Signalment:
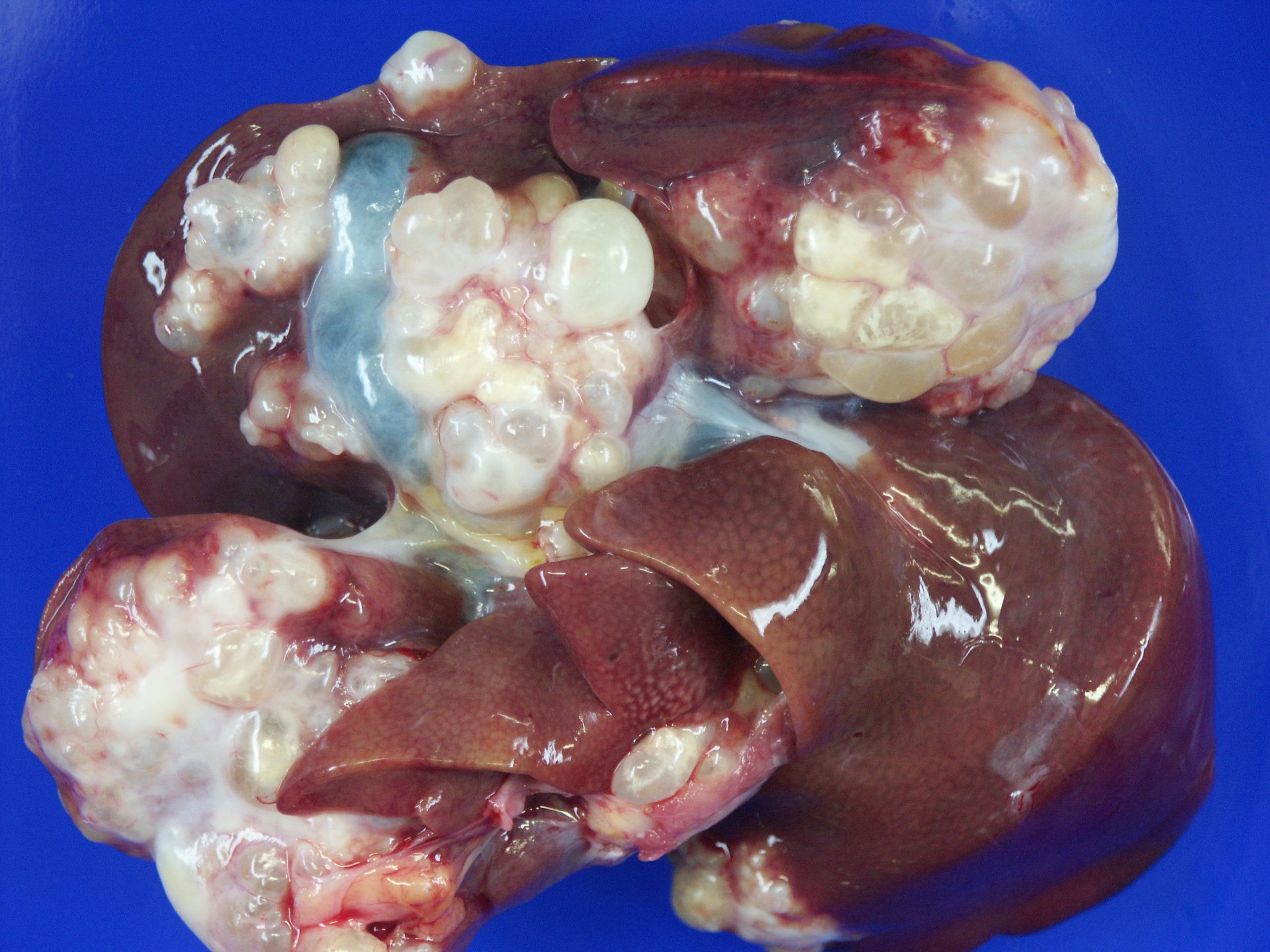
Gross Description:
Histopathologic Description:
Morphologic Diagnosis:
Lab Results:
Condition:
Contributor Comment:
The alveolar echinococcosis (AE) is an infectious disease which is caused by the second larval stage (metacestode) of the fox tapeworm. The adult parasite lives in the gastrointestinal tract of foxes of the genera Vulpes and Alopex, which are the definitive hosts. The eggs, including the first larval stage (oncosphere), get outside with the feces and are ingested by intermediate hosts, which typically are rodents of the family Arvicolidae. In the intermediate host, the oncosphere penetrates the intestinal wall and enters the blood system. Via the blood stream, the oncosphere reaches different organs, especially the liver. Once the oncosphere has reached the liver it starts to develop into the metacestode stage. In contrast to E. granulosus with the development of a unilocular cyst, the typical cyst of E. multilocularis shows a multilocular structure. The cyst with brood capsules and protoscolices may disturb the functions of the liver, depending on its size and location. The development of protoscolices can take several months. There may be several thousand protoscolices within a cyst. If protoscolices are ingested by a definitive host, they develop to the sexually mature adult tapeworm, approximately four to six weeks after infection. Thus, the cycle is closed(5).
All mammals (including man and nonhuman primates) in which metacestodes develop may be an intermediate host, but it is important to distinguish between a real intermediate host, which plays a role in the perpetuation of the cycle, and an accidental intermediate host like this monkey, which is a dead end for the parasite.
Many monkey species are susceptible to infection with E. multilocularis(1,2,6,7). In recently described small outbreaks, only a few species were affected (five M. fascicularis and two Gorilla gorilla(7) or 12 M. fuscata(8)). A larger outbreak at the German Primate Center affected three different Old World monkey species simultaneously in a period of 12 years(9). In this largest reported outbreak, cynomolgus monkeys are the species at risk. As previously reported, the percentage of infected cynomolgus monkeys among colonies of captive primates was conspicuously high (> 50%)(3).
Since the 1990s, E. multilocularis infection is spreading geographically and increasing infection rates of red foxes have been noted in Eastern and Western European countries(4). In Northern Germany, Denmark and Poland, prevalence rates in foxes are usually < 5%, but focal areas of higher prevalence exist. Animals in zoological gardens and in institutional colonies in the northern hemisphere are at risk and alveolar echinococcosis must be considered as an emerging disease. Certain species of non-human primates are very susceptible to alveolar echinococcosis and may thus indicate previously unknown areas of high transmission.
JPC Diagnosis:
1. Liver: Hydatid cyst, multiloculated, with hepatocellular loss and fibrosis, and mild granulomatous hepatitis.
2. Liver: Amyloidosis, diffuse, moderate.Â
Conference Comment:
References:
2. Brack M, Tackmann K, Conraths FJ, Rensing S. Alveolar hydatidosis (Echinococcus multilocularis) in a captive rhesus monkey (Macaca mulatta) in Germany. Trop Med Int Health 2(8):754-759, 1997
3. Deplazes P, Eckert J. Veterinary aspects of alveolar echinococcosis-a zoonosis of public health significance. Vet Parasitol 98(1-3):65-87, 2001
4. Eckert J, Conraths FJ, Tackmann K. Echinococcosis: an emerging or re-emerging zoonosis? Echinococcosis: an emerging or re-emerging zoonosis? Int J Parasitol 30(12-13):1283-1294, 2000
5. Meyers WM, Neafie, RC, Marty AM, Wear DJ. Hydatidosis. In: Pathology of Infectious diseases, Vol. I Helminthiasis, pp. 145-164. Armed Forces Institute of Pathology, 2000
6. Rehmann P, Grone A, Lawrenz A, Pagan O, Gottstein B, Bacciarini LN. Echinococcus multilocularis in two lowland gorillas (Gorilla g. gorilla). J Comp Pathol 129:85-88, 2003
7. Rehmann P, Grone A, Gottstein B, Sager H, Muller N, Vollm J, Bacciarini LN.: Alveolar echinococcosis in the zoological garden Basle. Schweiz Arch Tierheilkd 147(11):498-502, 2005
8. Sato C, Kawase S, Yano S, Nagano H, Fujimoto S, Kobayashi N, Miyahara K, Yamada K, Sato M, Kobayashi Y. Outbreak of larval Echinococcus multilocularis infection in Japanese monkey (Macaca fuscata) in a zoo, Hokkaido: western blotting patterns in the infected monkeys. J Vet Med Sci 67:133-135, 2005
9. Tappe D, Brehm K, Frosch M, Blankenburg A, Schrod A, Kaup F-J, M+�-�tz-Rensing K. Echinococcus multilocularis infection of several Old World monkey species in a breeding enclosure. Am J Trop Med Hyg.77(3):504-506, 2007
10. Zachary JF. Mechanisms of microbial infections. In: Zachary JF, McGavin MD, eds. Pathologic Basis of Veterinary Disease. 5th ed. St. Louis, MO: Elsevier; 2012:287-8.