Signalment:
1-year-old intact male Romney sheep (
Ovis aries).This sheep was one of several rams purchased 10 days earlier from the sale yards for a research trial. The animals had been transported for approximately 2 hours by truck to the sale yards and had been drenched with an anthelmintic on two occasions during the 10 days since purchase. This animal was found dead in its paddock without showing any previous signs of illness. The vaccination history was unknown.
Gross Description:
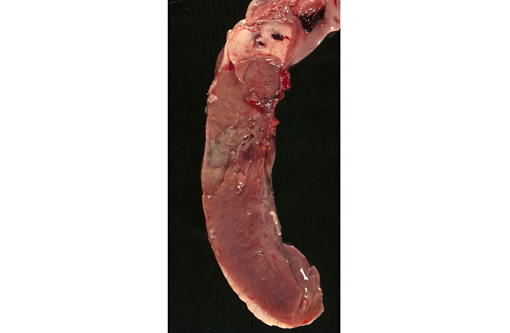
The ram was in good physical condition but was relatively small for its age, weighing 36.5kg. Although the abdomen was distended and blood-stained froth was exuding from the nostrils, there was little evidence of post-mortem decomposition at the time of examination. The lungs were diffusely red and edematous and contained multiple 2-3 mm dark red foci. The thoracic cavity contained approximately 1.5 liters of red fluid. Clear, yellow fluid was also present in the pericardial sac and the epicardial surface of the heart was almost completely covered by a thick, loosely adherent sheet of fibrin. On cut surface, the myocardium contained several sharply demarcated, irregular 2-3 cm diameter foci which varied from pale tan to grey/green, and often had a red margin. One focus involved the interventricular septum and extended to the luminal surface of the right ventricle, where it was covered by a fibrin thrombus.Â
Histopathologic Description:
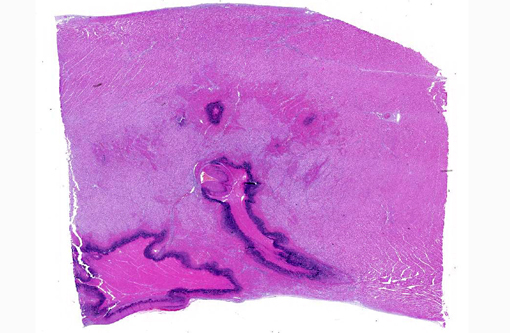
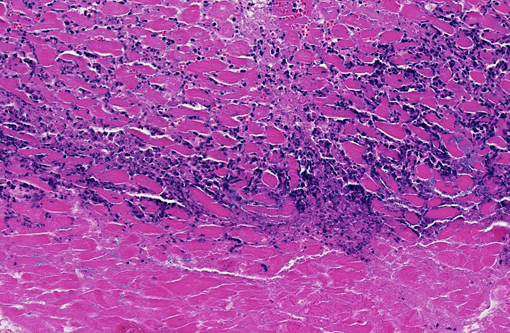
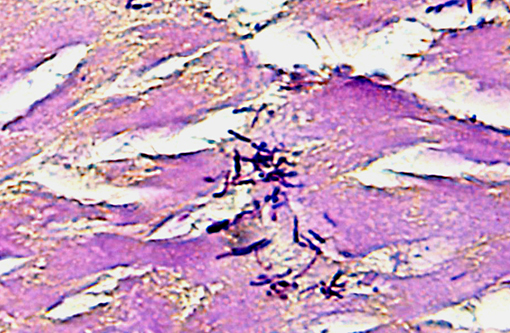
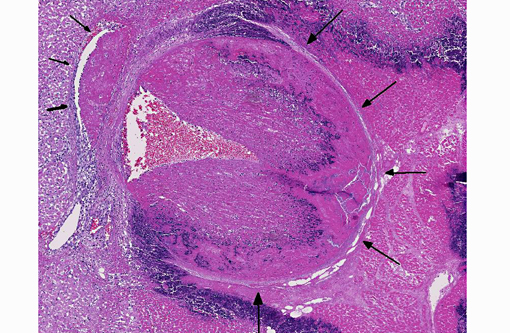
The focal lesions observed grossly in the heart consisted of acute coagulation necrosis surrounded by a thick layer of degenerate neutrophils. Within the necrotic areas, the interstitium was expanded with fibrinous exudate and many interstitial blood vessels and lymphatics contained fibrin thrombi. Large numbers of long, rod-shaped, gram-positive bacteria were present in necrotic areas, particularly near the margins. In surrounding areas of myocardium there was variable acute necrosis of myocardial fibers, neutrophilic interstitial infiltration and edema. The epicardium in some areas was markedly thickened with a layer of fibrin and moderate to large numbers of neutrophils. Necrotic foci that extended to the endocardial surface were covered with a layer of fibrin containing degenerate neutrophils.
Immunohistochemistry for
C. chauvoei, C. septicum, C. novyi and
C. sordelli performed at CAHFSL, San Bernardino (courtesy of Dr. F. Uzal) revealed organisms reacting positively for both
C. chauvoei and
C. septicum.
In the lungs, there was diffuse congestion of alveolar capillaries and proteinaceous alveolar oedema. There was also multifocal intra-alveolar haemorrhage and fibrinous exudation. Moderate numbers of neutrophils were present in the interstitium and in many alveoli.Â
Morphologic Diagnosis:
1. Acute multifocal necrotising myocarditis.
2. Acute diffuse fibrinous epicarditis
Condition:
Clostridial myocarditis
Contributor Comment:
Clostridial myocarditis is a rare disease of sheep. To our knowledge, it has only been reported once previously (in Australia)(2) and is probably analogous to the myocardial form of "blackleg" in cattle, which is caused by
Clostridium chauvoei.(3) No lesions were present in skeletal muscles of the ram, supporting primary myocardial involvement. The myocardial lesions were characterized by a greater neutrophilic response than would be expected in clostridial myositis involving skeletal muscles. This was also a feature of lambs and calves in previous reports of clostridial myocarditis.(2,3) An unexpected finding in this case was the presence of both
C. chauvoei and
C. septicum in the myocardial lesions, suggesting that latent spores of both organisms were present in the myocardium.Â
In published reports of clostridial myocarditis in lambs and calves, potential risk factors included lush pasture growth following a period of high rainfall, and stress associated with yarding and management procedures. Both factors existed in the present case. It is possible that the stress involved in recent transport and yarding for drenching caused focal catecholamine-induced myocardial necrosis, allowing germination of latent clostridial spores. Vaccination with a 5 in 1 clostridial vaccine would have been expected to prevent the disease, suggesting that this ram had not been vaccinated.
JPC Diagnosis:
Heart: Myocarditis, necrotizing, multifocal to coalescing, moderate with large colonies of bacilli, necrotizing arteritis and thrombosis.
Conference Comment:
There is moderate slide variation in this case; some sections contain a large arteriole adjacent to a focus of myocardial necrosis which is 80-90% occluded by an organizing fibrin thrombus, while in other sections, necrotizing arteritis and fibrin thrombi are limited to smaller coronary vessels within the endocardium. The presence of these fibrin thrombi supports the interpretation of the well demarcated areas of necrosis as infarcts. Additionally, most sections of myocardium contain rare protozoal cysts packed with numerous bradyzoites (
Sacrocystis sp.), without evidence of associated inflammation; infection with Sarcocystis sp. is a common, incidental finding in the skeletal and cardiac muscle of ruminants.(4) Histochemical staining with a Giemsa as well as a Gram stain highlighted the presence of numerous gram-positive bacilli with multifocal subterminal spores, a characteristic feature of
Clostridium sp.
Members of the genus
Clostridium are spore forming, gram-positive, anaerobic bacilli, whose spores are ubiquitous in soil, highly resistant to environmental changes and disinfectants, and able to persist in the environment for many years. Germination of spores, which are often ingested by ruminants during grazing, requires specific conditions, including low oxygen levels and an alkaline pH; necrotic lesions and penetrating wounds provide appropriate conditions for growth of clostridial spores. Clostridia typically produce disease via production of exotoxins which cause widespread necrosis with edema and extensive gas formation.(1,4,5) See table 1 for an abbreviated list of
Clostridium sp. important in veterinary medicine.
Table 1: Select clostridial diseases in veterinary species.(1,4,5)
| Name | Species |
| C. perfringens type: | |
A
(α toxin) |
- Enterocolitis in many species:
- Foals, piglets, lambs, calves
- Necrotic enteritis- chickens
- Hemorrhagic canine gastroenteritis
- Hemorrhagic bowel syndrome- dairy cattle
- Yellow lamb disease- IV hemolysis (rare)
- Gas gangrene- humans and animals
|
B
(α, β, ε toxins) |
- Enterocolitis in Europe, South Africa, Middle East
- Lamb dysentery; similar condition in neonatal calves
|
C
(α, β toxins) |
Enterocolitis in many species:
- Struck in adult sheep in the UK; similar condition in goats and feedlot cattle
- Necrohemorrhagic enteritis- lambs, calves, piglets, foals
|
D
(α, ε toxins) |
- Enterocolitis:
- overeating disease/pulpy kidney- sheep, goats, cattle
- Focal symmetric encephalomalacia- sheep and (rarely) goats
|
E
(α, ι toxins) |
- Enterotoxemia- calves, lambs, guinea pigs, rabbits
|
| C. haemolyticum | - Bovine bacillary hemoglobinuria (redwater) 2o to fluke migration
|
| C. septicum |
- Necrohemorrhagic abomasitis (braxy)
- Malignant edema
- Postparturient vulvovagnitis and metritis in dairy cattle
|
| C. novyi |
- Hepatic necrosis (black disease) 2o to fluke migration
- Wound infections
|
| C. chauvoei | - Necrotizing myositis (blackleg) in cattle and sheep
|
| C. colinum | - Ulcerative enteritis- quail disease in gallinaceous birds
|
| C. spiroforme | - Spontaneous or antibiotic-induced enterotoxemia in rabbits and rodents
|
| C. difficile | - Spontaneous or antibiotic-induced enterocolitis in hamsters, rabbits, guinea pigs, dogs, swine and horses
|
| C. piliforme |
- Necrotizing enteritis/colitis, hepatitis, myocarditis in many species (Tyzzers disease)
- Obligate intracellular, gram-negative
|
| C. carnis, C. histolyticum | - Wound infections in many species
|
| C. tetani | - Tetanus- muscle spasms, stiff gait, rigid posture, trismus (lockjaw)
|
| C. botulinum | - -�-� Botulism- flaccid paralysis â respiratory paralysis
|
References:
1. Brown CC, Baker DC, Barker IK. Alimentary system. In: Maxie MG, ed. Jubb, Kennedy and Palmers Pathology of Domestic Animals. Vol. 2. 5th ed. Philadelphia, PA: Elsevier; 2007:213-222.
2. Glastonbury JRW, Searson JE, Links IJ, Tuckett LM. Clostridial myocarditis in lambs. Australian Veterinary Journal. 1988;65:208-209.
3. Uzal FA, Paramidani M, Assis R, Morris W, Miyakawa MF. Outbreak of clostridial myocarditis in calves. Vet Rec. 2003;152:134-136.
4. Van Vleet JF, Valentine BA. Muscle and tendon. In: Maxie MG, ed. Jubb, Kennedy and Palmers Pathology of Domestic Animals. Vol. 1. 5th ed. Philadelphia, PA: Elsevier; 2007:259-267.
5. Zachary JF, McGavin MD, eds. Pathologic Basis of Veterinary Disease. 5th ed. St. Louis, MO: Elsevier; 2012:175-175, 192-196, 377-379, 866, 891-898.