Signalment:
Gross Description:
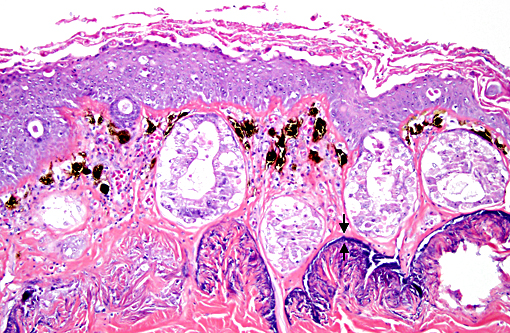
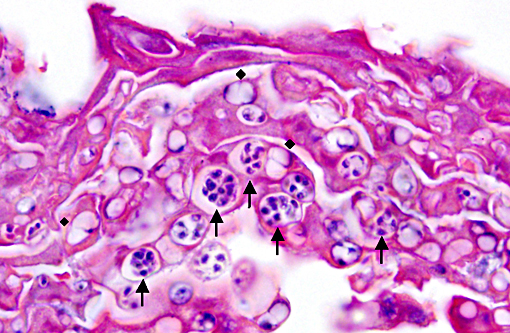
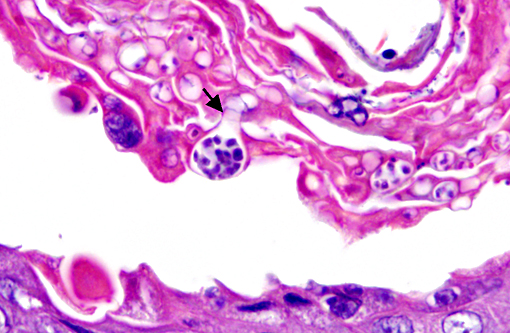
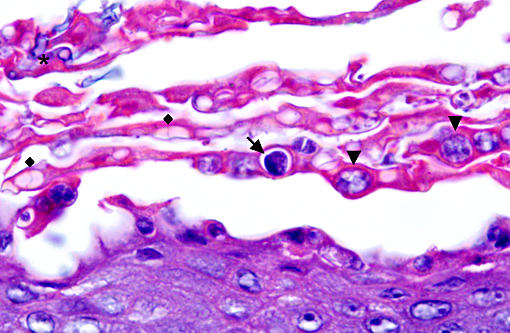
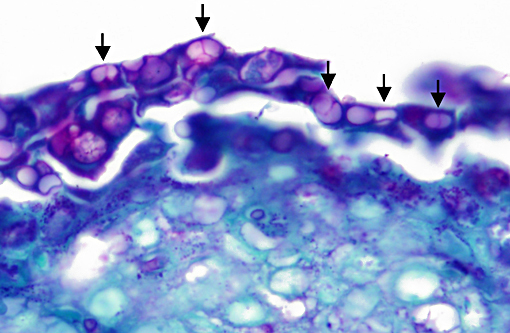
Histopathologic Description:
Morphologic Diagnosis:
Condition:
Contributor Comment:
Bd only affects keratinizing stratified squamous epithelium, which includes the mouthparts in tadpoles and the skin in adults. Clinical signs are variable and may include anorexia, lethargy, reluctance to place the ventrum on substrate, or even loss of righting reflex. Gross lesions most commonly occur on the ventrum and feet. Lesions include excessive skin shedding (dysecdysis), skin discoloration, and roughening of the skin. In tadpoles, depigmentation of the jaw sheath is commonly seen. Lack of gross lesions does not rule out chytridiomycosis, as was seen in this case.(5)
Histopathologic lesions of Bd include hyperplastic and hyperkeratotic epithelium containing the distinctive thalli within the keratin layers. There are 3 morphologic types of thalli which can range from 7 - 20 microns in diameter. Zoosporangia are the mature form, which contain 2 - 3 micron diameter basophilic zoospores and occasionally a flask-shaped discharge tube. Other types of thalli include a uninucleate stage with homogenous basophilic cytoplasm, and a multinucleated thallus with stippled to microvacuolated cytoplasm. Empty thalli with fine internal septations (colonial thalli) are zoosporangia that have discharged their zoospores and are a common finding. In many cases the empty thalli may be the only indication of Bd. Periodic acid-Schiff may be helpful in highlighting empty thalli, which could be interpreted to be an artifact in hematoxylin and eosin stained sections. Rhizoids are thin, root-like extensions from thalli and may be visible in silver-stained sections. Inflammation is usually minimal to absent. Secondary bacterial, fungal, and oomycete infections are common.(5)
The life cycle of Bd is well described. The zoospore is the infective stage and is released from a mature zoosporangium through the discharge tube. Transmission of the infectious motile flagellated zoospores occurs via direct animal contact or contact with water or substrates contaminated by affected animals. The zoospores become encysted in a new keratinocyte to develop into a thallus. Cleavage of the thallus results in the development of new zoospores within a zoosporangium. All stages can be found in the keratin layers of an infected animal.(5)
Infection with chytrid fungi lead to morbidity and mortality through the disruption of both innate and cell-mediated immunity and loss of the physiologic function of the skin. The normal skin flora, including Janthinobacterium lividum, produce the antifungal toxin, violacein, which may be important for innate immunity.(2) Cell-mediated immunity may also be affected by toxins released from the fungus that inhibit T and B lymphocyte proliferation and cause lymphocyte apoptosis.(1) The major contributing factor to death in affected animals is disruption of normal cutaneous function. The skin of frogs is important for water absorption, osmoregulation, and respiration. In experimental infections, frogs became hyponatremic, hypokalemic, hypomagnesemic, and hypochloremic compared to controls. Disruption of cutaneous electrolyte transport is considered responsible.(6) As in this animal, secondary fungal and bacterial infections leading to epidermal necrosis may also contribute to death.
Other methods for the diagnosis of chytridiomycosis include cytology and real-time polymerase chain reaction (PCR). Cytology may be useful in identification of thalli or zoospores in shed skin or a skin scraping; however, differentiation from yeasts and oomycetes can be difficult. Confirmation by histopathology or PCR may be necessary. PCR is most commonly used for screening animals in quarantine or prior to translocation, surveying captive and free-ranging populations, and confirming a positive result on wet mount or cytology. PCR is highly sensitive and may help identify subclinical cases.(5)
In addition to the lesion presented in this case, sections also demonstrate the Eberth-Kastschenko (EK) layer that is characteristic of many frog species. The EK layer is a deeply basophilic layer of calcium deposits within the dermis. The function of this layer is unclear but speculated to be involved with protection from dessication, storage and mobilization of calcium, or the interchange of substances between the animals internal and external environment.(3)
JPC Diagnosis:
Conference Comment:
The contributor provided extensive detail regarding the clinical presentation and pathogenesis of this entity. The discussed electrolyte disturbances induced by the loss of skin function often results in death due to cardiac arrest.(4) Interestingly, some species appear to be resistant to infection, including the well-studied Xenopus laevis which has contributed significantly to the understanding of the immune systems role in evoking this resistance. The immunoglobulins IgG and IgY secreted within the mucus from the skin appear to play a prominent role in providing protection. Their innate immune system also prevents colonization through the release of antimicrobial peptides, whose secretion is dramatically increased during periods of stress.(4)
References:
1. Fites JS, Ramsey JP, Holden WM, et al. The invasive chytrid fungus of amphibians paralyzes lymphocyte response. Science. 2013;342:366-369.
2. Harris RN, Brucker RM, Walke JB, et al. Skin microbes on frogs prevent morbidity and mortality caused by a lethal skin fungus. ISME J. 2009;3:818-824.
3. Katchburian E, Anoniazzi MM, Jared C, et al. Mineralized dermal layer of the Brazilian tree-frog Corythomantis greeningi. J Morphology. 2001;248:56-63.
4. Pessier AP. Chytridiomycosis. In: Current Therapy in Reptile Medicine and Surgery. St. Louis, MO: Saunders:2014:255-270.
5. Ramsey JP, Reinart LK, Harper LK, Woodhams DC, Rollins-Smith LA. Immune defenses against Batrachochytrium dendrobatidis, a fungus linked to global amphibian declines in the South African clawed frog, xenopus laevis. Infect Immun. 2010;78(9):3981-3992.
6. Voyles J, Young S, Berger L, et al. Pathogenesis of chytridiomycosis, a cause of catastrophic amphibian declines. Science. 2009; 326:582-585.