Signalment:
Gross Description:
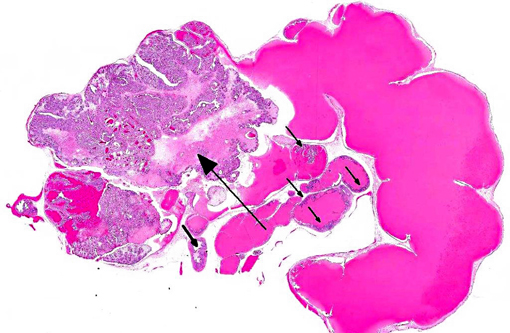
Histopathologic Description:
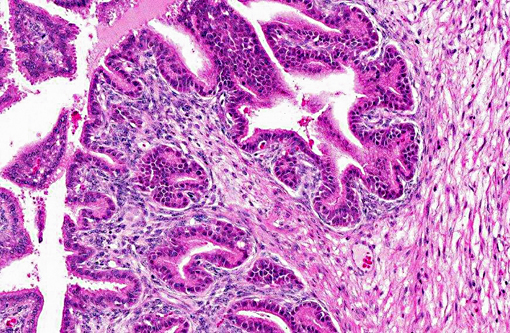
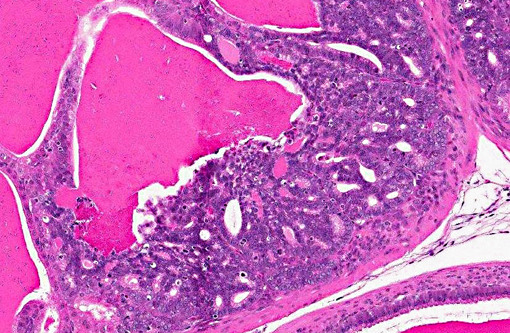
The lumen and ducts are often variably ectatic, lined by attenuated epithelium, and contain homogeneous, brightly eosinophilic, proteinaceous material/colloid with occasional hemorrhage. Multifocally, occasional foci of necrotic debris are noted within the stroma associated with breaching of the lining epithelium into the lumen admixed with eosinophilic debris and hemosiderin-laden macrophages. Multifocally, other areas of the lining epithelium of the seminal vesicle extend as nodular, papillary proliferations into the lumen and have piling of the epithelial cells, and mildly expanded supporting stroma with increased mitoses.Â
Morphologic Diagnosis:
Condition:
Contributor Comment:
Typically, lesions noted in TRAMP mice include the prostate, for which lesions may be assessed using a well-characterized grading scheme.(6) In addition to prostate neoplasms, tumors are also noted in the seminal vesicles.(7)
The normal luminal surface of seminal vesicles in mice is lined by an anastomosing glandular epithelium lined by cuboidal to tall columnar cells forming an intricate arrangement of primary, secondary and tertiary folds. These folds are normally present and should not be confused with hyperplastic changes. Brightly eosinophilic luminal secretions expand the seminal vesicles, resulting in an extended vesicular wall lined by short epithelial papillae, a scanty submucosal layer, and a thin smooth muscle layer.
As in the present case, seminal vesicle tumors in TRAMP mice typically exhibit features of biphasic organization. Although stromal cells proliferate rapidly, they are always lined by a single-layered epithelial component and do not form solid stromal masses such as those seen in uterine stromal tumors. Also, compared to prostatic tumors in TRAMP mice, seminal vesicle tumors display prominent proliferation of stromal cells with frequent mitotic figures lined by bland epithelium, whereas prostatic tumors display epithelial tumor cell proliferation with scant stromal components.(7)
Although the tumors of TRAMP seminal vesicles resemble epithelialstromal tumors found in the breast, prostate, and seminal vesicles in humans, no reports of this type of tumor in any other rodents exist, and these tumors are considered to be TRAMP mouse-specific lesions.
JPC Diagnosis:
Conference Comment:
Accessory genital glands, found along the length of the pelvic urethra in males, have two purposes: providing nutrition and a transport medium for spermatozoa.(1) There are four main male accessory genital glands: the ampullae, seminal vesicles (vesicular glands), prostate and bulbourethral glands (Cowpers glands). Their presence, size, type and number vary among species. The ampullae are paired glands located on the dorsal neck of the bladder. They are composed of secretory alveoli lined by cuboidal to columnar pseudostratified epithelial cells with few basal cells. The seminal vesicles are paired saccular glands that are located between the prostate gland and the ampulla.(1,4) Seminal vesicles consist of lobules of tubuloalveolar glands with pseudostratified epithelium with sparse basal cells, divided by interlobular septa that contains either abundant smooth muscle (in ruminants) or connective tissue with little smooth muscle (stallions and boars).1 The prostate, which has a higher proportion of connective tissue than the seminal vesicles, originates at the urinary bladder and surrounds the urethra.(4) The prostate is composed of either the corpus (body) of compact prostatic tissue located external to the urethra, and/or the disseminated prostate, characterized by diffuse prostatic tissue found along the length of the pelvic urthera.(4) Prostatic glands are classified as compound tubuloacinar glands, and are lined predominantly by pseudostratified and occasionally low cuboidal or squamous epithelium. Bulbourethral glands are paired, dense glands containing abundant fibrous connective tissue located below the prostate and are composed of compound tubuloalveolar glands.(4)
The moderator provides the following chart summarizing the presence and relative size of accessory genital glands in various species:
(Cowpers) |
||||
| Body | Disseminate | |||
| Bull | ++ | + | + | + |
| Ram | + | - | + | ++ |
| Boar | +++ | + | + | +++ |
| Stallion | +++ | ++ | -(+) | + |
| Dog | - | ++ | - | - |
| Cat | - | ++ | - | ++ |
| Rodent | +++ | ++ | - | ++ |
| Human | + | ++ | - | + |
References:
2. Foster RA. Male reproductive system. In: McGavin MD, Zachary JF, eds. Pathologic Basis of Veterinary Disease. 5th ed. St Louis, MO: Elsevier Mosby; 2012:1128.Â
3. Gupta S, Hastak K, Ahmad N, et al. Inhibition of prostate carcinogenesis in TRAMP mice by oral infusion of green tea polyphenols. Proc Natl Acad Sci USA. 2001;98:10350-10355.
4. Johnson L, Welsh Jr TH, Curley Jr KO, Johnston CE. Anatomy and physiology of the male reproductive system and potential targets of toxicants. In: McQueen CA, ed. Comprehensive Toxicology. Oxford, UK: Elsevier. 2010:5-59. Accessed online at http://www.sciencedirect.com/science/article/pii/B9780080468846011027 on 3 February 2013.Â
5. Nyska A, Suttie A, Bakshi S, et al. Slowing tumorigenic progression in TRAMP mice and prostatic carcinoma cell lines using natural anti-oxidant from spinach, NAO--a comparative study of three anti-oxidants. Toxicol Pathol. 2003;31:39-51.
6. Suttie A, Nyska A, Haseman JK, et al. A grading scheme for the assessment of proliferative lesions of the mouse prostate in the TRAMP model. Toxicol Pathol. 2003;31:31-38.
7. Tani Y, Suttie A, Flake GP, et al. Epithelial-stromal tumor of the seminal vesicles in the transgenic adenocarcinoma mouse prostate model. Vet Pathol. 2005;42:306-314.
8. Wechter WJ, Leipold DD, Murray ED, Jr., et al. E-7869 (R-flurbiprofen) inhibits progression of prostate cancer in the TRAMP mouse. Cancer Res. 2000;60:2203-2208.