Signalment:
Gross Description:
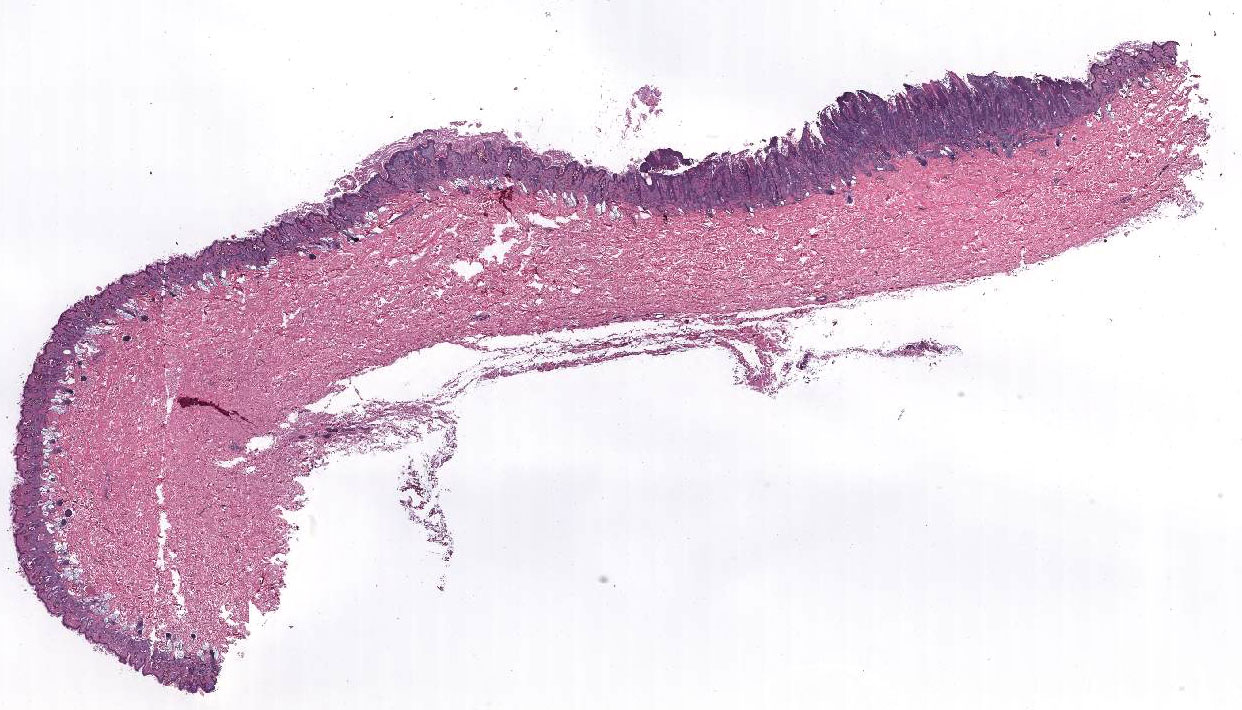
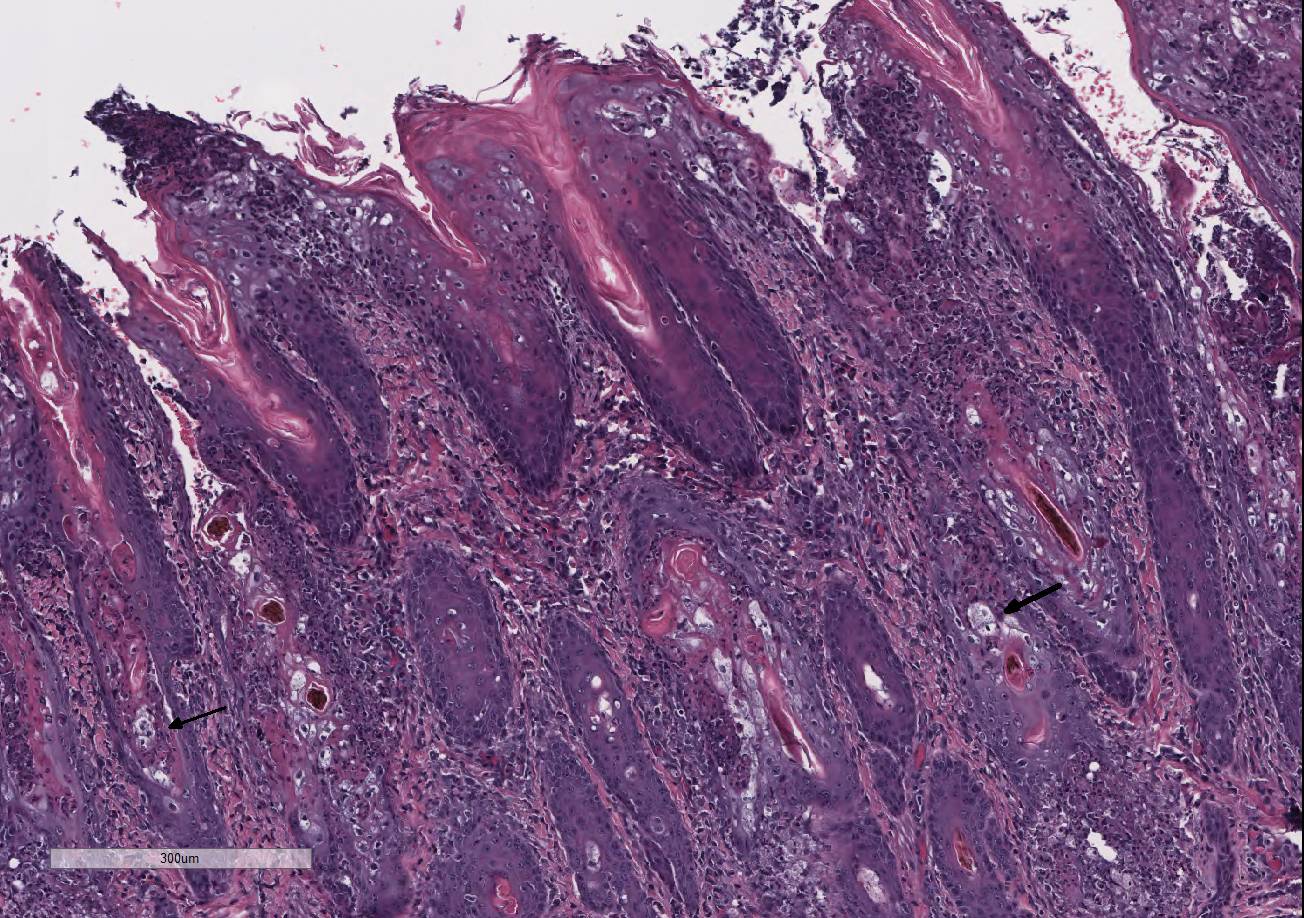
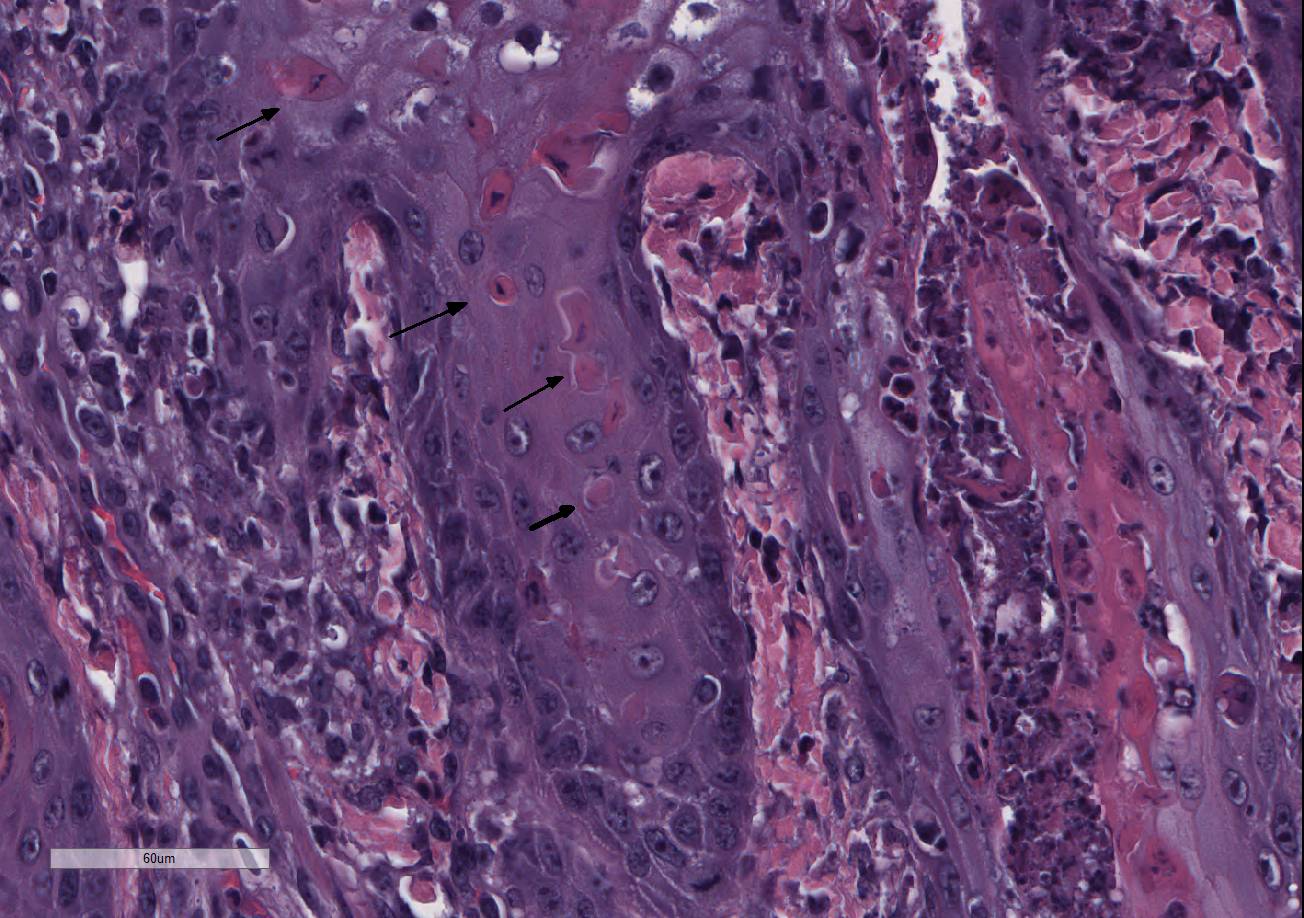
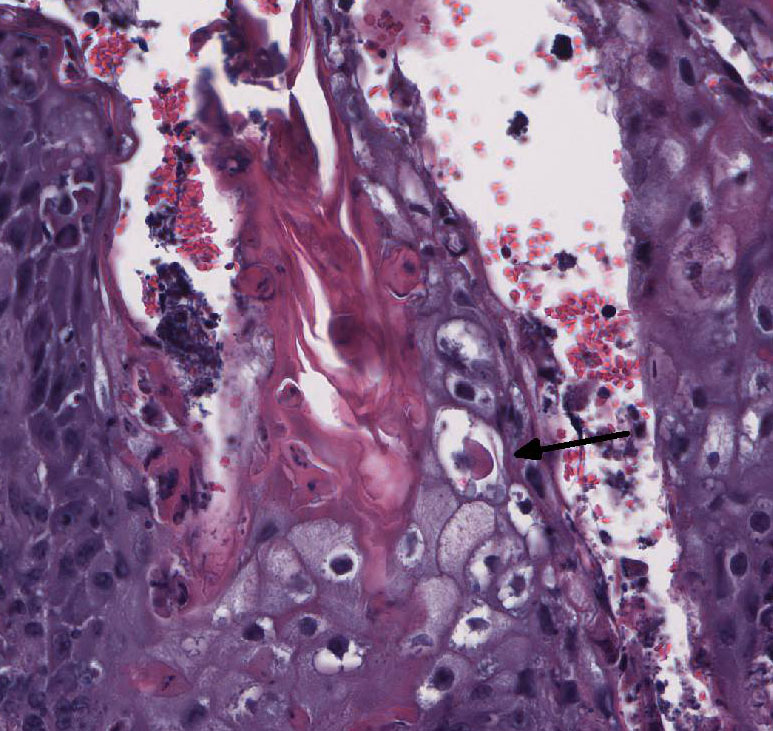
Histopathologic Description:
Morphologic Diagnosis:
Lab Results:
Condition:
Contributor Comment:
There are at least 36 types of ichthyosis described in humans.14 Ichthyosis in animals has not been well correlated with human forms. However, there are five human ichthyosis correlates described in animals, which include ichthyosis vulgaris, x-linked ichthyosis, epidermolytic hyperkeratosis, lamellar ichthyosis and harlequin ichthyosis.9 Ichthyosis is a rare skin condition that has been shown to affect cattle, dogs, pigs, chickens, mice and llamas9, among others. Two inherited forms of ichthyosis, both through autosomal recessive genes, have been reported in cattle: ichthyosis fetalis and ichthyosis congenita. Ichthyosis fetalis is generally fatal and affected animals usually survive for only a few days after birth. This disease has been described in Norwegian red poll, Friesian, and brown Swiss calves, and this entity most closely resembles human harlequin ichthyosis.2 Hairless skin is associated with ichthyosis fetalis and is characterized by large, horny, plates separated by deep clefts. Microtia, cataracts, thyroid hypoplasia, and eversion of mucocutaneous junctions are common features of this type of ichthyosis. These manifestations were not apparent in this case.
Ichthyosis congenita, a milder variant of ichthyosis, has been reported in Jerseys, Pinzgauer, Chianina, and Holstein-Friesian breeds.9 The lesions of ichthyosis congenita are similar to those of ichthyosis fetalis, but less severe and often localized to the skin over the abdomen, inguinal region, joints and the muzzle. Ichthyosis congenita most closely resembles human lamellar ichthyosis. Due to the older age of this calf and clinical presentation, ichthyosis congenita is suspected in this case. Eight genes have been linked to congenital ichthyosis: TGM1 (in people and Jack Russell terriers, but shown to not be the case in cattle 3, ABCA121, ABHD5(CG158)7, 2 lipoxygenases (ALOXE3 and ALOX12B5, NIPAL4 (ICTHYIN, Golden retrievers10), LIPN4, CYP4F2213, and PNPLA-1(American bulldogs16). The listed mutations result in disruption of the normal protective barrier that the skin provides while ichthyosis represents the local response to restore that barrier4. For example, TGM deficiency results in the abnormal cross-linking of the cornified envelope. Lipoxygenase deficiency in ALOXE3 and ALOX12B mutations are directly associated with abnormal lipid metabolism, impairing the structural integrity of the lipid bilayers. GJB2 mutations affect gap-junction integrity. Accumulation of large amounts of keratin and impairment of the permeability barrier promote the colonization and subsequent infection of skin. The surface Malassezia sp. infection, suppurative folliculitis, and poxviral dermatosis likely represent secondary lesions to the ichthyosis.
Follicular dysplasia or hypotrichosis is not an initial feature of ichthyosis congenita. However, hairlessness was present at birth in this calf, which suggests a primary rather than secondary process. Histologically, the follicular morphology is most consistent with follicular dysplasia. Based on genetic studies, an autosomal recessive component for follicular dysplasia has been described in cattle, such as for Herefords, Polled Herefords, Ayrshires, Guernseys, Jerseys, Holsteins and Black Angus.8,12,15
JPC Diagnosis:
Conference Comment:
Both lethal and viable variants of autosomal recessive and dominant forms of congenital hypotrichosis have been reported with generalized alopecia associated with excessive scaling, infundibular hyper-keratosis, easily broken and misshapen hair shafts, keratinocyte degeneration, and dilated apocrine glands. Other reported findings include subcapsular hepatic fibrosis, anemia, and neurologic deficits.8,11 Non-genetic causes of hypotrichosis in calves include iodine deficiency, adeno-hypophyseal hypoplasia, maternal ingestion of the toxic plant Veratrum album, and intrauterine infection with bovine pestivirus.9
References:
2. Chittick EJ, Olivry T, Dalldorf F, Wright J, Dale B a, Wolfe B a. Harlequin ichthyosis in two greater kudu (Tragelaphus strepsiceros). Vet Pathol. 2002;39:751-756.
3. Dardano S, Gandolfi B, Parma P, et al. Characterization of bovine TGM1 and exclusion as candidate gene for ichthyosis in Chianina. J Hered. 2008;99(1):81-83.
4. Elias PM, Williams ML, Holleran WM, Jiang YJ, Schmuth M. Pathogenesis of permeability barrier abnormalities in the ichthyoses: inherited disorders of lipid metabolism. J Lipid Res. 2008;49(4):697-714.
5. Jobard F, Lefèvre C. Lipoxygenase-3 (ALOXE3) and 12 (R)-lipoxygenase (ALOX12B) are mutated in non-bullous congenital ichthyosiform erythroderma (NCIE) linked to chromosome. Hum Mol Genet. 2002;11(1):107-113.
6. Jubb, Kennedy & Palmers Pathology of Domestic Animals 5th edition
7. Lefèvre C, Jobard F, Caux F, et al. Mutations in CGI-58, the gene encoding a new protein of the esterase/lipase/thioesterase subfamily, in Chanarin-Dorfman syndrome. Am J Hum Genet. 2001;69(5):1002-1012.
8. Mansell JL. Follicular dysplasia in two cows. Vet Dermatol. 1999;10(2):143-147.
9. Mauldin EA, Peters-Kennedy J. Integumentary system. In: Maxie MG ed. In: Jubb Kennedy and Palmer's Pathology of Domestic Animals. Vol 1. 6th ed. Philadelphia, PA: Elsevier Saunders; 2016:530-532, 538-540.
10. "'>Mauldin EA, Wang P, Evans E, et al. Autosomal Recessive Congenital Ichthyosis in American Bulldogs Is Associated With NIPAL4 (ICHTHYIN) Deficiency. Vet Pathol. 2015;52(4):654-662.
11. Mecklenburg L. An overview on congenital alopecia in domestic animals. Vet Dermatol. 2006;17(6):393-410.
12. Miller WH, Scott DW. Black-hair follicular dysplasia in a Holstein cow. Cornell Vet. 1990;80(3):273-277.
13. Ohno Y, Nakamichi S, Ohkuni A, et al. Essential role of the cytochrome P450 CYP4F22 in the production of acylceramide, the key lipid for skin permeability barrier formation. Proc Natl Acad Sci U S A. 2015;112(25):7707-7712.
14. Oji V, Tadini G, Akiyama M, et al. Revised nomenclature and classification of inherited ichthyoses: Results of the First Ichthyosis Consensus Conference in Sorèze 2009. J Am Acad Dermatol. 2010;63(4):607-641.
15. Scott DW. Color Atlas of Farm Animal Dermatology. 1st ed.; 2008.
16. Tamamoto-Mochizuki C, Banovic F, Bizikova P, Laprais A, Linder KE, Olivry T. Autosomal recessive congenital ichthyosis due to PNPLA1 mutation in a golden retriever-poodle cross-bred dog and the effect of topical therapy. Vet Dermatol. May 2016.