Signalment:
Gross Description:
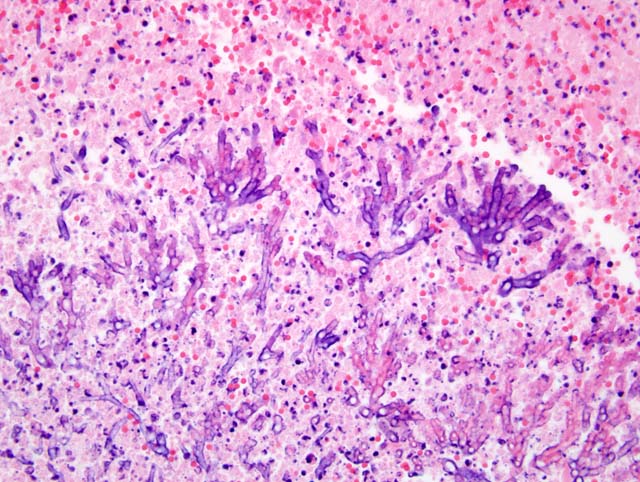
Histopathologic Description:
Morphologic Diagnosis:
Lab Results:
Condition:
Contributor Comment:
Pathogenic factors of Candida species are important in determining their relative virulence in the host.(3) Candida albicans can invade the columnar epithelium of the intestines.(3) The yeast form of C. albicans colonizes epithelia, while hyphae are the more invasive form and are found within deeper tissue invasion.(3)
Local proliferation of Candida species on mucosal surfaces is the first step in the spread of infection. Overgrowth of Candida species within the gastrointestinal tract is inhibited by mucosal microflora.(1) Factors that upset the balance of normal endogenous microflora may cause Candida organisms to proliferate. Intestinal candidiasis may be sequelae to parvoviral infections or alterations in microflora caused by systemic antibiotic therapy.(3,1,5) Mycotic ileitis and colitis in cats have been reported as a secondary complication of infection with feline panleukopenia virus.(5,7) Localized candidiasis has been reported to occur in chronically immunosuppressed cats and in those with nonhealing ulcers of the gastrointestinal tract.(3) However, in one study retroviral status (feline leukemia virus, FeLV and feline immunodeficiency virus, FIV) did not influence the ability to isolate C. albicans from feline cutaneous and mucosal samples.(6) The FeLV and FIV status of this patient was not established and routine bloodwork, that may have demonstrated panleukopenia, was not performed.
Histologic examination of lesions reveals multifocal abscesses or areas of necrosis that contain abundant blastoconidia, pseudohyphae and true hyphae surrounded by mixed inflammatory cells.(3) Infiltrates tend to be minimal in profoundly immunosuppressed or leukopenic animals.(3) The proprial infiltrate in the cecum from this cat would suggest it was not immunosuppressed or leukopenic.
Candida species grow well on blood agar and are often isolated from specimens submitted for bacterial culture. Organisms may sometimes be cultured from many tissues at surgery or necropsy, but such results should be interpreted cautiously.(3) Culture of Candida species from cutaneous, mucosal or exudates alone are not an indicator of infection.(3) Histologic confirmation of invasion and host reaction are essential.(3) Mucosal and cutaneous biopsies should be submitted for histologic and culture examination simultaneously. A definitive histologic diagnosis can sometimes be made in the presence of negative culture results.
JPC Diagnosis:
1. Cecum (per contributor): Typhlitis, necrotizing and pyogranulomatous, diffuse, severe, with vasculitis, fibrin, hemorrhage, edema, intralesional hyphae and pseudohyphae, and rare eosinophilic intranuclear inclusion bodies.
2. Lymph node: Lymphoid depletion, diffuse, severe, with draining hemorrhage, edema, and rare eosinophilic intranuclear inclusion bodies.
Conference Comment:
| PV | FAE | CoV | EUE | |
| Necrosis | crypts | crypts (less severe) | villar | crypts |
| Inflammation | â (T cells) | â â (T cells) | â â (lymphocytes) | â â (macrophages) |
| Lymphoid tissue | depleted | Normal to hyperplastic | depleted | |
| Bone marrow | â | Slight â | Slight â | Slight â |
References:
2. Gelberg HB: Alimentary system. In: Pathological Basis of Veterinary Disease, eds. McGavin MD, Zachary JF, 4th ed., pp. 378-380. Mosby, St. Louis, MO, 2007
3. Greene CE and Chandler FW. Candidiasis and Rhodotorulosis. In: Infectious Diseases of the Dog and Cat, ed. Greene CE, 3rd ed., pp. 627- 632. Saunders Elsevier, London, UK, 2006
4. Kipar A, Kremendahl J, Jackson ML, Reinacher M: Comparative examination of cats with feline leukemia virus-associated enteritis and other relevant forms of enteritis. Vet Pathol 38:359-371, 2001
5. Lorenzini R and De Bernardis F: Antemortem diagnosis of an apparent case of feline candidiasis. Mycopathologia 93:13-14, 1986
6. Ochiai K, Valentine B and Altschul M: Intestinal candidiasis in a dog. Vet Rec 146:228-229, 2000
7. Sierra P, Guillot J, Jacob H, Bussieras S, Chermette R: Fungal flora on cutaneous and mucosa surfaces of cats infected with feline immunodeficiency virus or feline leukemia virus. American Journal of Veterinary Research 61:158-161, 2000