Signalment:
1-year-old intact domestic shorthair cat, (
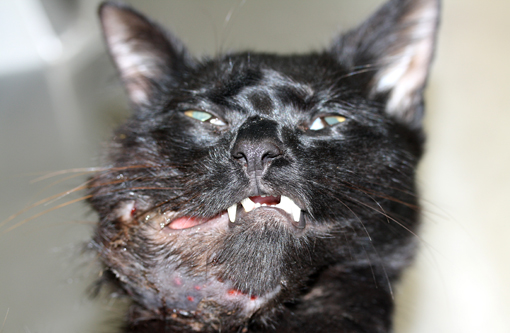
Felis cats).A recently rescued approximately 1-year-old stray, intact DSH cat, weighing 3.2 kg was presented to the Ross University Veterinary Teaching Hospital (RU-VTH) for routine physical examination and vaccination. A 1 cm smooth, firm to hard, unilateral, non-painful mass was detected on the right mid-mandible. Multifocal areas of ulceration were present on the buccal surface of the gums along the molar portion of the body of the mandible. Despite antibiotic therapy the cats overall physical condition deteriorated and two months later it was brought back to the RU-VTH for evaluation. The mass was fast growing, reaching 6 cm in its largest dimension. The cat was euthanized due to poor body condition and progression of the lesion despite therapy.
Gross Description:
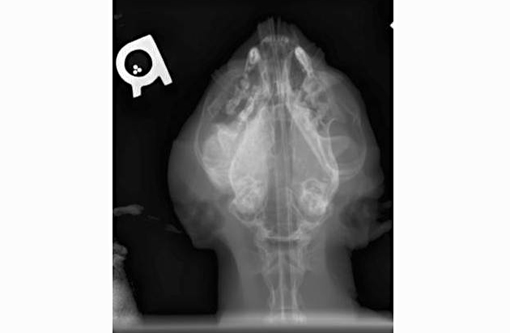
Abnormal findings were confined to the right mandible and the regional lymph nodes. The mandible was brittle and could be sliced with the necropsy knife. The right lower gingival mucosa was diffusely red, firm and swollen. The swelling extended to the right sublingual inter-mandibular space. The right submandibular and parotid lymph nodes were markedly enlarged (up to 2 cm in the largest dimension), firm and wet.
Histopathologic Description:
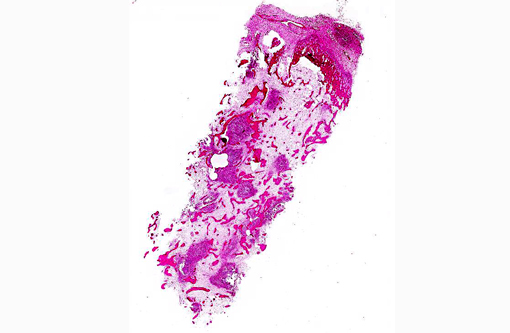
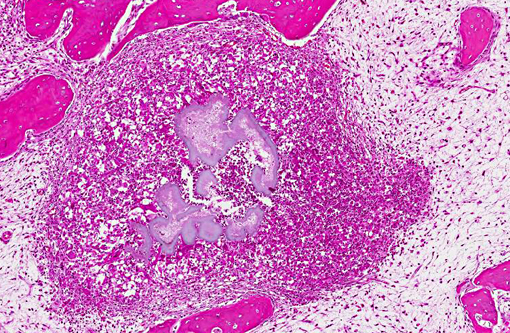
Mandibular mass: Routine H&E sections from the core biopsy revealed focal new bone formation around areas of osteolysis and inflammatory cell infiltration surrounding large, irregularly-shaped, faintly basophilic clusters of filamentous bacteria rimed by fine eosinophilic amorphous material (Splendore-Hoeppli phenomenon). Bacterial colonies were surrounded by neutrophils, lesser numbers of plasma cells, macrophages, and occasional multinucleated giant cells and fibroblasts at the outermost layer. Anastomosing trabeculae of newly deposited woven bone (bone proliferation) were seen around inflammatory foci.Â
Morphologic Diagnosis:
Chronic multifocal pyogranulomatous osteomyelitis with intralesional filamentous bacteria.Â
Lab Results:
A core bone marrow biopsy was used for culture and incubated at 37°C in aerobic and anaerobic conditions. Microscopically the cultured organisms were characterized as Gram positive, acid fast negative, filamentous bacteria. Molecular testing of the isolates by amplification and sequencing of the 16S rRNA gene identified the isolates as
Nocardia cyriacigeorgica.
Condition:
Nocardial osteomyelitis
Contributor Comment:
Mandibular osteomyelitis, also known as lumpy jaw in ruminants and other domestic and wild animal species, is uncommon in domestic cats. It is a condition characterized by pyogranulomatous inflammation resulting in bone destruction and remodeling. Lumpy jaw in hoofed animals is believed to occur secondary to oral infection, poor drainage, coarse forage ingestion, dental eruptions and oral abrasions. Common bacterial etiologies for lumpy jaw are
Actinomyces bovis in ruminants and
Fusobacterium necrophorum in kangaroos.(1)
Nocardia spp. are aerobic actinomycetes ubiquitous in soil and water, and are known to cause opportunistic infections in terrestrial and marine mammal species.(4) The severe pyogranulomatous mandibular osteomyelitis diagnosed in this cat was the result of infection with a recently described Nocardia species,
Nocardia cyriacigeorgica. In recent years, Nocardia species identification and taxonomy has greatly improved with the availability of molecular methods such as 16S rRNA and hsp65 gene sequence analysis.Â
Nocardia cyriacigeorgica is an actinomycete which, since its first description in 2001, has been reported as a causative agent of human disease in Europe, Asia, Canada and the USA.(5) While
Nocardia cyriacigeorgica infection has not been described in domestic species, it has been reported in a captive beluga whale,(4) and is currently regarded as an emergent pathogen in people.(3) To our knowledge this is the first report of
N. cyriacigeorgica-induced mandibular osteomyelitis in a cat. Interestingly, gross and microscopic findings of the mandibular lesions caused by this potentially zoonotic bacterium closely resemble those of mandibular actinomycosis (lumpy jaw) in cattle.
JPC Diagnosis:
Bone: Osteomyelitis and cellulitis, pyogranulomatous, multifocal to coalescing, marked, with large colonies of filamentous bacteria
Conference Comment:
This case is interesting in that
Nocardia spp. is not generally classified as a large colony forming bacterium in tissue;
Yersinia spp.,
Actinomyces spp.,
Actinobacillus spp.,
Corynebacterium spp.,
Staphylcoccus spp. and
Streptococcus spp. (see
WSC 2013-14, conference 1, case 3) are the most common bacteria that form large colonies in tissues in veterinary species. The main etiologic rule-out for the gross and histopathologic findings is
Actinomyces spp., a gram-positive, non-acid fast filamentous bacteria commonly associated with cutaneous infection secondary to bite wounds and penetrating injuries from grass awns or other foreign bodies. The gross lesions of actinomycosis and nocardiosis are typically identical (cellulitis, abscesses, draining tracts, osteomyelitis), and although it is a more common feature of actinomycosis, both conditions can produce sulfur granules (grains composed of necrotic debris, aggregates of bacteria and Splendore-Hoeppli material). Both bacteria are gram-positive, while
Nocardia spp. is often, but not always, acid fast; if negative, it cannot be differentiated microscopically from
Actinomyces spp.(2) In this case, filamentous bacteria were both gram-positive and acid-fast; these histochemical staining characteristics, along with the culture results reported by the contributor, support a diagnosis of nocardiosis.Â
References:
1. Brookins MD, Rajeev S, Thornhill TD, Kreinheder K, Miller DL. Mandibular and maxillary osteomyelitis and myositis in a captive herd of red kangaroos (Macropus rufus). J Vet Diagn Invest. 2008;20:846-849.
2. Ginn PE, Mansell JEKL, Rakich PM. Skin and appendages. In: Maxie MG, ed. Jubb, Kennedy and Palmers Pathology of Domestic Animals. Vol.1. 5th ed. Philadelphia: Elsevier; 2007:686-687.Â
3. Schlaberg R, Huard RC, Della-Latta P. Pathogen in the United States Nocardia cyriacigeorgica, an emerging pathogen in the United States. J Clin Microbiol. 2008;46:265-273.
4. St. Leger JA, Begeman L, Fleetwood M, et al. Comparative pathology of nocardiosis in marine mammals. Vet Pathol. 2009;46:299-308.
5. Yassin AF, Raieny FA, Steiner U. Nocardia cyriacigeorgici sp. nov. Int J Syst Evol Microbiol. 2001;51:1419-1423.