CASE IV: 18042E (JPC 4136503)
Signalment:
4-year-old male, Red-Bellied Short-Necked turtle (Emydura Subglobosa)
History:
The turtle was housed with 9 other turtles, in an appropriately sized tank. The animal presented to the rDVM with a history of progressive ascites and edema. A treatment of ceftazidime was initiated after thoracic radiographs were suspicious for pneumonia. The animal improved briefly, but then clinical signs recurred. At the submitting institution's exotic clinic, examination revealed ecchymoses, edema, ascites, and loss of keratin/epidermis on plastron. Due to poor prognosis, the owner elected euthanasia.
Gross Pathology:
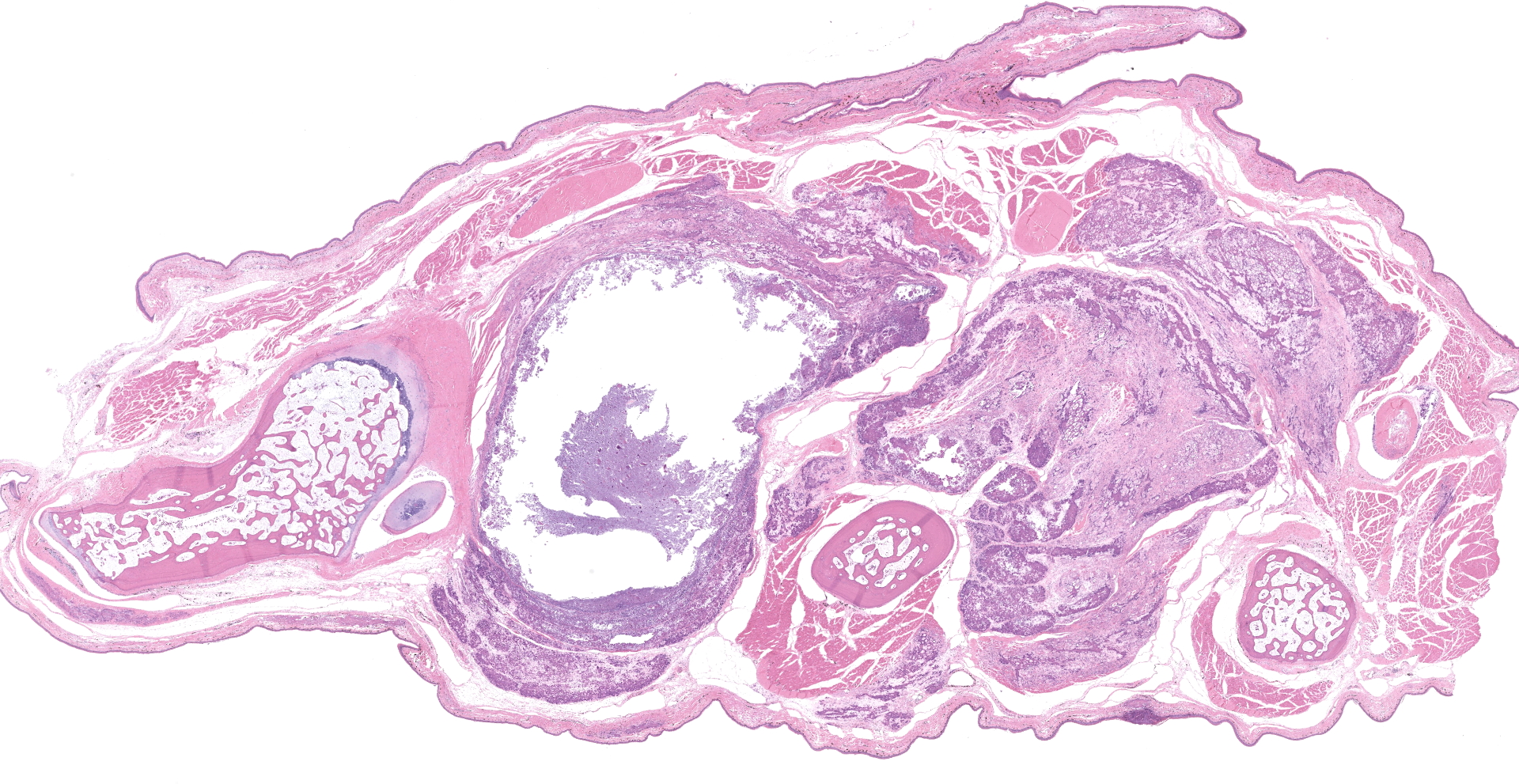
The turtle was in extremely poor nutritional status (no fat stores appreciable within the carcass). The plastron was devoid of epithelium, and there was a focal 0.5 cm by 0.5 cm indentation in the left cranial portion of the plastron. The carapace had multifocal to coalescing ulcerations. Upon opening the carcass, bright yellow fluid spilled out, and when the plastron was removed, there was clear fluid within the coelomic cavity. All tissues were very pale and contained little blood (anemia). All muscles within the coelom had a thick layer of yellow firm pitted tissue (possible fibrin or myonecrosis). The neck muscles had a sharply demarcated line between the yellow outer material covering (or replacing the muscle) caudally underneath the carapace and more normal appearing muscle (albeit pale) cranially. The subcutaneous tissues and eyelids were edematous and there was approximately 75 mL of free fluid within the coelom and approximately 10 mL within the pericardial sac. There were multiple white nodules located in the joints of at least one digit from each limb. The nodules contained thick white material (possible gout). When the metacarpals of the front right limb were cut, tan material oozed out. The lungs looked unremarkable grossly. The kidneys were not readily apparent.
Laboratory Results:
Abnormal bloodwork included inclusion bodies of unknown origin in red blood cells, hypoglycemia, PCV 10% and Total Solid too low to read.
Microscopic Description:
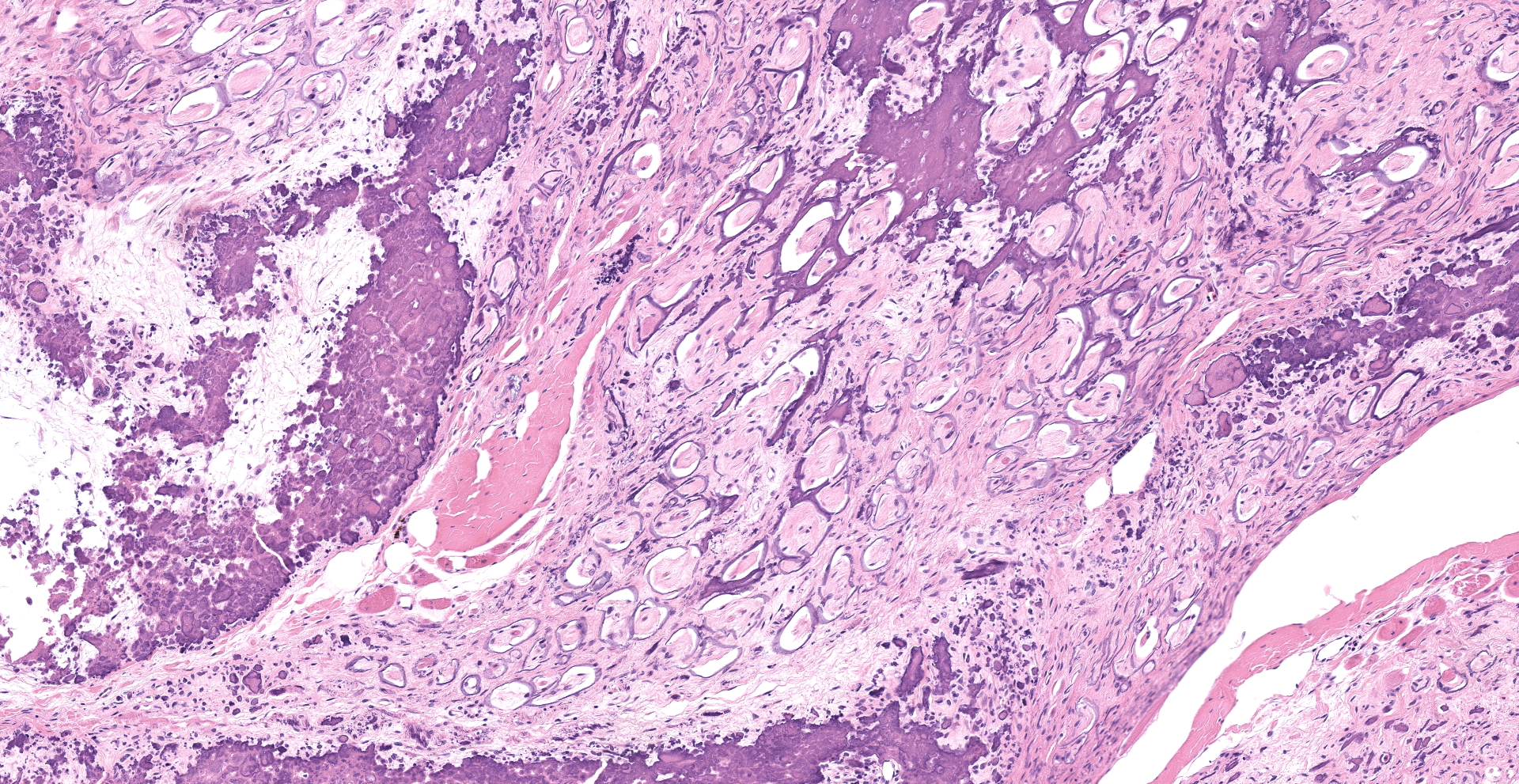
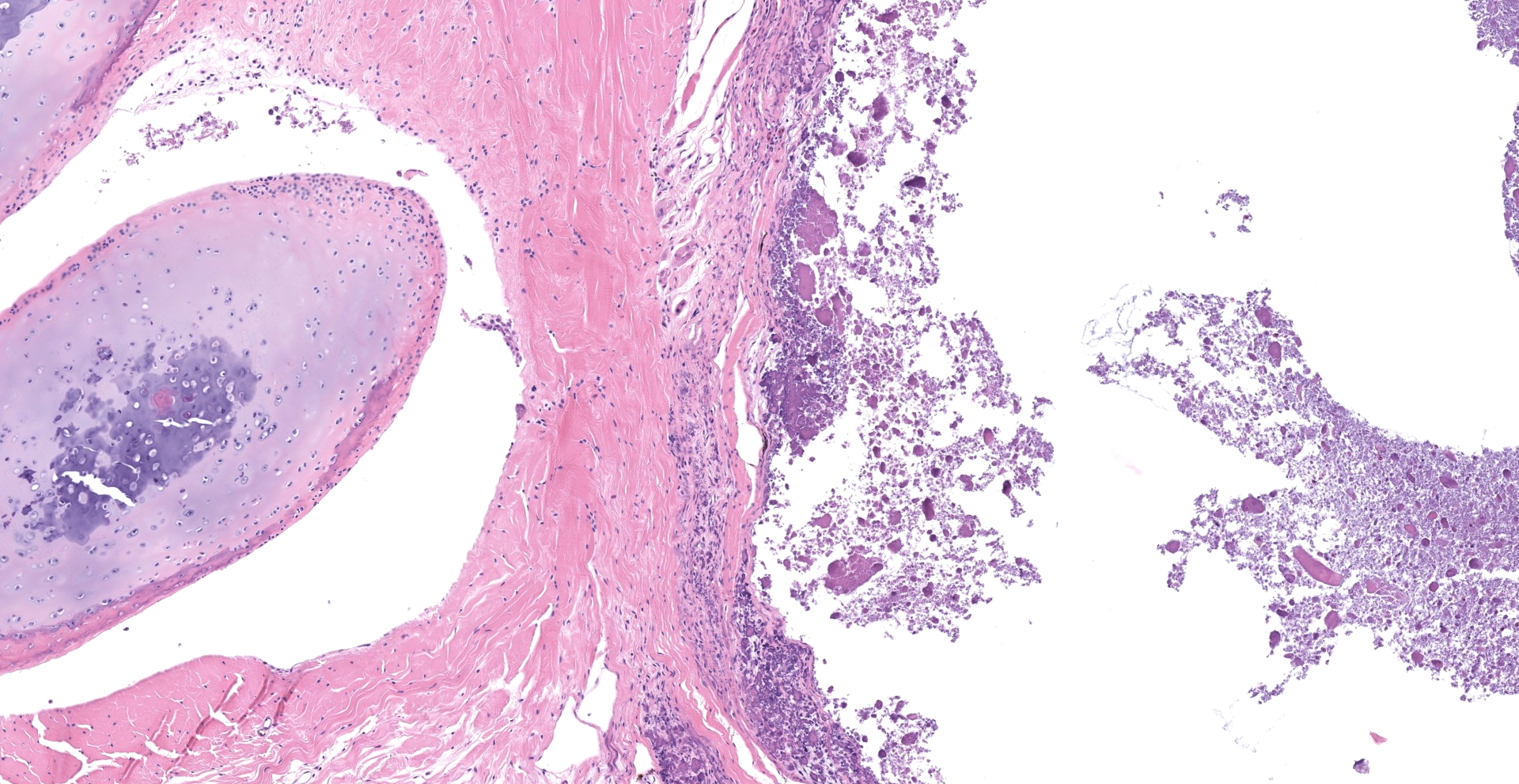
Joint with surrounding muscle/tissue (foot). The joint space has marked accumulation of amorphous fine, globular to dense deposits of mineralized material. Mineralization extends over joint surfaces and cartilage is eosinophilic with small numbers of lacunae containing cellular debris. Surrounding capsule has loss of epithelium with marked mineralization and occasional small numbers of granulocytes, lymphocytes and macrophages; latter are occasionally multinucleated. The adjacent skeletal muscle has widespread atrophy, degeneration with internalization of nuclei, loss of striations, increased eosinophilia, hypercontraction bands to fragmentation of cytoplasm and often blue stippled to dense deposits of mineral. Myocytes in affected areas are often ringed by a dense band of mineralized material with partial to complete loss of cytoplasm. In atrophic areas myocytes are separated by a very loose arrangement of fibrocytes and collagen.
In some slides, subcutaneous tissue has separation of fibrocytes and collagen; the latter has multifocal areas of light to dense mineralization. Vessels, when present, have occasional mineralized thin band within inner layer and multifocal deposits in arterial tunica muscularis.
Other tissues (not included) with similar, but less striking lesions: cornea (eye), kidney, mesentery and skin.
Contributor's Morphologic Diagnoses:
Severe, systemic mineralization with myonecrosis and pseudogout
(Hydroxyapatite deposition disease ? HADD)
Contributor's Comment:
Mineralization is the term used to describe deposition of insoluble, inorganic minerals, and is often categorized as either dystrophic, metastatic, idiopathic or iatrogenic.4 The nature of the mineral involved, in most species, is usually calcium paired with phosphate or carbonate.4 However, when deposition of crystalline material is seen in a reptile's joint(s), one must be careful to differentiate the nature of the mineralization, to accurately diagnose as either gout (urate crystals) or 'pseudogout' (other, including calcium), considering that in these species; gout can manifest as solely articular, separate from visceral or renal involvement. Pre-mortem assessment can include cytologic preparation, where once stained and observed under polarized light, the crystals will appear as strongly birefringent and needle-like, or radiography, since uric acid deposits are radiolucent and could be differentiated from other mineral-based deposits.3 However, caution must be exercised, since crystals can dissolve during staining and chronic gout lesions may mineralize3, giving false negatives resulting in challenging interpretation.
The submitter believes this periarticular mineralization to be pseudogout, also known in the literature as false gout, tumoral calcinosis, calcinosis circumscripta, and hydroxyapatite deposition disease (HADD). In other species, similar lesions may be calcium pyrophosphate deposits. Terminology, when referring to these deposition conditions, have been inconsistent and makes literature review difficult.
Animals with HADD present with non-ulcerated, firm, multinodular swellings that may impede their mobility.3 When radiographed, the nodules appear vibrantly radiopaque.3 Recommendations are often directed at husbandry and diet to slow progression of lesions, as regression has not been reported. On transection, the nodules are sharply demarcated, filled with white to yellow, pasty to chalky substance within periarticular tissues.3
Histology shows affected tissues expanded by multiple irregular, nodular aggregates of granular to amorphous, basophilic or eosinophilic non-birefringent material.3 Adjacent to mineral deposits, there is fibrous tissue and variable numbers of inflammatory cells, predominantly epithelioid macrophages and multinucleated giant cells.3
The exact cause of HADD, or articular tumoral calcinosis in reptiles, has not been elucidated. In other species, similar lesions have been linked to trauma, renal failure, hyperparathyroidism, hypervitaminosis D, and other tissue or bone conditions (e.g., infection or neoplasia); however, evidence of any of the previously mentioned underlying conditions have not been linked to any reports published so far.3
Previous articles describing HADD were almost exclusively covering juveniles, and the majority of cases were in turtles. The literature reports HADD in 10 Red-Bellied Short-Necked Turtles1, Painted Turtles2 and a Red-Eared Slider3. There was also a report in a Uromastyx sp. lizard.1 Between 2017 and 2019, the submitting institution has had 4 cases of periarticular mineralization: the current case and 3 Red-Eared Sliders, ages 25, 27 and another adult (exact age unknown).
The severity and extensive mineralization in multiple tissues seen in the present case was not reported in literature, and may indicate a systemic involvement, such as renal failure or calcium/phosphorus imbalance, and could not be ruled out due to constraints in testing pre-mortem.
Contributing Institution:
Veterinary Pathology ? Western College of Veterinary Medicine (https://wcvm.usask.ca/departments/vet-pathology.php)
JPC Diagnosis:
1. Leg, skeletal muscle: Fibrosis and dystrophic calcification, multifocal to coalescing, severe, with myofiber atrophy and loss.
2. Bone marrow, adipose tissue: Atrophy, diffuse, moderate to severe.
JPC Comment:
The contributor provides an excellent review of hydroxyapatite deposition disease (HADD) in reptiles. Although HADD has predominantly been described in aquatic turtles, it has also rarely been reported in lizards and should be considered as differential diagnosis for reptiles presenting with gout-like lesions.
Various molecular forms of calcium salts may be deposited in tissues. Analysis of crystals forming periarticular deposits in cases of HADD are often composed of hydroxyapatite (Ca10(PO4)2H2O), the primary mineral component of bone. Other species, such as humans, NHPs, and canines1, and avians6 are affected by similar periarticular calcium deposits. However, in these species, the calcium salt is often composed of structurally dissimilar calcium pyrophosphate dehydrate (Ca2P2O42H2O). Therefore, the term "hydroxyapatite deposition disease" has been suggested to specifically apply to disease associated with hydroxyapatite while "pseudogout" be applied to disease associated with calcium pyrophosphate dehydrate. Furthermore, human literature typically applies the term "tumoral calcinosis" for nodular periarticular deposits of hydroxyapatite whereas veterinary literature typically applies the term "calcinosis circumscripta" for similar lesions.1
Although unlikely in this case, the pathogenesis of gout is worthy of discussion given the commonality of the disease amongst reptiles. Gout occurs as the result of hyperuricemia, due to excess uric acid, and results in the deposition of monosodium urate crystals (known as tophi) in organs and soft tissues. In terrestrial reptiles, uric acid is the main break down product of protein, purines, and other non-protein sources of nitrogen. Excess uric acid may occur as the result of overproduction (i.e. primary gout), such as in cases of excessive dietary purines from insect rich diets, or may accumulate in the body due to decreased renal excretion (i.e. secondary gout) in cases such as starvation, dehydration, and damage to the renal tubules. Deposition of tophi is frequently associated with granulomatous inflammation and often results in nodular periarticular swellings observed in association with joints.5
A useful tool for differentiating gout from calcium containing minerals such as calcium pyrophosphate when analyzing joint fluid is the combined use of a polarizing filter with a red compensator filter. Gout crystals composed of monosodium urate are negatively birefringent, demonstrated by the colors yellow and blue when aligned parallel and perpendicular to the direction of polarization, respectively.5,6 Conversely, calcium pyrophosphate crystals in cases of pseudogout demonstrate positive birefringence, demonstrating a blue color when parallel to the slow axis of the compensator and yellow when perpendicular.6 Histologically, Von Kossa and Alizarian red S stains are useful for identification of calcium containing mineral in nondecalcified histologic sections, such as in cases of HADD and pseudogout.6 However, confirmation of calcium deposition is not pathognomonic, since chronic gout lesions may also undergo mineralization, as previously noted by the contributor. In regard to this case, Von Kossa stain confirms the presence of calcium containing mineral within the section, further supporting the diagnosis of HADD.
An additional finding noted by conference participants is significant atrophy of adipose tissue within the bone marrow. Adipose atrophy is an indication metabolic demand chronically exceeding dietary intake, resulting in the mobilization and eventual depletion of fat stores. Although reptilian adipose tissue is typically concentrated in abdominal fat bodies or the tail, the bone marrow is a useful location to assess the nutritional status of an animal at the time of its death.
References:
1. Burns RE, Bicknese EJ, Westropp JL, Shiraki R, Stalis IH. Tumoral calcinosis form of hydroxyapatite deposition disease in related red-bellied short-necked turtles, Emydura subglosoba. Vet Path. 2013; 50(3): 443-450.
2. Chambers JK, Suzuki T, Une Y. Tophaceous pseudogout of the femorotibial joint in a painted turtle (Chrysemys picta). J. Vet. Med. Sci. 2009; 71(5): 693-695
3. Graham EA, Burns RE, Ossiboff RJ. Depositional diseases. In: Garner MM, Jacobson ER, ed. Noninfectious Diseases and Pathology of Reptiles: Color Atlas and Text. Vol 2. 2nd ed. Boca Raton, FL: CRC Press; 2021: 109-114.
4. Hargis AM, Myers S. The Integument. In: James F. Zachary's Pathologic Basis of Veterinary Disease. 6th ed. St Louis, MO: Elsevier; 2017: 1030.
5. Pennick KE, Holicky RA, Wilkerson MJ. What is your diagnosis? Joint and associated tissue aspirates from a Bearded Dragon (Pogona vitticeps). Vet Clin Pathol. 2017;46(2):363-364.
Terio KA, McAloose D, St. Leger J, eds. Pathology of Wildlife and Zoo Animals, Cambridge, MA Academic Press; 2018: 699,833,877-8.