Joint Pathology Center
Veterinary Pathology Services
Wednesday Slide Conference
2018-2019
Conference 15
16 January 2019
CASE III: 13004 (JPC 4048861).
Signalment: 8 weeks, male, Wistar/Crlj, Rattus norvegicus, rat
History: The rat was administered alloxan (50mg/kg) by an intra-vein injection for single time at the 7 weeks. The rat lost its body weigh gradually after injection. Both the food intake and the urine volume were also less than other diabetic rats. It was sacrificed at the 8 weeks.
Gross Pathology: Both kidneys were enlarged at the time of necropsy.
Laboratory results:
General parameters and blood glucose (BG)
|
time |
Purchased day |
injective day |
Day1 |
Day 2 |
Day 3 |
Day 4 |
Day 5 |
Day 6 |
Day 7 |
|---|---|---|---|---|---|---|---|---|---|
|
BW(g) |
237.3 |
239.6 |
/ |
232.4 |
/ |
/ |
/ |
/ |
202.4 |
|
Food Consumption(g) |
23.38 |
13.36 |
10.55 |
1.51 |
3.09 |
5.76 |
9.95 |
14.45 |
|
|
Urine volume (ml) |
28 |
28 |
42 |
22 |
22 |
32 |
26 |
36 |
56 |
|
BG?mg/dl? |
92 |
/ |
418 |
/ |
/ |
/ |
/ |
/ |
455 |
Microscopic Description:
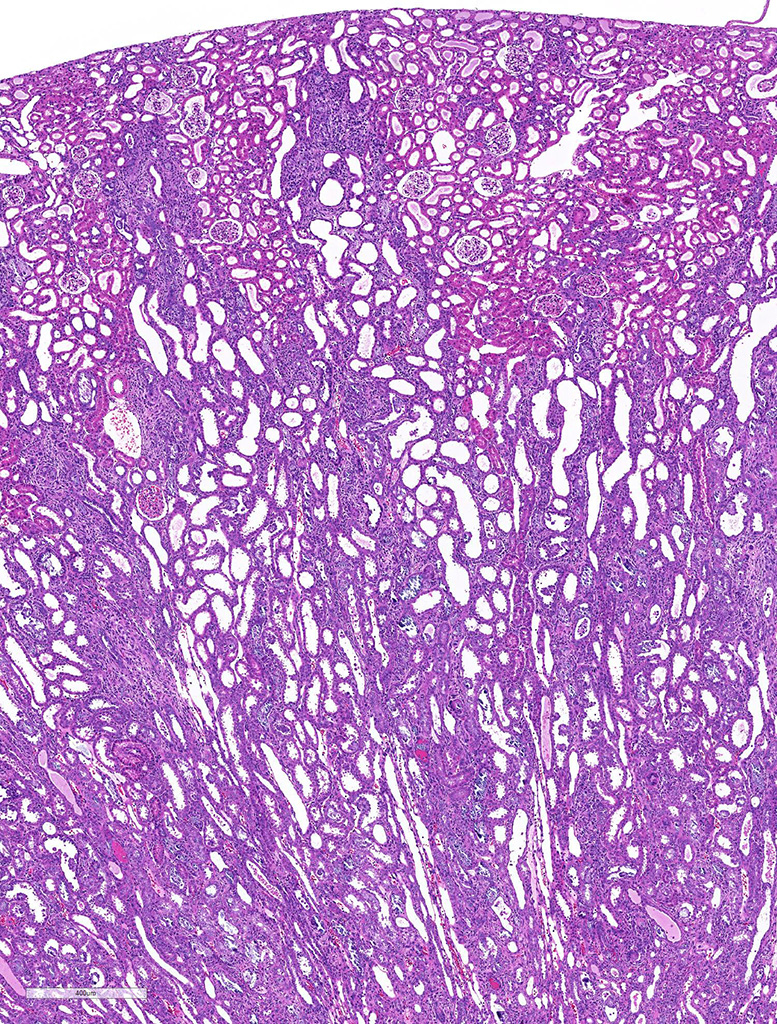
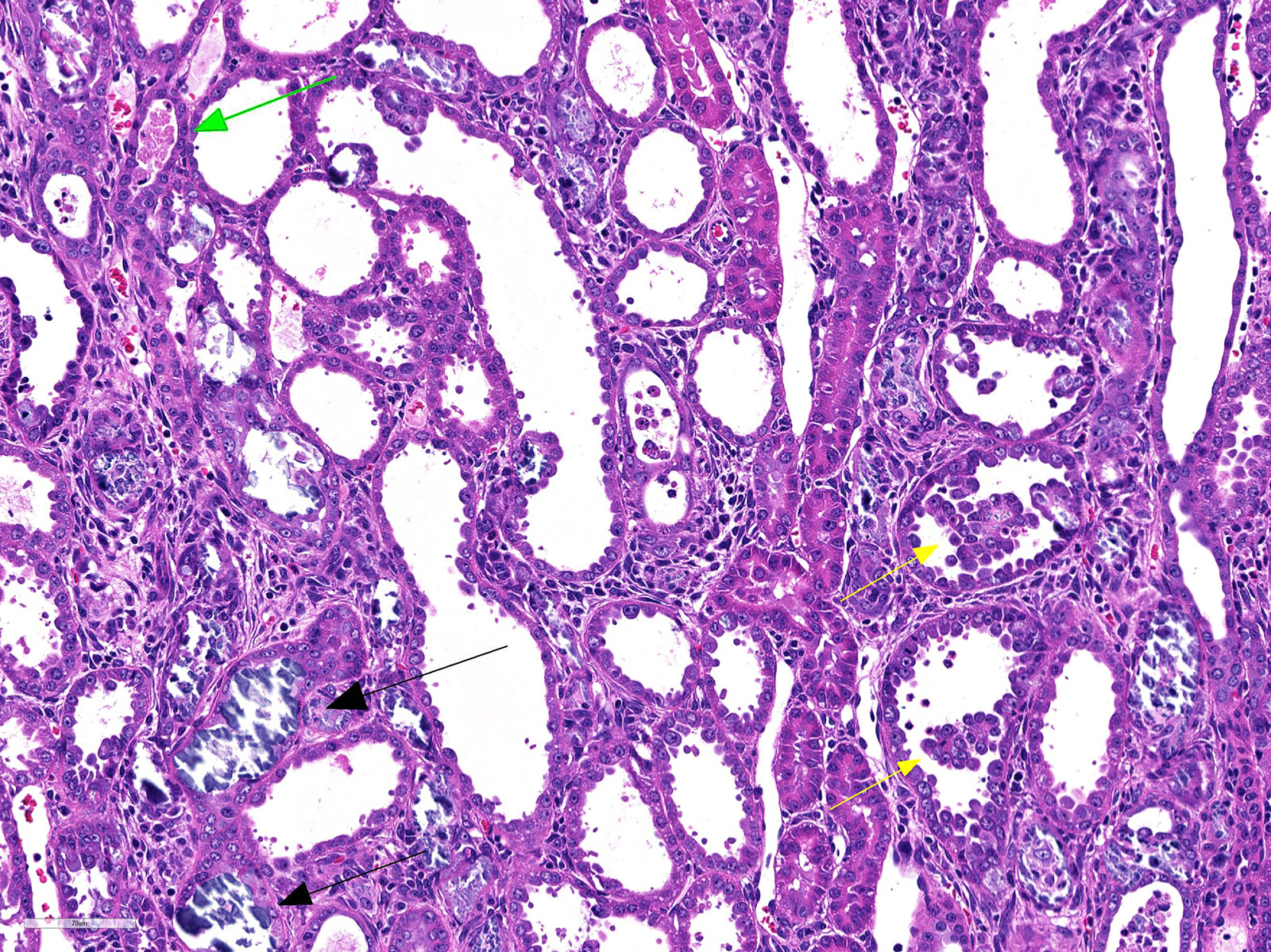
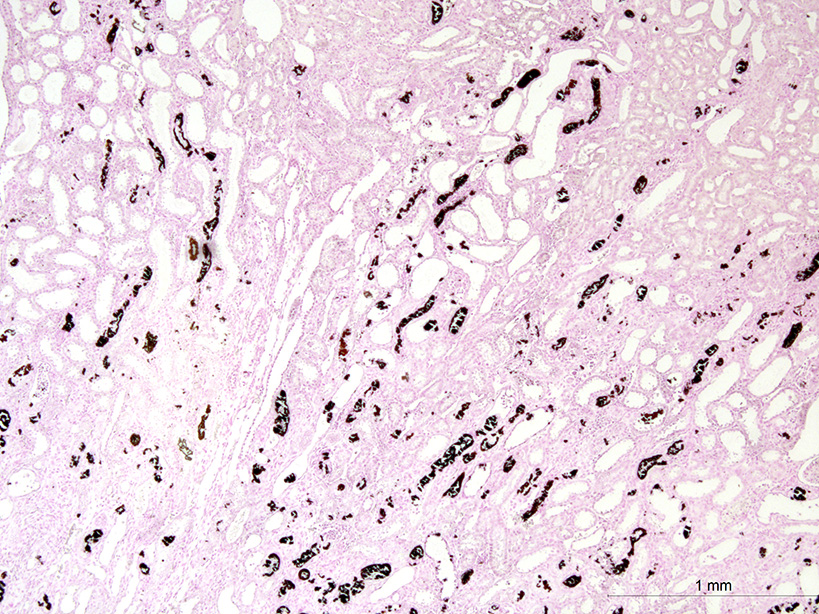
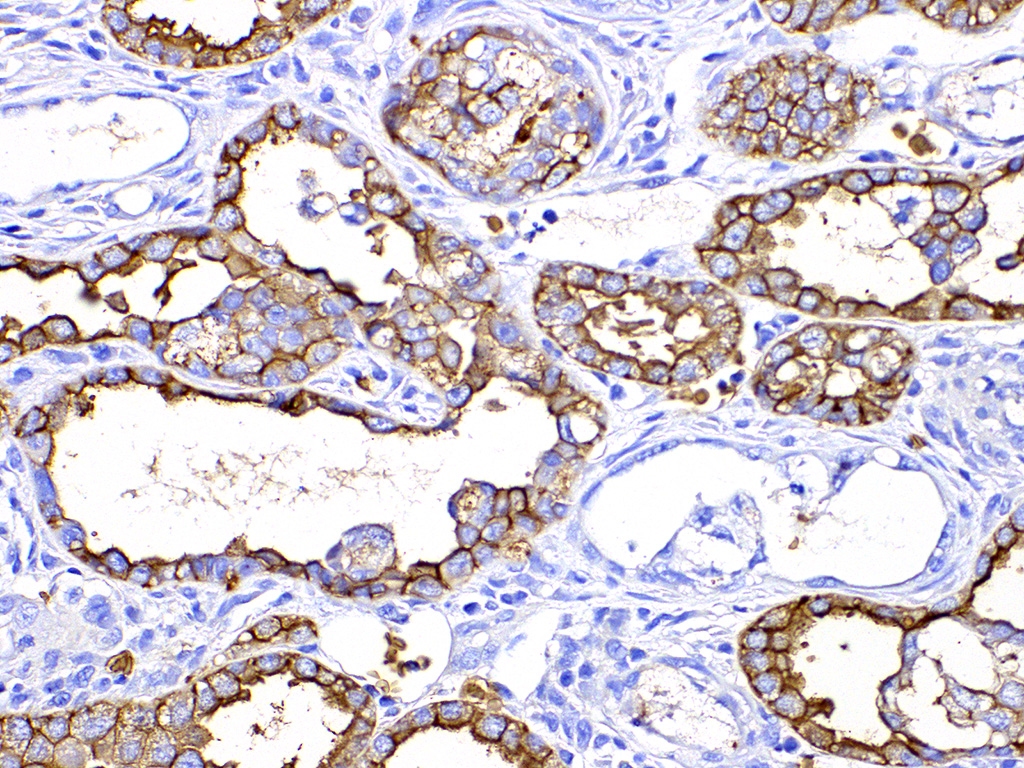
Both two kidneys showed similar lesions. Many dilated and occluded tubules were segmentally observed in inner cortex and outer medulla. Degeneration and necrosis of tubular epithelial cells were seen with/without tubular obstruction by cell debris and crystal/mineral. Dilated tubules were lined with flattened- and attenuated-epithelium with basophilic cytoplasm. In the interstitium around degenerated tubules, multinuclear giant cells, macrophages and lymphocytes infiltrated with fibrosis. Some multinuclear giant cells formed granuloma containing the crystal and mineral . The crystals and minerals located mainly inside the tubules, and some occluded the tubular lumen. In some tubules, they were enveloped by tubular epithelial cells, and tubular basement membrane was disrupted by these crystals and minerals. Mineral was positive for von Kossa’s method, but crystal was negative, and both showed non-birefringence with polarized microscopy. Mineral was confirmed as calcium salt. Damaged tubules were mainly the proximal tubules that have brush border and immunopositive for aquaporin-1, but some were distal tubules that were negative for aquaporin-1 and positive for Na+/K+ pomp.
Contributor’s Morphologic Diagnoses:
Kidney: Granulomatous tubulointerstitial nephritis with tubular necrosis, degeneration and regeneration, and mineralization.
Contributor’s Comment: Type 1 diabetes in the rat induced by intravenous injection of alloxan is one of the most common experimental diabetic model which was firstly discovered by Dunn et al. Alloxan induces selectively necrosis of pancreatic beta cells. Alloxan molecule is structurally similar to glucose, and it can enter beta cells via glucose transporter 2 (GLUT2).2 In the presence of intracellular thiols, especially glutathione, alloxan generates reactive oxygen species (ROS) in a cyclic redox reaction with its reduction product, dialuric acid. Autoxidation of dialuric acid generates superoxide radicals, hydrogen peroxide and, in a final iron-catalyzed reaction step, hydroxyl radicals. These hydroxyl radicals are ultimately responsible for the death of the beta cells.4 Nephrotoxicity is a dominating feature of the toxicity of alloxan after systemic administration.4 As alloxan is an unstable substance, it disappears within five minutes and almost entirely converted to the relative stable alloxanic acid or alloxanates in plasma and these might exist in the largest amount in urine. Therefore nephrotoxicity is so severe that it causes fatal renal failure in the animals before diabetes can develop.4 The expression of GLUT2 in tubular epithelial cells may explain why the toxins can cause damage to the kidney.4
The nephrotoxic changes induced by alloxan include extensive swelling and vacuolar degeneration of renal tubular cells, necrosis of tubular cells with disappearance of brush border and tubular dilution, cellular infiltrates resembling granuloma in the interstitium from the third day to fourteen days.5 Histological changes of the kidney observed in alloxan-induced diabetic rats were vacuolar change/ glycogen deposition of the tubular cells and glomerular lesions. Thus, the histologic changes in our case could be differentiated from diabetic change.
Alloxan promotes intratubular mineral deposition and granulomatous interstitial nephritis. These minerals were also localized in the interstitium. The mechanism of translocation of mineral from tubular lumen to interstitium might be similar to that of calcium oxalate crystals1. Crystal in the tubules is surrounded by tubular epithelial cells and then entered the interstitium through disrupted basement membrane of the tubules and then engulfed by macrophages to form foreign granuloma.
Contributing Institution:
Department of Pathology, Faculty of Pharmaceutical Sciences, Setsunan University,
45-1 Nagaotohge-cho, Hirakata, Osaka 573-0101, Japan
http://www.setsunan.ac.jp/~p-byori/
JPC Diagnosis: Kidney, distal convoluted tubules and collecting ducts: Tubular degeneration, necrosis and regeneration, diffuse, severe, with granulomatous tubular interstitial nephritis, rare intratubular crystals, and marked tubular mineralization.
Conference Comment: Alloxan, a pyrimidine derivative is one of the world’s oldest named organic compounds. It was originally derived by the oxidation of uric acid by nitric acid, with early investigators using various sources of uric acid, including human kidney stones as well as boa constrictor excrement (which contains up to 90% ammonium acid urate ) The compound was named by Wohler and von Leibig in 1838, who began with the synthesis of urea in 1828, then of uric acid, and some additional 13 derivatives of uric acid of which alloxan is one.4 In another one of those cruel twists of fate over naming compounds, Wohler and Leibig were previously aware of similar research conducted in 1818 by Brugnatelli (working in Italy and publishing in Italian) who created the compound almost two decades previously and had named it “erythric acid” as a result of the red staining it caused on his fingers. The word “alloxan” is derived from a combination of “allantoin”, and oxalic acid. A number of reductive products from alloxan exist, including dialuric acid (also diabetogenic in animals), and an anhydrous dimer, alloxantin (produced by the partial reduction of alloxan with hydrogen sulfide.)4
As discussed by the contributor, alloxan’s similarity to glucose allows it to be taken up by GLUT2 glucose transporters in pancreatic beta cells, as well as renal tubular epithelium, resulting in the characteristic necrosis in both cell types, and ultimately “alloxan diabetes” and a change in the kidney initially referred to as “cortical sponge kidney”5 (an appellation that like erythric acid, has fortunately not survived the test of time.) Without its structural similarity to glucose, alloxan would not be able to penetrate the cell membrane as it is not lipid soluble.
The basic mechanism of action of alloxan is its generation of reactive oxygen species and selective necrosis of beta cells. The reduction of alloxan within beta cells to dialuric acid results in the generation of the superoxide radical as well as hydrogen peroxide, and in the presence of iron, hydroxyl radicals via the Fenton reaction.3 In addition, alloxan also inhibits glucokinase, inhibiting glucose-induced insulin secretion. This action occurs within a minute of alloxan administration, following alloxan’s almost immediate inhibition of glucose oxidation and generation of ATP necessary to stimulate insulin release from beta cells. 3
Another commonly used diabetogenic agent is streptozotocin, a chemotherapeutic alkylating agent and antineoplastic, which was first identified as a diabetogenic agent in 1963. 3 This nitrosourea analogue exhibits its effects of beta cells through methylation, or a damaging transfer of a methyl group from streptozotocin to the DNA and other protein molecules of the beta cell following selective uptake through the GLUT2 glucose transporter.3
References:
- de Water R, Noordermeer C, van der Kwast TH, Nizze H, Boeve ER, Kok DJ, Schroder FH: Calcium oxalate nephrolithiasis: effect of renal crystal deposition on the cellular composition of the renal interstitium. Am J Kidney Dis 33: 761-771, 1999
- Elsner M, Tiedge M, Guldbakke B, Munday R, Lenzen S: Importance of the GLUT2 glucose transporter for pancreatic beta cell toxicity of alloxan. Diabetologia 45: 1542-1549, 2002
- Lenzen S: The mechanisms of alloxan- and streptozotocin-induced diabetes. Diabetologia 51: 216-226, 2008
- McLetchie NGB. : Alloxan diabetes: a discovery albeit a minor one. J Rou Coll Phsyicans Edinb 2002; 32:134-142.
- Vargas L, Friederici HH, Maibenco HC: Cortical sponge kidneys induced in rats by alloxan. Diabetes 19: 33-44, 1970