Joint Pathology Center
Veterinary Pathology Services
Wednesday Slide Conference
2017-2018
Conference 24
May 2nd, 2018
CASE I: AP17-0483 (JPC 4101485).
Signalment: 1-year-old, female, Zebrafish (Danio rerio), pisces.
History: A subset of TP53 mutant zebrafish from the colony were found with significantly distended coelomic cavities, which necessitated their removals from the study.
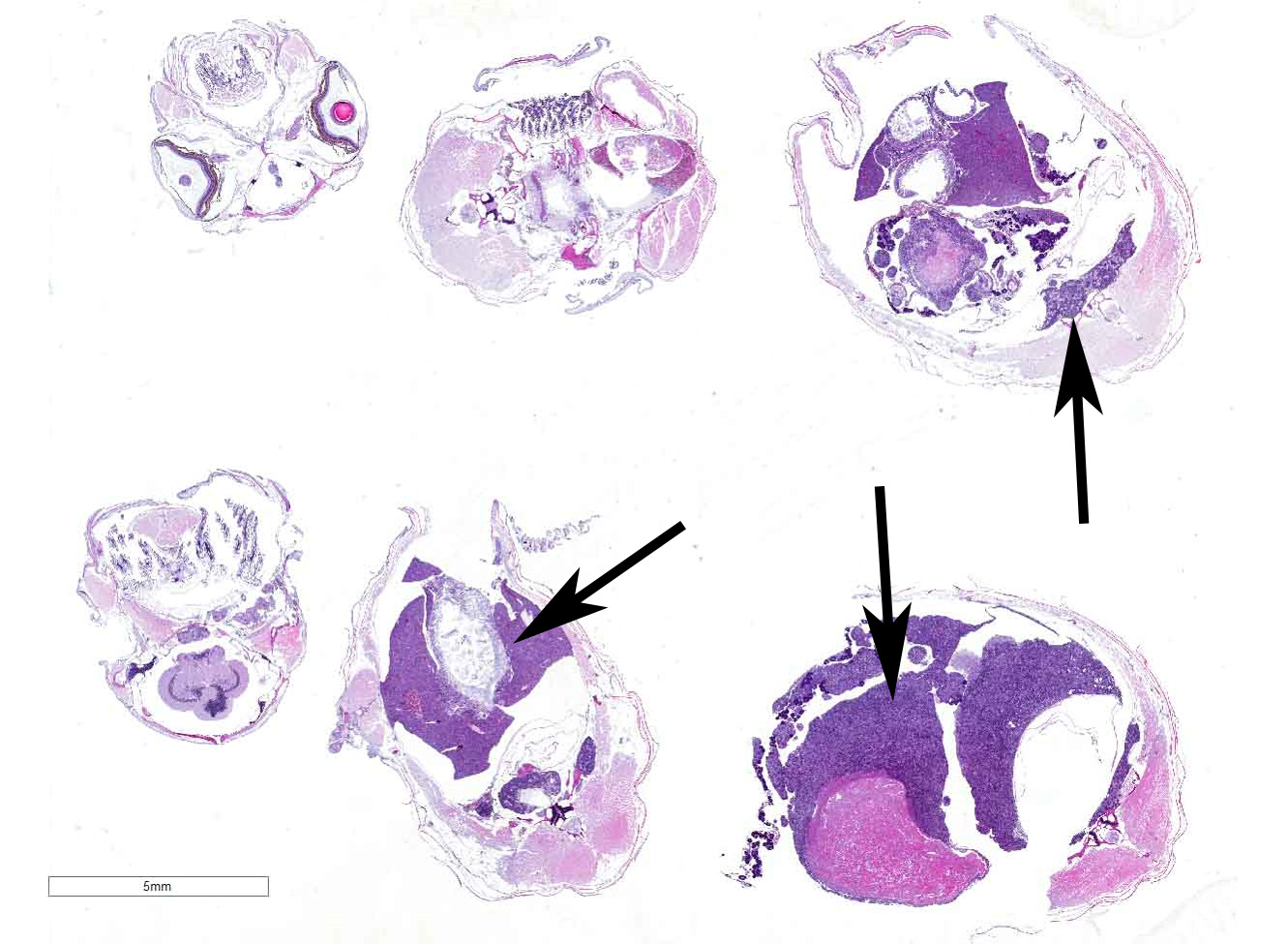
Gross Pathology: The coelomic cavities of the affected fish were grossly distended and, upon opening, contained variably sized, firm, tan masses that compressed and effaced organs and infiltrated the body wall.
Laboratory Results (clinical pathology, microbiology, PCR, ELISA, etc.): None provided.
Microscopic Description:
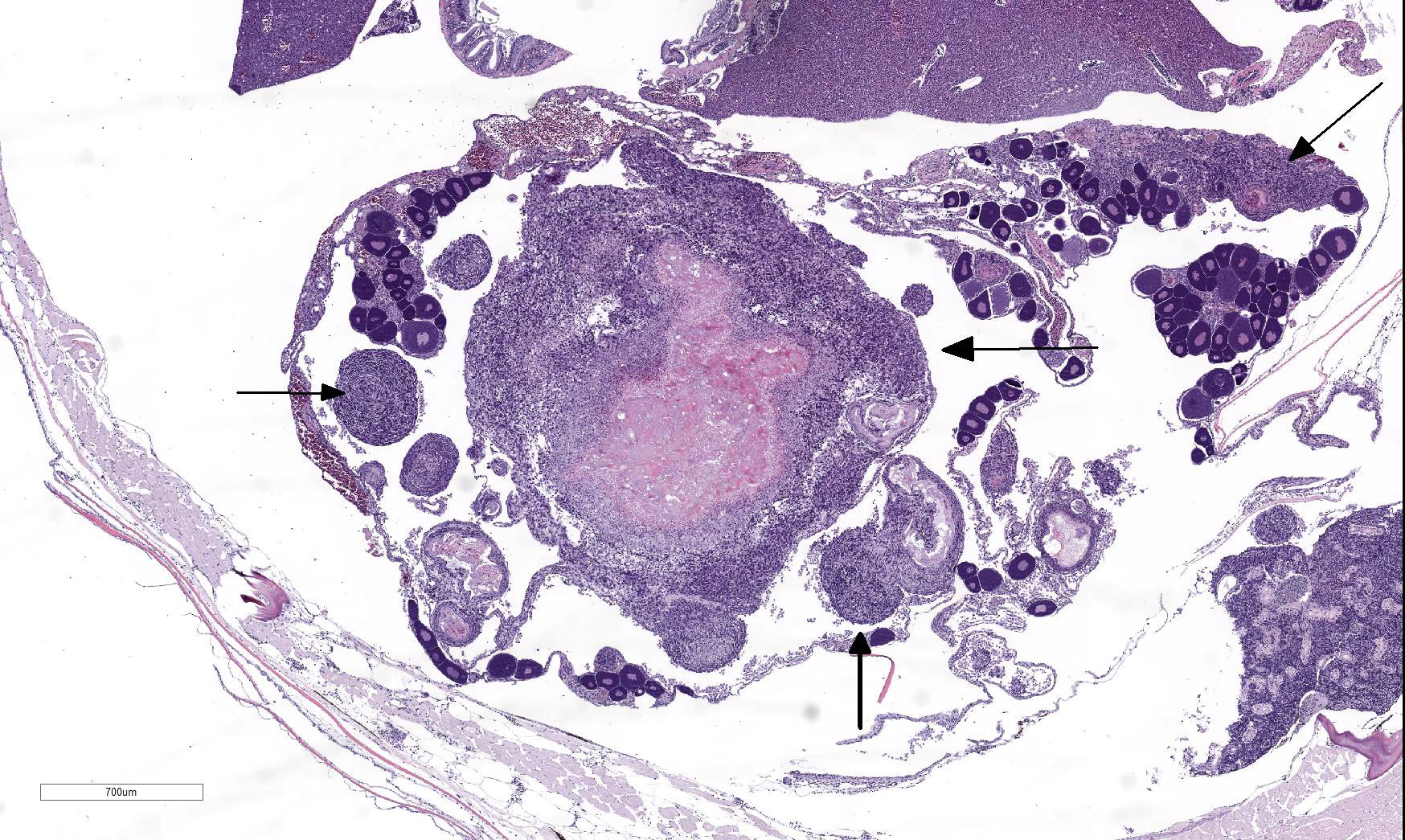
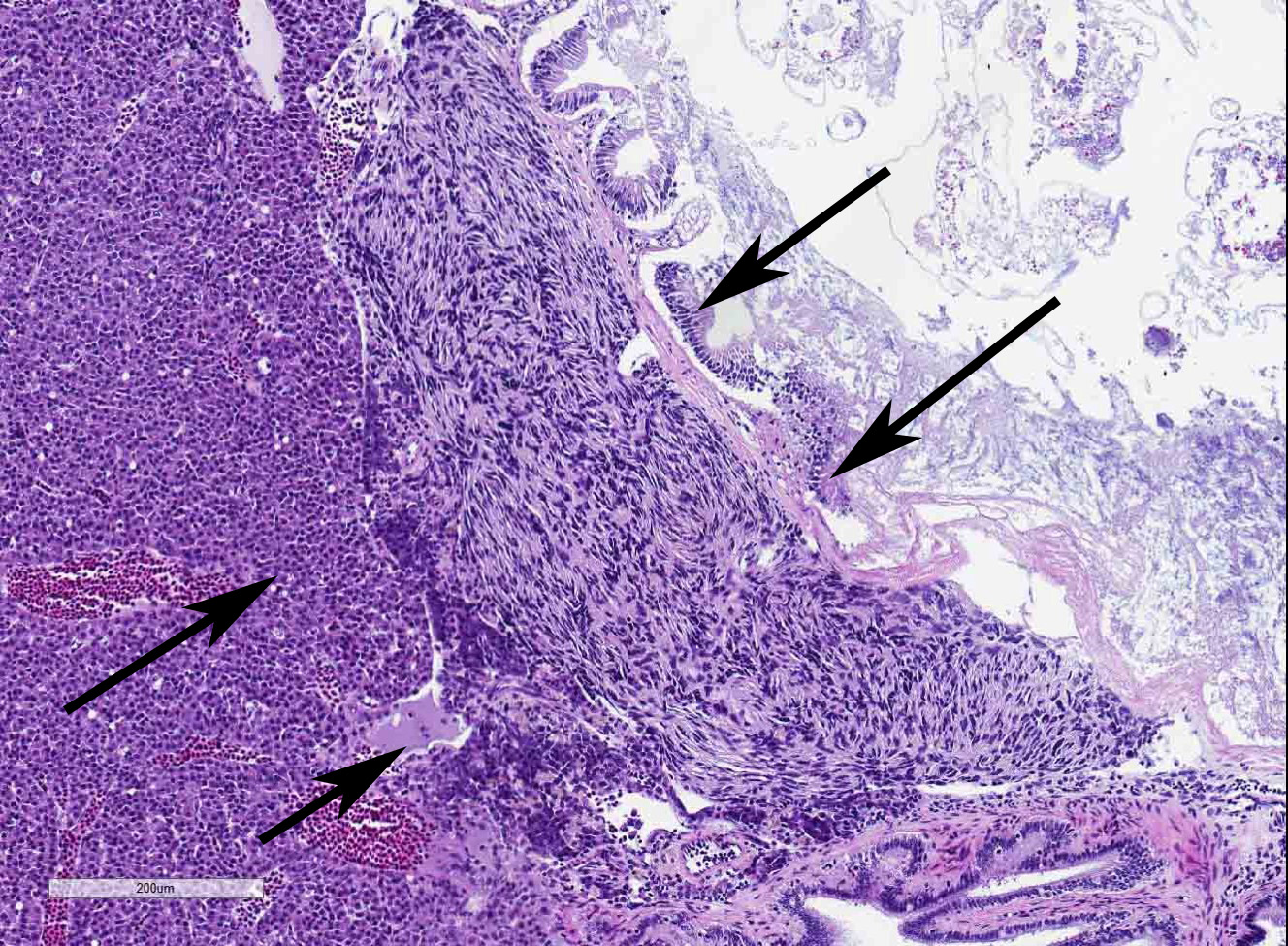
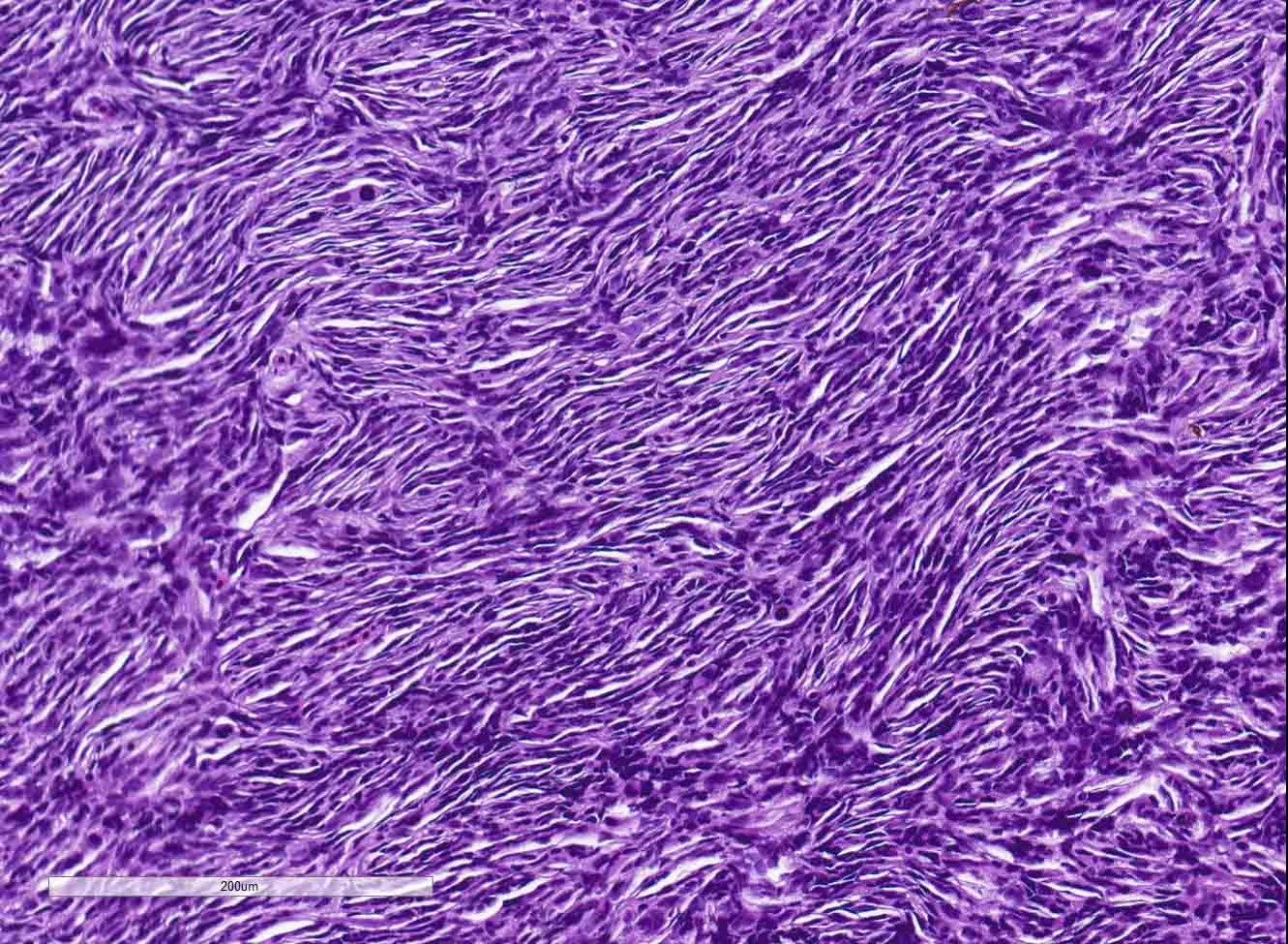
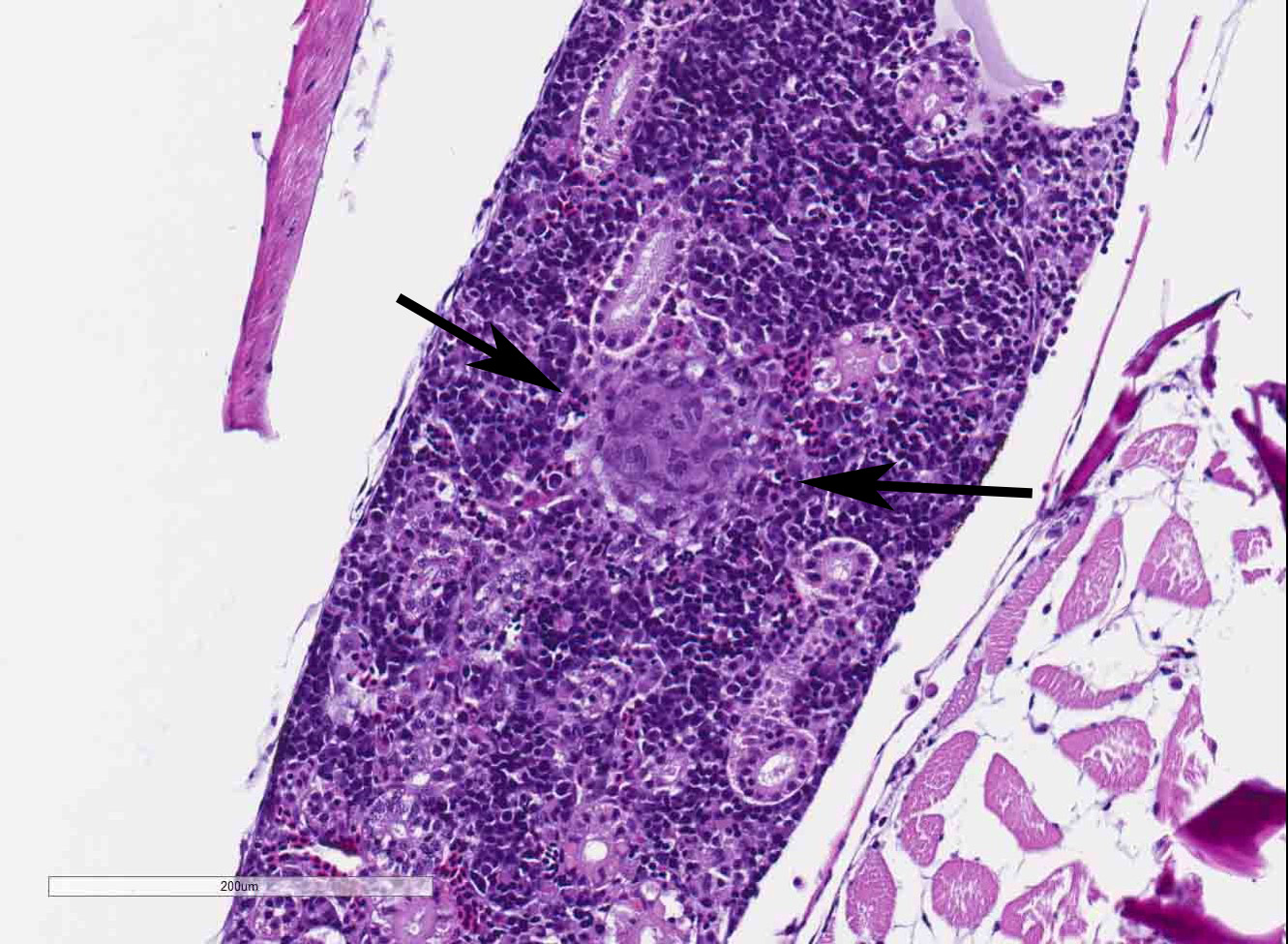
The coelomic cavity is expanded by a large, unencapsulated neoplasm that is arranged in variably sized and tightly arranged bundles with regions of indistinct nuclear palisading and situated within an eosinophilic, collagenous matrix. Neoplastic cells have poorly defined cellular borders, are spindle-shaped and have moderate amounts of cytoplasm. Nuclei are oval to fusiform in shape and have either reticulated or hyperchromatic patterning with 1 to 2 nucleoli that are variable in prominence based on the orientation of the tumor cells. There is marked anisocytosis and anisokaryosis with atypical giant cells scattered throughout the tumor. Mitoses range from 0-4 per 400x/hpf. There are varying combinations and concentrations of small lymphocytes, heterophils and histiocytes that are distributed throughout the neoplasm as well as randomly scattered foci of necrosis that often coalesce together. There are also rare, small sized granulomas that are centered on clear spaces as well as entrapped remnants of ovarian tissues in different states of degeneration. Neoplastic cells contact and extend into portions of the body wall. Metastasis is not observed.
Contributor’s Morphologic Diagnosis:
Coelomic cavity: Malignant peripheral nerve sheath tumor, Zebrafish
Contributor’s Comment: The tumors that were observed in the Tp53-mutant zebrafish are classified as malignant peripheral nerve sheath tumors (MPNSTs) based on cellular pleomorphism, invasiveness, the presence of necrosis, and, most importantly, the clinical course of the disease that was documented for this colony. In humans and animals peripheral nerve sheath tumors (PNSTs) may arise throughout the body and are largely defined by their morphologic patterning and include neurofibromas, schwannomas and perineurinomas. Further subdivision into benign versus malignant tumors is currently based on the degree of cellular differentiation, mitotic count, presence of necrosis and clinical staging. Schwann cells are the putative cells of origin for most PNSTs. Benign PNSTs most consistently express S100, which is frequently used to distinguish them from other neuroepithelial and mesenchymal neoplasms with similar morphologies.3,5,9 PNSTs are not currently documented in zebrafish outside of experimental manipulations affecting Tp53 status or ribosomal protein genes, although viral infection resulting in neurofibromatosis has been described in damselfish.1,7,10 One study has confirmed that sarcomas arising in laboratory zebrafish, which had mutations of both Tp53 and Brca2, are derived from Schwann cell populations as assessed by the positive labeling of the tumor cells with anti-CD57 and anti-S100 antibodies.13
The zebrafish has been an important animal model for studying human MPNSTs, especially in the context of hereditary cancer syndromes, because t mutated zebrafish MPNSTs often have comparable genetic alterations as human tumors.12,14 Since many of the functions of P53 are conserved the spectrum of tumors that may arise from perturbations to P53-mediated cellular processes frequently result in lymphomas, leukemias, sarcomas and carcinomas in both humans and animals. However, there are often species specific differences in the frequencies of certain tumors, such as the predisposition for zebrafish to form malignant rather than benign PNSTs.6,8,12 These differences suggest that a combination of time and environmental dependent factors are important for the types of P53 dependent tumors that develop and their biological behaviors.2 While these differences in biology between humans and animal species exist, these dissimilarities are being used to further understand the essential genetic and environmental events that are important for the biology of PNSTs.
JPC Diagnosis: 1. Omentum, ovary, pancreas: Malignant peripheral nerve sheath tumor, Zebrafish (Danio rerio), pisces.
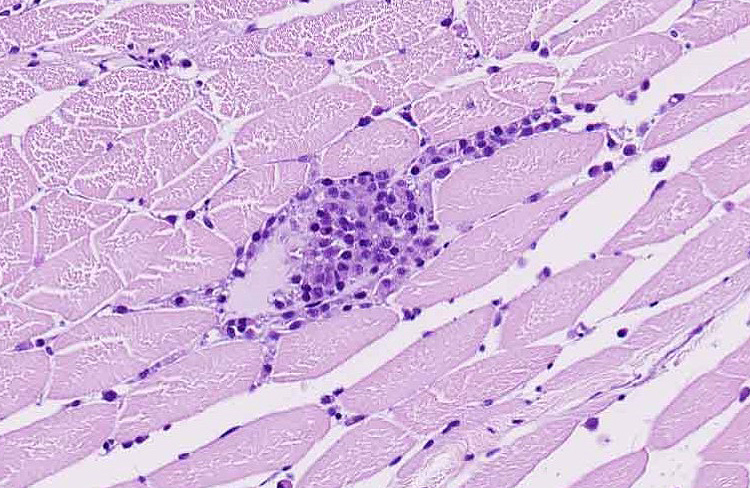
- Kidney, ovaries, omentum: Granulomas, multiple.
Conference Comment: In humans, malignant peripheral nerve sheath tumors (MPNSTs) are rare but devastating tumors, asthey are aggressive and have high rates of relapse following chemotherapy. In approximately half of cases, MPNSTs occur in association with neurofibromatosis type I which results from loss of function mutations to the tumor suppressor neurofibromin. Neurofibromatosis type I, an autosomal dominant condition, is the most common human syndrome which predisposes to cancer. Clinical signs include: cutaneous hyperpigmentation (known as “café-au-lait spots”) and multiple cutaneous neurofibromas which can transform into MPNSTs.
In addition to NF-1, TP53 mutations have also been associated with development of MPNSTs – this is where zebrafish are particularly useful.4 Historically, human cancer studies utilized rodent models. However, disadvantages such as increased time for tumor development and cost encouraged identification of a new model. Zebrafish are more economic and undergo neoplastic transformation and growth quicker. Most importantly, many aspects of carcinogenesis in humans are similarly expressed in zebrafish.
Several methods are used to generate effective models for research including: chemical carcinogenesis, forward or reverse genetics screens, transgenic models, and xenotransplantation in embryos.11 Currently, zebrafish are used as models for the following types of cancer:
Table 1: Zebrafish models of cancer11
|
Tissue of origin |
Cancer model |
|
Cutaneous neoplasia |
Benign nevus; melanoma; papilloma; epidermal tumors |
|
Muscular, adipocytic, and vascular neoplasia |
Embryonal rhabdomyosarcoma; rhabdomyosarcoma; liposarcoma; hemangiosarcoma |
|
Intestinal, pancreatic, and hepatic neoplasia |
Intestinal adenoma; malignant intestinal tumors; pancreatic acinar cell adenoma; pancreatic carcinoma; hepatoma; hepatocellular carcinoma |
|
Hematopoietic, lymphoid, and small round blue cell neoplasia |
T-cell leukemia; T-cell or B-cell acute lymphocytic leukemia; T-cell lymphoblastic lymphoma; acute myeloid leukemia; myeloproliferative neoplasm; small round blue cell tumors |
|
Neural and neuroendocrine neoplasia |
MPNST; neuroblastoma; neuroepithelioma; pituitary corticotroph adenoma; pancreatic neuroendocrine carcinoma |
Peripheral nerve sheath tumors can be further differentiated as benign and malignant Schwannoma, neurofibroma, or neurofibrosarcoma. In most cases, the Schwann cell likely the progenitor cell. A common rule out in fish is chromatophoroma (pigment cell tumors). The bi-color damselfish was once thought to be a naturally-occurring model of neurofibromas before it was discovered they were virally induced.10
During the conference, the moderator pointed out that there is increased hematopoietic tissue and vacuolated tubular epithelial cells within the anterior kidney, multiple granulomas, and histiocytes focally within skeletal muscle. Conference participants discussed what constitutes a “malignant” PNST and concluded that invasiveness is a key indicator but that spread to other organs via blood or lymphatics is not required.
Contributing Institution:St Jude Children’s Research Hospital
Department of Pathology
MS 250, Room 5031
262 Danny Thomas Place
Memphis, TN, 38105-3678
https://www.stjude.org/research/departments-divisions/pathology.html
References:
- Amsterdam A, Sadler KC, Lai K, Farrington S, Bronson RT, Lees JA, et al.: Many Ribosomal Protein Genes Are Cancer Genes in Zebrafish. PLOS Biology 2004:2(5):
- Berghmans S, Murphey RD, Wienholds E, Neuberg D, Kutok JL, Fletcher CDM, et al.: tp53 mutant zebrafish develop malignant peripheral nerve sheath tumors. Proceedings of the National Academy of Sciences of the United States of America 2005:102(2):407-412.
- Cantile C, Youssef S: Chapter 4 - Nervous System IN: Maxie, M. Grant Jubb, Kennedy & Palmer's Pathology of Domestic Animals: Volume 1 (Sixth Edition): W.B. Saunders; 2016: 250-406.
- Farid M, Demicco EG, Garcia R, Ahn L, et al. Malignant peripheral nerve sheath tumors. The Oncologist. 2014; 19(2):193-201.
- Louis DN, Ohgaki, H., Wiestler, O.D., Cavenee, W.K.: WHO Classification of Tumours of the Central Nervous System, Fourth Edition. IARC WHO Classification of Tumours, No 1
- Lozano G: Mouse Models of p53 Functions. Cold Spring Harbor Perspectives in Biology 2010:2(4):
- MacInnes AW, Amsterdam A, Whittaker CA, Hopkins N, Lees JA: Loss of p53 synthesis in zebrafish tumors with ribosomal protein gene mutations. Proceedings of the National Academy of Sciences 2008:105(30):10408-10413.
- Olivier M, Hollstein M, Hainaut P: TP53 Mutations in Human Cancers: Origins, Consequences, and Clinical Use. Cold Spring Harbor Perspectives in Biology 2010:2(1):
- Rodriguez FJ, Folpe AL, Giannini C, Perry A: Pathology of Peripheral Nerve Sheath Tumors: Diagnostic Overview and Update on Selected Diagnostic Problems. Acta neuropathologica 2012:123(3):295-319.
- Schmale MC, Gibbs PDL, Campbell CE: A virus-like agent associated with neurofibromatosis in damselfish. Diseases of Aquatic Organisms 2002:49(2):107-115.
- Shive HR. Zebrafish models for human cancer. Vet Pathol. 2013; 50(3):468-482.
- Storer NY, Zon LI: Zebrafish Models of p53 Functions. Cold Spring Harbor Perspectives in Biology 2010:2(8).
- White LA, Sexton JM, Shive HR: Histologic and Immunohistochemical Analyses of Soft Tissue Sarcomas From brca2-Mutant/tp53-Mutant Zebrafish Are Consistent With Neural Crest (Schwann Cell) Origin. Veterinary Pathology 2017:54(2):320-327.
- Zhang G, Hoersch S, Amsterdam A, Whittaker CA, Lees JA, Hopkins N: Highly aneuploid zebrafish malignant peripheral nerve sheath tumors have genetic alterations similar to human cancers. Proceedings of the National Academy of Sciences 2010:107(39):16940-16945.