Signalment:
Gross Description:
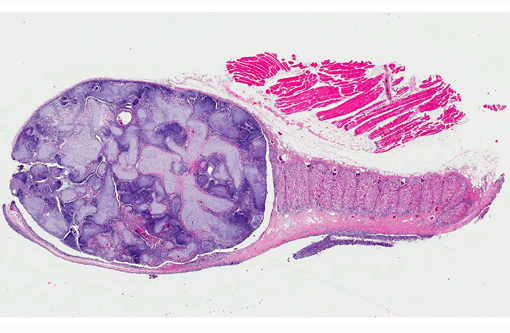
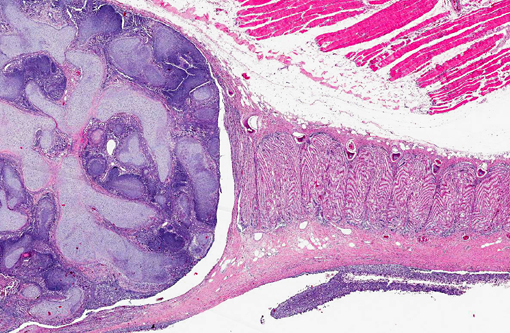
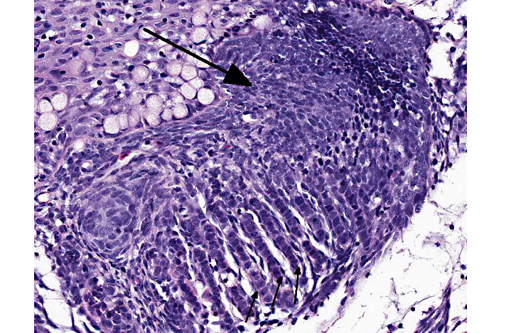
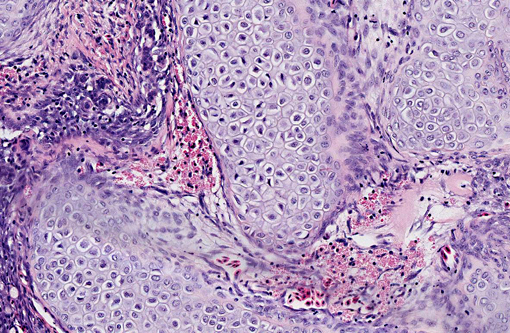
Histopathologic Description:
Morphologic Diagnosis:
Condition:
Contributor Comment:
Branchioblastoma is a tumor of embryologic blast-type cells with mesenchymal and epithelial components. It is histologically considered a benign tumor because it is expansile and often well demarcated. However, the mass can interfere with gaseous exchange in the gill with potentially fatal consequences. Affected fish may present with respiratory distress, an inability to close their mouth or multilobulated mass/masses appearing to originate from a gill arch or the pseudo branch.(3)
Select differentials for a multilobular lesion in the region of the operculum, gill and oral cavity may include granulomatous or inflammatory lesions due to infection by bacteria (eg. Mycobacterium sp.), fungi (eg. Banchiomyces), microsporidia (eg. Loma sp.), myxozoa (eg. Myxobolus sp.), branchial trematodes, and iridovirus (eg. lymphocystis).
Neoplasia in fish is likely multifactorial and has previously been associated with exposure to carcinogens,(6) genetic causes,(5) and retrovirus infection.(1) No underlying cause was identified in this case.
JPC Diagnosis:
Conference Comment:
In addition to branchioblastoma, which can arise from the gill or the pseudobranch and may be spontaneous or carcinogen-induced,(3) conference participants briefly discussed teratoma as a rule-out with similar histological features. Branchioblastoma, as noted by the contributor, is composed of embryologic blast-type cells with mesenchymal and epithelial components; similarly, teratomas are classically defined as having at least two of the three embryonic layers- endoderm, mesoderm, and ectoderm (see WSC 2013-2014, conference 8, case 2). Teratomas occur most frequently in the gonads; however, these tumors can also develop at extragonadal locations, usually along the midline.(2) They are rarely (if ever) reported in fish. The moderator points out that branchioblastoma and teratoma have analogous etiopathogeneses; however, the neoplastic cell types comprising branchioblastomas are appropriate to the anatomic location (i.e. this tumor appears to be attempting to form normal gill tissue in a relatively normal location), while teratomas tend to produce poorly differentiated, disorganized tissue that does not belong (i.e. foci of squamous epithelium, bone and tooth within the gonad). Based on these broad attributes, this neoplasm is most consistent with a branchioblastoma.
References:
1. Coffee L, Casey J, Bowser P. Pathology of tumors in fish associated with retroviruses: a review. Vet Pathol. 2013;50:390-403.
2. Foster RA. Male reproductive system. In: McGavin MD, Zachary JF, eds. Pathologic Basis of Veterinary Disease. 5th ed. St. Louis, MO: Elsevier; 2012:1142-1143.
3. Kn+â-+sel R. Brandes R, Lechleiter S, Schmidt-Posthaus H. Two independent cases of spontaneously occurring branchioblastomas in koi carp (Cyprinus carpio). Vet Pathol. 2007;44:237-239.
4. Mumford S, Heidel J, Smith C, Morrison J, MacConnell B, Blazer V. Fish Histology and Histopathology. US Fish and Wildlife Service, National Conservation Training Center. http://nctc.fws.gov/resources/course-resources/fish-histology/index.html. Accessed April 26, 2014.
5. Shin J, Padmanabhan A, de Groh E, Lee J-S, Haidar S, Dahlberg S, et al. Zebrafish neurofibromatosis type 1 genes have redundant functions in tumorigenesis and embryonic development. Dis Model Mechan. 2012;5:881-894.
6. Williams D, Bailey G, Reddy A, Hendricks J, Oganesian A, Orner G, et al. The rainbow trout (Oncorhynchus mykiss) tumor model: recent applications in low-dose exposures to tumor initiators and promoters. Toxicol Pathol. 2003;31:58-61.
7. Wirgin I, Waldman J. Altered gene expression and genetic damage in North American fish populations. Mutation Res. 1998;399:193-219.