Joint Pathology Center
Veterinary Pathology Services
Wednesday Slide Conference
2018-2019
Conference 21
24 March 2019
CASE II: A-231/15 B (JPC)
Signalment: Meat type chicken (Gallus gallus domesticus, broiler), 3 weeks old.
History: Sudden onset of mortality affecting 10% of the flock. Sick birds adopt a crouching position with ruffled feathers and die within 48 hours.
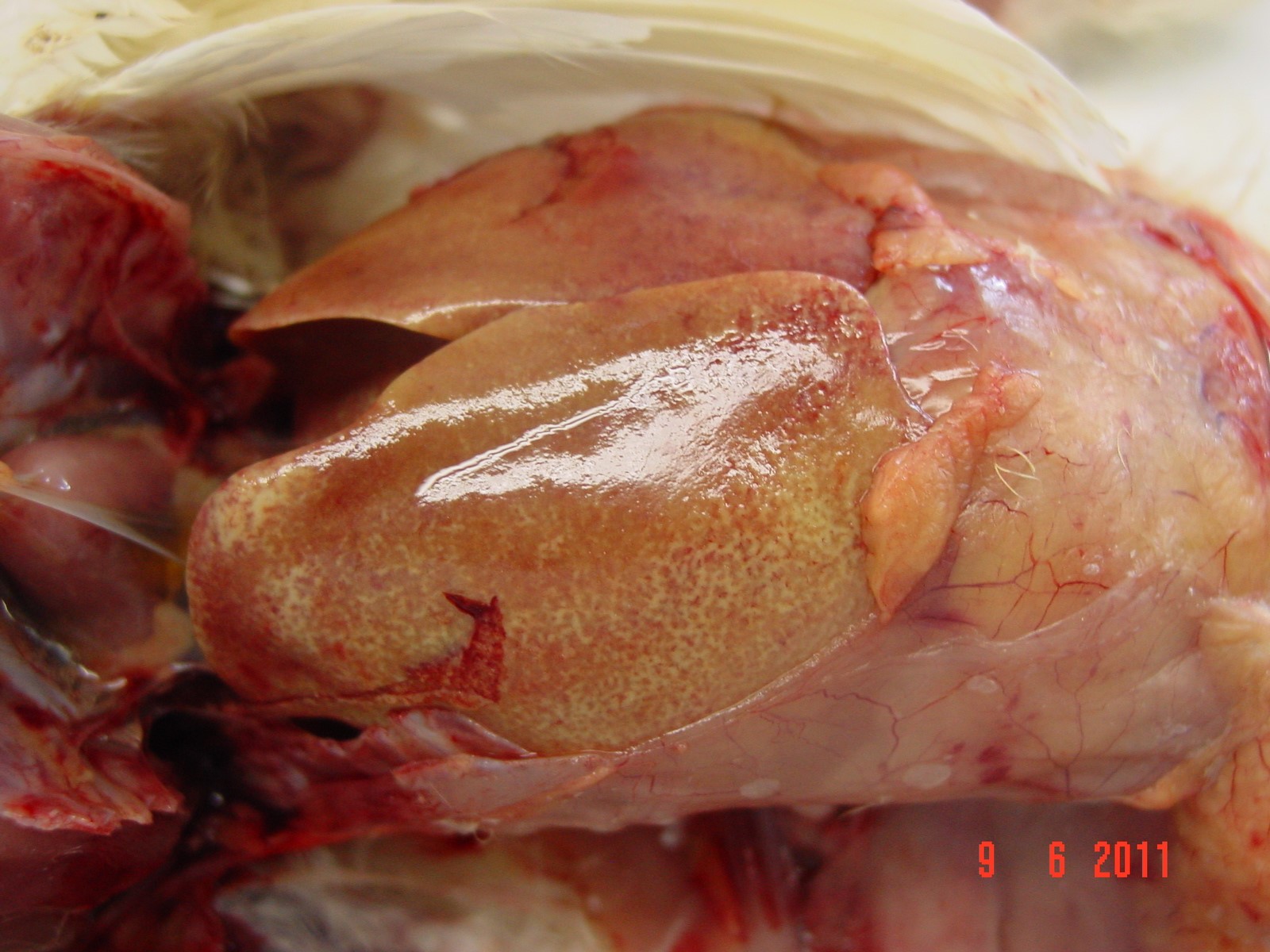
Gross Pathology: At necropsy, diffuse yellowish-pale, friable and swollen livers are seen. Multiple petechiae beneath the capsule are present in some livers.
Laboratory results: None given.
Microscopic Description:
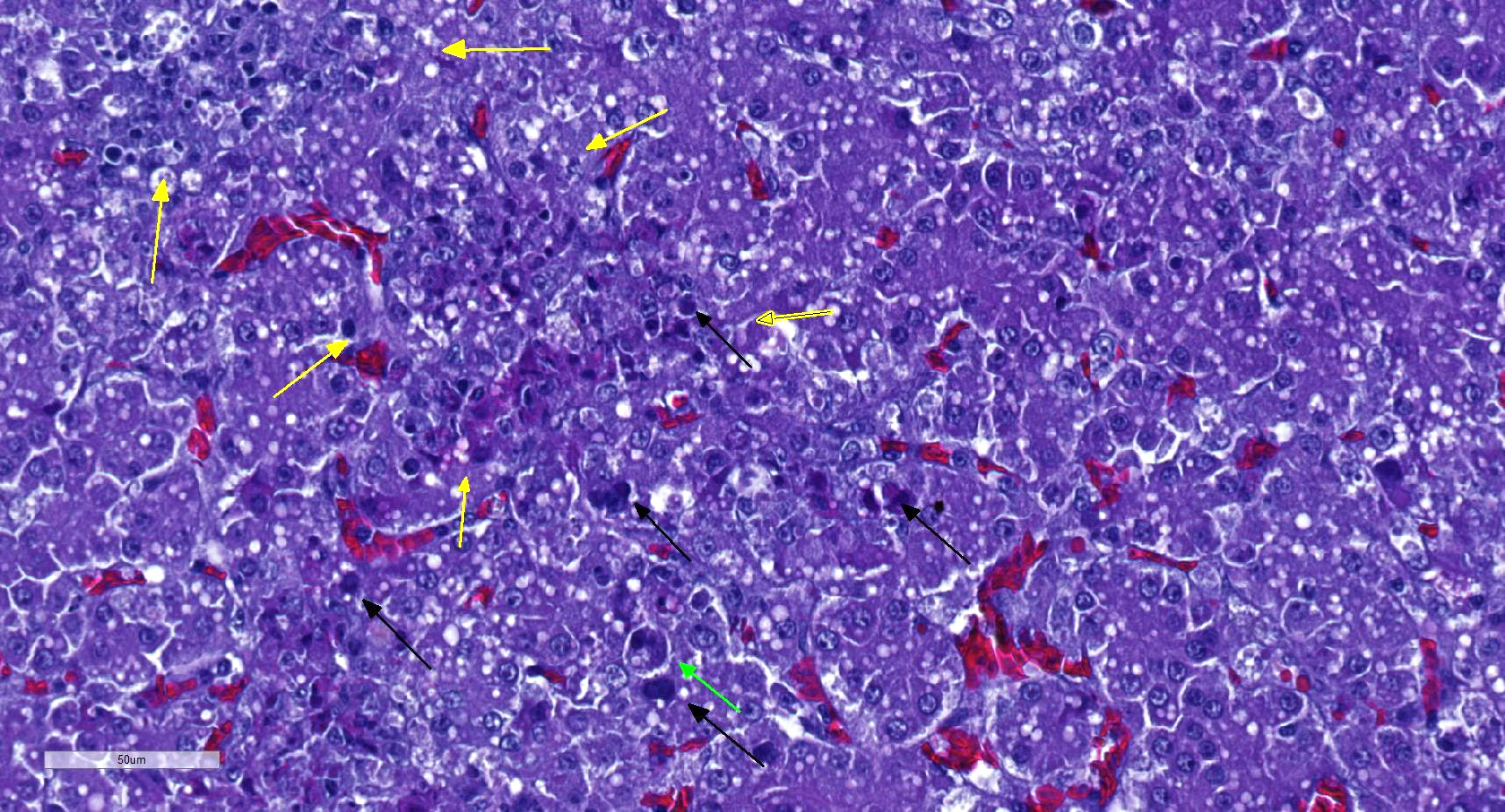
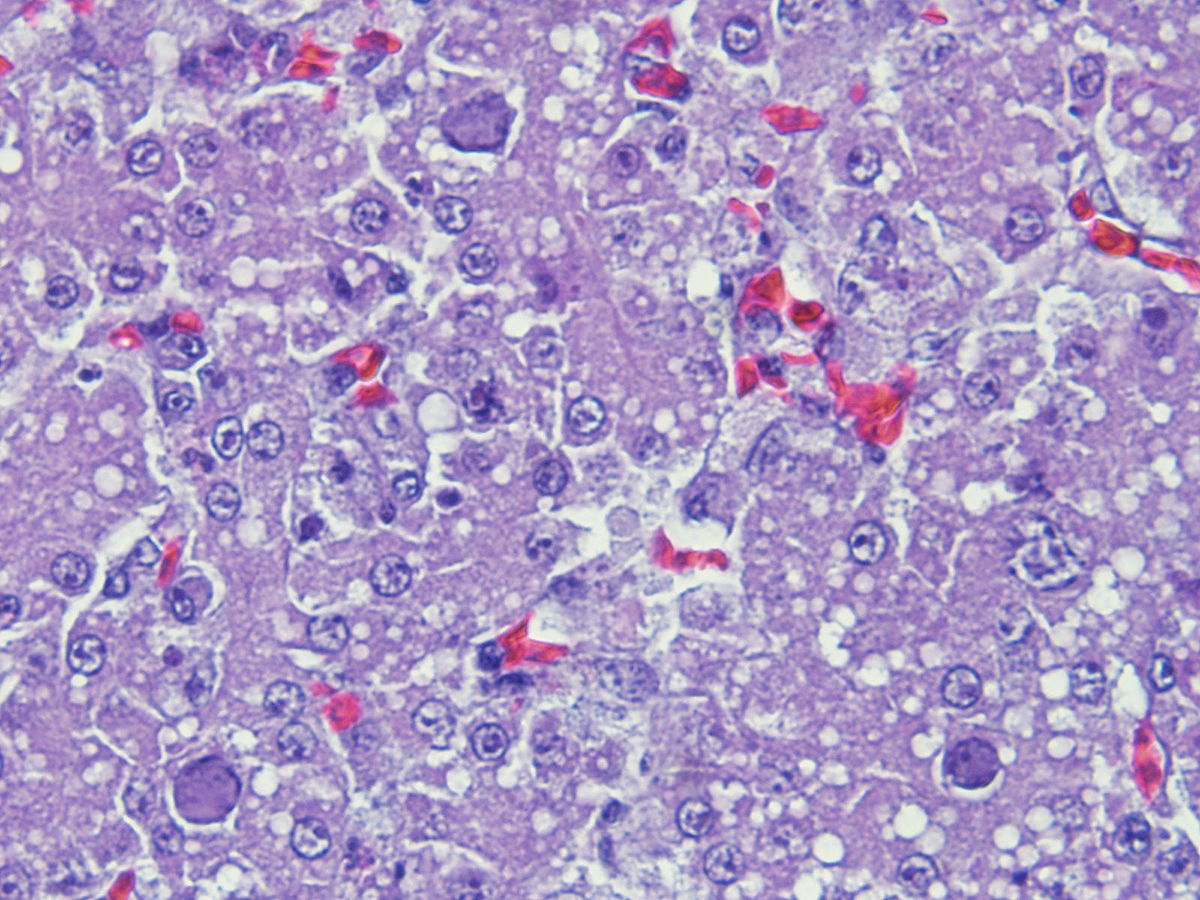
There is a disruption of the hepatic parenchyma due to the presence of multifocal to coalescing randomly distributed foci of degenerated hepatocytes. These hepatocytes are swollen and show hypereosinophilic and highly vacuolated cytoplasm, and pyknotic nucleous with karyorrhexis and/or karyolysis. Associated to these foci and randomly scattered throughoutt the parenchyma some hepatocytes present marked karyomegaly with chromatin condensation at the nuclear membrane and large basophilic intranuclear inclusion bodies. Moderate lymphoplasmacytic and heterophilic inflammatory infiltrate in the periportal areas is observed. Cytoplasmic vacuolation is diffusely present in remaining hepatocytes. Occasionally, focal widening and infiltration of sinusoids with lymphocytes, heterophils and histiocytes are present.
Contributor’s Morphologic Diagnosis:
Acute, severe, multifocal to coalescing necrotizing hepatitis with intranuclear inclusion bodies in hepatocytes
Contributor’s Comment: Inclusion body hepatitis (IBH) is a viral disease produced by a member of the family Adenoviridae, genus Aviadenovirus,1 which was first described in chickens by Helmboldt and Frazier in 1963.3 IBH is a ubiquitous disease in commercial and farm birds2, although in the last years the infection has been proven in wild and exotic birds, producing the same characteristic hepatic lesions.3
The liver is the primary organ affected.1 The infection produces a multifocal necrotizing hepatitis with intranuclear inclusion bodies in the hepatocytes.1,5 Intranuclear inclusion body description are variable: large, eosinophilic or basophilic, round or irregular shaped, but always replacing and displacing peripherally the chromatin, and producing karyomegaly.1
Contributing Institution:
Servei de Diagnostic de Patologi Veterinaria, Facultat de Veterinaria, Bellaterra (Barcelona), 08193 Spain.
JPC Diagnosis: Liver: Hepatitis, necrotizing, multifocal and random, mild to moderate with numerous intrahepatocytic nuclear viral inclusions.
JPC Comment: Within the aviadenoviruses, there are six species (FAdV A-E and good adenoviruses, and twelve FAdV serotypes. Although all 12 serotypes have been identified in outbreaks of inclusion body hepatitis, FAdV species D and E are most often isolated. The virus may be transmitted both vertically and horizontally, and is environmentally resistant when passed within the feces or by aerosol.
The clinical course of the disease is based on the virulence of the viral strain, age of the infected birds, as well as the presence of other viruses, to include immunosuppressive viruses such as avian circovirus and avian bunyavirus. The disease hits younger birds harder with mortalities of up to 85% in 2-day old chicks, but mortality averages 10% in 3-5 week old birds. Clinical signs are non-specific with depression and watery droppings. Grossly, livers are pale, swollen and friable. PCR for FAdV and typing is required for definitive diagnosis; the identification of FadV in feces is not diagnostic, and FAdV are considered ubiquitous.
In more recent years, cases of hydropericardium syndrome, with and without associated IBH have been identified as resulting from infection with FAdV serotype 4. These cases are characterized by accumulation of straw-colored fluid within the pericardial sac, nephritis, and hepatitis with mortality rates of up to 70%.7
In addition to the inclusion body/hydropericardium syndrome, aviadenoviruses result in a number of other syndromes in chickens including necrotizing pancreatiis as well as gizzard erosions. Fowl aviadenovirus type A has been identified in outbreaks in Europe and Japan, resulting in necrosis and hemorrhage within the koilin layer, as well as adenoviral inclusions within the epithelium. Random foci of hemorrhage, unassociated with viral inclusions are also seen in this condition.
The moderator commented on the difficulties of differentiating propagating intranuclear crystalling arrays from nucleoli (which are especially prominent in turkeys), and mentioned that it is imperative to see large karyomegalic basophilic viral inclusions characteristic of adenovirus before diagnosing this condition. He also emphasized that due to the ubiquitous nature on non-pathogenic adenovirus in many flocks, isolation of an adenovirus in poultry is of little clinical importance in the absence of disease. He also commented on the presence of glomerulonephritis in many cases of inclusion body hepatitis and the need to examine kidneys in these cases.
References:
- Adair BM, Fitzgerald SD. Group I Adenovirus infections. In: Diseases of Poultry. 12th Iowa State Press; 2008:252-266.
- Garmyn A, Bosseler L, Braeckmans D, Van Erum J, Verlinden M. Adenoviral gizzard erosions in two Belgian broiler farms. Avian Dis 2018; 62(3):322-325.
- Hollell JD, McDonald W, Christian RG. Inclusion body hepatitis in chickens. Can Vet J 1970; 11:99-101.
- Ramis A, Marlasca MJ, Majo N, Ferrer L. Inclusion body hepatitis (IBH) in a group of eclectus parrots (Eclectus roratus). Avian Pathol 1992; 21(1):165-9.
- Randall CR and Reece RL. Color Atlas of Avian Histopathology. Mosby-Wolfe, Times Mirror International Publishers Limited; 1996: 95-96.
- Yugo DM, Hauck R, Shivaprasad HL, Meng XJ. Hepatitis virus infectious in poultry. Avian Dis 2016; 60:576-588.
- Zhao J, Zhong Q, Zhao Y, Hu Y, Zhang GZ. Pathogenicity and complete genme characterization of fowl adenoviruses isolated from chickens associated with inclusion body hepatitis and hydropericardium syndrome in China. Plos One 2015 doi 10.1371/journal.pone.0133073