranging naturally infected neotropical primates and experimentally infected Rhesus monkeys.1,15,16,18
Another hepatic injury observed in these cases was lipidosis, which has been reported in both human and neotropical primates.6,15,16,21 Inflammatory infiltrate is usually mild during YF infection, mostly because of the transforming growth factor beta (TGF-β), an inductor of apoptosis that also acts as an anti-inflammatory cytokine.12,15,20 Acute tubular necrosis causing renal failure is a major complication associated with human fatal cases of yellow fever, however this is not an important lesion associated with primate infection.11,12,16
The case reported here was part of a recent study16 that demonstrated histopathological patters in different species of neotropical primates naturally infected with the yellow fever virus. The pattern observed in the liver of Alouatta sp. is associated with severe hepatic failure caused by loss of most of the hepatocytes. Histologically, primates of this specie showed severe hepatocyte necrosis, mainly of the midzonal region expanding to the portal and central regions of the hepatic lobule.
Contributing Institution:
Escola de Veterinária, Universidade
Federal de Minas Gerais ? www.vet.ufmg.br
JPC Diagnosis:
Liver: Hepatocellular degeneration and necrosis, centrilobular and midzonal, diffuse, severe, with Councilman bodies and intracytoplasmic lipid.
JPC Comment:
The contributor provides an excellent overview of yellow fever, the most severe of five arboviruses that have emerged or continually reemerge over recent decades in the Americas. The other four arboviruses include dengue, West Nile, Chikungunya, and Zika.13
Yellow fever is flavivirus that likely originated in Africa and was imported to the Americas during the 1600s along trans-Atlantic trade routes.13,14,7 The virus was responsible for hundreds of thousands of deaths throughout the 18th and 19th centuries, including 10% of the population of Philadelphia in 1793, the United States capital at the time. Nearly a century later, Cuban epidemiologist Carlos Finlay first proposed the disease was mosquito-borne, which was verified by US Army physician Walter Reed and the Yellow Fever Commission in 1900. Subsequent mosquito control and sanitation efforts essentially eliminated yellow fever from the United States and non-endemic areas of the Americas.13
In 1937, virologist Max Theiler developed a vaccine still in use today that provides lifetime immunity in up to 99% of vaccinated individuals.13,19 Theiler was awarded the 1951 Nobel Prize in Physiology or Medicine, the first and only Nobel Prize given for the development of a vaccine. Interestingly, Max Theiler's father was Sir Arthur Theiler, a Swiss veterinarian and researcher who also made significant contributions in the realm of scientific discovery.19
Yellow fever was substantially suppressed though the combination of vector control, sanitation, and vaccination campaigns; however, the virus continues to persist due to a sylvatic transmission cycle maintained between forest mosquitos (Haemagogus sp. and Sabethes sp.) and susceptible non-human primates. Periodically, the virus becomes reintroduced into the human population (i.e. urban cycle) and is readily transmitted by the vector Aedes aegypti, resulting in outbreaks. These outbreaks occur in parts of Africa and Central and South America, resulting in an estimated 84,000 to 170,000 severe cases and 29,000 to 60,000 related human deaths per year.13
In South America, outbreaks occur every 5 to 10 years. Human cases are preceded by a rise in nonhuman primate cases, which is due to an increased susceptibility of the nonhuman primate population.14 Both old and new world primates are susceptible to yellow fever; however, new world primates are particularly susceptible, especially howler monkeys (Alouatta sp.).14 In this context, highly susceptible nonhuman primate species likely play an important role in amplifying the virus and therefore infecting large numbers of invertebrate vectors. Therefore, monitoring the occurrence of yellow fever in nonhuman primates serves as an early warning to viral circulation in a given area, which facilitates identification of exposed human populations and use of targeted vaccination programs, as a preventive measure.14
In contrast, animals that develop a mild yellow fever virus infection and long-lasting immunity, including resistant new world primates, probably suppresses circulation of yellow fever virus between outbreaks.14
As noted by the contributor, yellow fever is classified as a viral hemorrhagic fever. Although much attention has been paid to the hepatic pathology of viral hemorrhagic fevers, and abnormal liver function tests are common, clinically significant liver disease and death from liver failure are rare except in yellow fever. Disseminated intravascular coagulation and depletion of vitamin-K dependent clotting factors due to decreased production as the result of hepatic necrosis contribute to the recognized bleeding diathesis.22
Apoptotic hepatocytes were first clearly described in yellow fever by William Thomas Councilman and therefore have often been referred to as "Councilman bodies". Although apoptosis occurs in many forms of liver disease, by convention this eponym is restricted to use in regard to yellow fever.9
References:
1. Almeida MAB. Yellow fever. In: Fuentes A, The international encyclopedia of primatology. 1th ed. Hoboken, NJ: John Wiley & Sons, Inc; 2017:1-2.
2. Almeida MAB, Santos E, Cardoso JC, et al. Yellow fever outbreak affecting Alouatta populations in southern Brazil (Rio Grande do Sul State), 2008?2009. Am J Promatol. 2012;74(1):68-76.
3. Bacha HA, Johanson GH. Yellow fever. Rev Assoc Med Bras. 2017;63(4):291-292.
4. Cunha MS, Costa AC, Fernandes NCCA, et al. Epizootics due to Yellow Fever Virus in São Paulo State, Brazil: viral dissemination to new areas (2016?2017). Scientific Reports. 2019;9(1):1-13.
5. Dietz JM, Hankerson SJ, Alexandre BR, et al. Yellow fever in Brazil threatens successful recovery of endangered golden lion tamarins. Sci Rep. 2019;9:12926.
6. Duarte-Neto AN, Cunha MP, Marcilio I, et al. Yellow fever and orthotopic liver transplantation: new insights from the autopsy room for an old but re-emerging disease. Histopathology. 2019;75:638-648.
7. Engelmann F, Josset L, Girke T, et al. Pathophysiologic and trancriptomic analyses of viscerotropic yellow fever in a rhesus macaque model. PLOS Negl Trop Dis. 2014;8(11):e3295.
8. Fernandes NCCA, Cunha MS, Guerra JM, et al. Outbreak of yellow fever among nonhuman primates, Espírito Santo, Brazil, 2017. Emerg Infect Dis. 2017; 23(12):2038-2041.
9. Gill RM, Kakar S. Liver and gallbladder. In: Kumar V, Abbas AK, Aster JC, eds. Robbins and Cotran Pathologic Basis of Disease. 9th ed. Philadelphia, PA: Elsevier; 2021:825.
10. Litvoc MN, Novaes CTG, Lopes MIB. Yellow fever. Rev Assoc Med Bras. 2018;64(2):106-113.
11. Monath TP. Yellow fever: an update. Lancet Infect Dis. 2001;1(1):11-20.
12. Monath TP, Vasconcelos PFC. Yellow fever. J Clin Virol. 2015;64:160-173.
13. Norrby E. Yellow fever and Max Theiler: the only Nobel Prize for a virus vaccine. J Exp Med. 2007;204(12):2779-2784.
14. Paules CI, Fauci AS. Yellow Fever - Once Again on the Radar Screen in the Americas. N Engl J Med. 2017;376(15):1397-1399.
15. Quaresma JAS, Pagliari C, Medeiros DBA, et al. Immunity and immune response, pathology and pathologic changes: progress and challenges in immunopathology of yellow fever. Rev Med Virol. 2013;23(5):305-318.
16. Santos DO, Oliveira AR, Lucena FP et al. Histopathologic patterns and susceptibility of neotropical primates naturally infected with yellow fever virus. Vet Pathol, 2020. In press.
17. Silva NIO, Sacchetto L, Rezende IM, et al. Recent sylvatic yellow fever virus transmission in Brazil: the news from an old disease. Virol J. 2020;17(9):1-12.
18. Stokes A, Bauer JH, Hudson NP. The transmission of yellow fever to macacus rhesus: preliminary note. J Am Med Assoc, 1928;90(4):253-254.
19. Tan SY, Pettigrew K. Max Theiler (1899-1972): Creator of the yellow fever vaccine. Singapore Med J. 2017;58(4):223-224.
20. Vasconcelos PFC. Febre amarela. Rev Soc Bras Med Trop. 2003;36(2):275-293.
21. Vieira V, Pacheco L, Demetrio L, et al. Liver transplantation for acute liver failure due to yellow fever: a case report. Transplant Proc. 2019;51:1625-1628.
22. Wachtman L, Mansfield J. Viral diseases of nonhuman primates. In. Abee CR, Mansfield K, Tardif S, Morris T. eds. Nonhuman primates in biomedical research. Vol 2. 2nd ed. Oxford, UK: Elsevier; 2012:50-51.