Signalment:
Samples of mouthparts and a part of the foot and surrounding mantle from 3 affected blacklip abalone, 1, 2, and 3 were submitted in 10% buffered formalin for microscopic examination as well as a part of the foot with pedal nerves in alcohol for referral to Australian Animal Health Laboratory for PCR for abalone ganglioneuritis (ABG).
Gross Description:
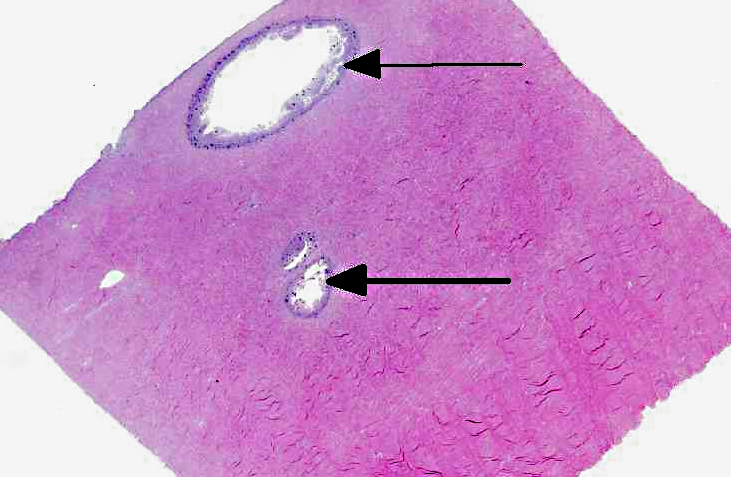
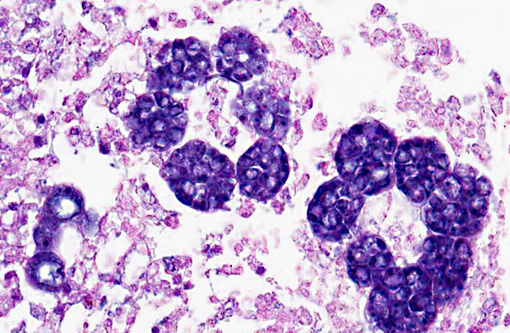
Histopathologic Description:
There were no histopathological lesions of ABG in the mouthparts (sections not presented).
Morphologic Diagnosis:
Lab Results:
Condition:
Contributor Comment:
ODonoghue et al linked P. olseni to widespread kills of abalone in the Gulf of St. Vincent side of the Yorke Peninsula, South Australia, and abalone stocks in this area have not recovered (S. Mayfield personal communication).(5) In the early 1990s, a significant proportion of H. rubra along approximately 500 km of the NSW coastline between Port Stephens and Jervis Bay died in association with P. olseni infections.
The route of entry of Perkinsus olseni is unknown. It proliferates in tissues but is eventually sequestered by the host and killed or ejected. During sequestration the parasite becomes surrounded by abalone pigment, causing the yellow/brown appearance of the nodules. These reduce market value, causing losses in fishery value. In some severe infections, death occurs without nodule formation, suggesting that the host is unable to mount an effective immune response in some cases. Infection is not always lethal to H. rubra and expression of disease is widely associated with environmental variables, primarily temperature, with infections becoming more severe at higher temperatures or when food availability diminishes.(4) Transmission occurs directly; prezoosporangia develop into zoosporangia in seawater which then release hundreds of motile, biflagellated zoospores (about 3 by 5 μm) which are infective to abalone and other mollusks.(3)
JPC Diagnosis:
Conference Comment:
Although this animal did not have abalone viral ganglioneuritis (AVG), participants discussed this disease as well. AVG is caused by a herpesvirus that affects only abalone; other mollusks appear to not be affected. Mortality rates in farmed abalone are up to 100%. The disease has been found to be slowly spreading in wild abalone along the Victorian (Australia) coast as well; however, the exact mortality rate in wild populations is uncertain.(2)
There was moderate slide variation, with some sections exhibiting more significant necrosis, and others showing cavitation of the pedal tissue.Â
References:
2. Corbeil S, McColl KA, Williams LM, et al. Abalone viral ganglioneuritis: establishment and use of an experimental immersion challenge system for the study of abalone herpes virus infections in Australian abalone. Virus Res. 2012;65(2):207-13.
3. Goggin, CL, Sewell KB, Lester RGJ. Cross-infection experiments with Australian Perkinsus species. Diseases of Aquatic Organisms. 1989:T:55-59.
4. Lester RJG, Hayward CJ. Control of Perkinsus Disease in Abalone. Final Report for FRDC Project no. 2005;2000/151.
5. O'Donoghue PJ, Phillips PH, Shepherd SA. Perkinsus (Protozoa: Apicomplexa) infections in abalone from South Australian waters. Transactions of the Royal Society of South Australia. 1991;115:77-82.
6. Petty D. Perkinsus infections in bivalve mollusks. University of Florida IFAS Extension. http://shellfish.ifas.ufl.edu/PDFs/Publications/Perkinsus%20Infections%20of%20Bivalve%20Molluscs.pdf. Accessed online 26 January 2013.Â