Joint Pathology Center
Veterinary Pathology Services
Wednesday Slide Conference
2017-2018
Conference 21
April 11th, 2018
CASE III: G071 (JPC 4100982).
Signalment: 4-week-old, female, outbred Hartley guinea pig (Cavia porcellus), guinea pig.
History: Experimentally infected with Ebola virus (guinea pig adapted Mayinga isolate) by intraperitoneal route 8 days prior to necropsy.
Gross Pathology: The animal was mildly dehydrated. The liver was pale and friable. Lungs were mottled and multifocally hemorrhagic. The gastrointestinal tract was filled with digested blood.
Laboratory Results (clinical pathology, microbiology, PCR, ELISA, etc.): None provided.
Microscopic Description:
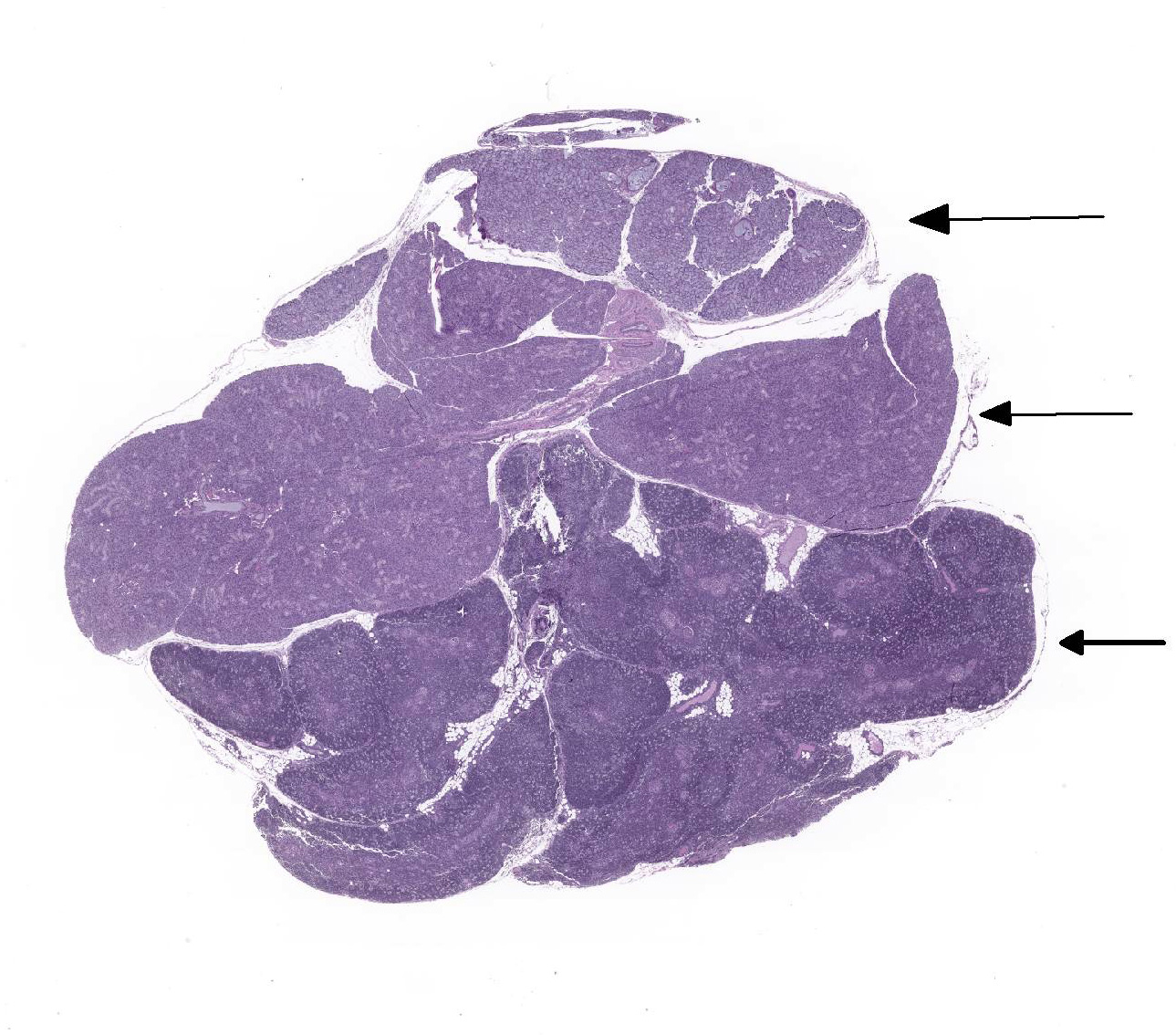
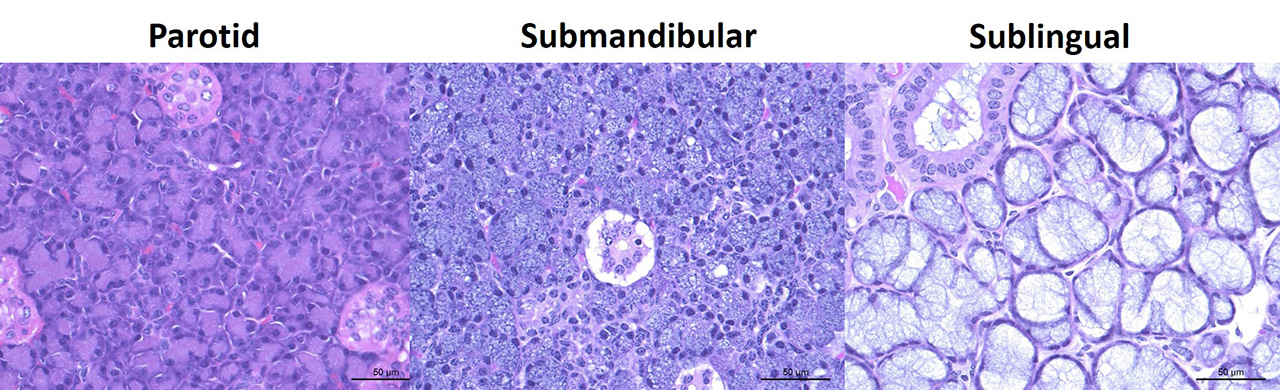
Slide contains sections of thymus and sublingual and parotid salivary glands. In the thymus, there is moderate diffuse lymphocytolysis with tingible body macrophages containing apoptotic cellular debris in the cortex and medulla, with blurring of the distinction between the two. Low numbers of macrophages with abundant vacuolated cytoplasm and small to large eosinophilic intracytoplasmic inclusion bodies are present in the medulla and occasionally cortex. Within the center of the Hassall’s corpuscles, there is pyknotic nuclear dust admixed with viable and degenerate heterophils and cornified epithelial cells, with scattered rare coarse mineralized concretions (normal feature of this species).
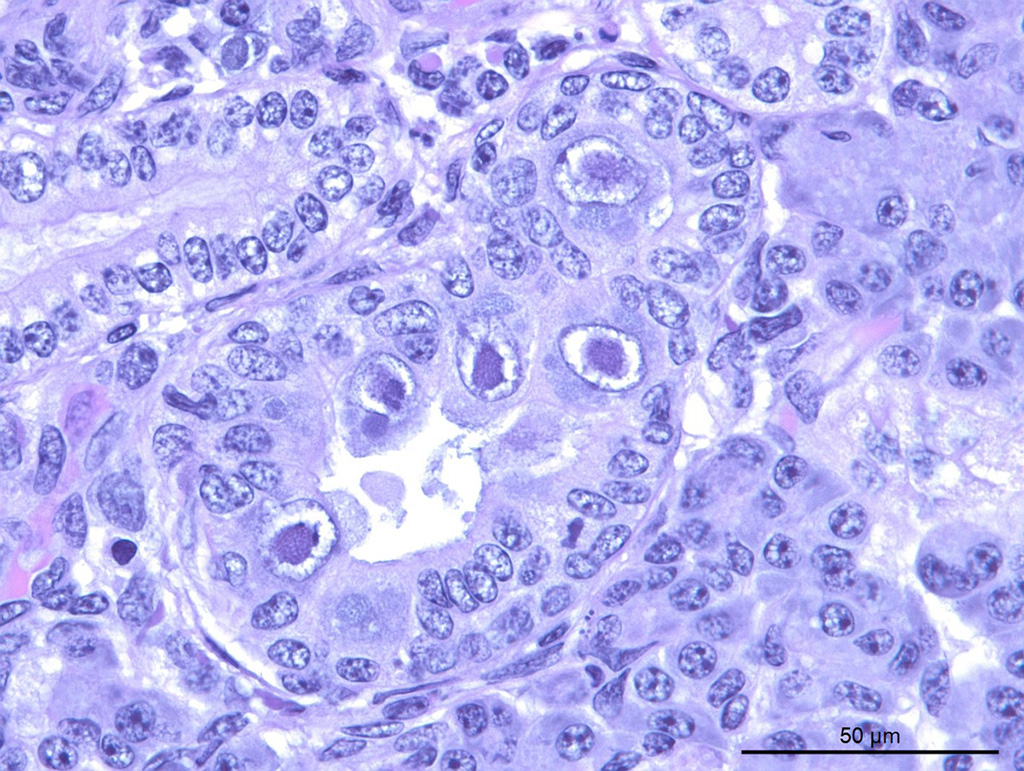
Within the both salivary glands there are low numbers of individual necrotic ductal cells. There are scattered individual and small clusters of apoptotic and necrotic acinar cells with rare ICIB. There are low numbers of vacuolated and inclusion bearing macrophages and fibroblasts in the interacinar and periductal interstitium. Within several small and medium ducts in the parotid gland, there are karyomegalic ductal epithelial cells containing very large eosinophilic to amphophilic (owl’s eye) intranuclear inclusions with a clear halo and peripheral margination of the chromatin (Cowdry type A). In both salivary glands there are several medium ducts that are mildly dilated and filled with wispy (sublingual) to homogenous (parotid) pale basophilic acellular material (inspissated saliva).
Contributor’s Morphologic Diagnosis:
- Thymus, lymphocytolysis, diffuse, moderate, with intrahistiocytic intracytoplasmic inclusion bodies
- Salivary glands, parotid and sublingual, sialoadenitis, necrotizing and histiocytic, multifocal, acute, mild with intracytoplasmic inclusion bodies
- Salivary gland, parotid, ductal epithelial cell karyomegaly, multifocal, mild with intranuclear inclusion bodies
Contributor’s Comment: Ebola virus, a filovirus, is an important high consequence pathogen causing significant human disease outbreaks, most recently in West Africa from 2014 to 2016, and again in 2017. Guinea pigs remain an important model for infectious disease research, and have a number of unique anatomical features. The guinea pig thymus is located in the cervical region, and the pustule like appearance of the Hassall’s corpuscles is normal for this species.1 Lesions in the thymus are consistent with experimental manipulations.4,5 Macrophages are a main target cell type of Ebola virus infection, with frequent (and often severe) bystander necrosis of lymphocytes. Ebola does not infect lymphocytes.9,10 Lymphocytolysis of the thymic cortex can be seen in a number of infectious disease processes, as well as corticosteroid administration or stress.
Lesions in the salivary glands represent two distinct processes, and are a mixture of experimental manipulation and adventitious spontaneous (background) infection. There is apoptosis and necrosis of ductal and acinar epithelial cells, with viral cytoplasmic inclusions present in acinar epithelial cells as well as interstitial fibroblasts and macrophages. These lesions are consistent with experimental Ebola virus infection, and saliva may transmit the virus during acute infections. Saliva is also one of several fluids that may remain positive for the Ebola genome in convalescent human disease survivors, although virus generally cannot be isolated from the saliva.2 Human disease transmission via semen from recovered individuals has been documented.3
Intranuclear inclusions in the parotid ductal epithelium are consistent with caviid betaherpesvirus-2 (guinea pig cytomegalovirus).1 In other species (rat, mouse), polyomavirus would be a differential etiology; guinea pigs have no described polyomavirus. Caviid betaherpesvirus-2 is not excluded by the supplier of these animals, and CMV is typically an incidental finding in this species. Systemic disease can occur in pregnant or weaned pigs. There were no CMV type inclusions in any other organ sampled in this guinea pig, nor in any of the other pigs in this study.
JPC Diagnosis:
- Thymus, lymphocytes: Apoptosis, diffuse, moderate, with numerous tingible body macrophages, outbred Hartley guinea pig (Cavia porcellus), guinea pig.
- Thymus & salivary gland, macrophages: Intracytoplasmic viral inclusions, occasional.
- Salivary gland, glandular epithelium: Necrosis, multifocal, minimal to mild.
- Salivary gland, ductal epithelium: Rare, intranuclear, karyomegalic viral inclusions.
Conference Comment: The family Filoviridae (negative-sense RNA viruses) has three genera: Marburgvirus (Marburg and Ravn viruses), Ebolavirus (Sudan virus, Ebola virus, Reston virus, Bundibugyo virus, and Tai Forest virus), and Cuevavirus (Lloviu virus). Filoviruses enter cells via macropinocytosis with subsequent binding to Niemann-Pick C1 (NPC1) receptor protein (a host cholesterol transport protein). Other cellular attachment molecules include: C-type lectins, phosphatidylserine, actin filaments, and cellular microtubules. Virus replication takes place in the cytoplasm and form prominent intracytoplasmic inclusion bodies. Viral maturation occurs via budding of preassembled nucleocapsids from the plasma membrane. As RNA viruses, Filoviruses mutate rapidly within infected individuals or reservoirs (different species of fruit bats).7 The mutation rate has been calculated to be similar to seasonal influenza at 2.0 x 10-3 substitutions per site per year.6
Non-human primates (NHPs) are highly susceptible to filovirus infections with outbreaks reported in wild gorillas (Gorilla gorilla) and chimpanzees (genus Pan). The incubation period is about 3-6 days, followed by onset of clinical disease characterized by petechiae, ecchymoses, hemorrhagic pharyngitis, hematemesis, melena, and prostration. The pathogenesis of filovirus infections is similar in NHPs and humans but the clinical course of disease in NHPs is shorter and almost always ends in death. Mice and guinea pigs are not susceptible to field strains of Marburg virus or Ebola virus but rodent-adapted strains have been developed for vaccine and therapeutics testing.8
In experimentally infected NHPs, filoviruses replicate in macrophages, dendritic cells, and endothelium resulting in dissemination throughout the body and necrosis of various organs which is most flagrant in the liver. Infected monocytes, macrophages, and dendritic cells also release inflammatory mediators like tumor necrosis factor and interleukin-8, as well as, nitric oxide which effect vascular permeability and coagulation, and tissue factor from infected macrophages and monocytes. Additionally, loss of functional hepatocytes yields reduced synthesis of clotting factors and further aggravation of dysfunctional hemostasis dramatically ending with disseminated intravascular coagulation.8
Conference participants failed to recognize the eosinophilic intracytoplasmic viral inclusion bodies in macrophages and fibroblasts within the salivary gland. According to the moderator (who is also the contributor of this case), viral inclusions were seen in the glandular epithelial cells as well with immunohistochemistry; the saliva is also a way that this virus may be spread. ,potentially contributing to virus spread via saliva. Ebolavirus results in lymphocytolysis in the thymic cortex indirectly as “bystander necrosis”. The main microscopic differential for this lesion is stress-induced lymphocytolysis.
Contributing Institution:
Pathology Department
NIH/NIAID
Integrated Research Facility
https://www.niaid.nih.gov/about/integrated-research-facility
References:
- Barthold, S.W., S.M. Griffey, and D.H. Percy, Guinea Pig, in Pathology of Laboratory Rodents and Rabbits, S.W. Barthold, S.M. Griffey, and D.H. Percy, Editors. 2016, Wiley Blackwell: Ames, IA. p. 213-252.
- Brainard, J., et al., Presence and Persistence of Ebola or Marburg Virus in Patients and Survivors: A Rapid Systematic Review. PLoS Negl Trop Dis, 2016. 10(2): p. e0004475.
- Christie, A., et al., Possible sexual transmission of Ebola virus - Liberia, 2015. MMWR Morb Mortal Wkly Rep, 2015. 64(17): p. 479-81.
- Connolly, B.M., et al., Pathogenesis of experimental Ebola virus infection in guinea pigs. J Infect Dis, 1999. 179 Suppl 1: p. S203-17.
- Cross, R.W., et al., Modeling the Disease Course of Zaire ebolavirus Infection in the Outbred Guinea Pig. J Infect Dis, 2015. 212 Suppl 2: p. S305-15.
- Jenkins GM, Rambaut A, Pybus OG, Holmes EC. Rates of molecular evolution in RNA viruses: A quantitative phylogenetic analysis. Journal of Molecular Evolution. 2002; 54(2): 156-165.
- Klapper, CE. The development of the pharynx of the guinea pig with special emphasis on the morphogenesis of the thymus. Am J Anat. 1946;78:139-180.
- MacLachlan NJ, Dubovi EJ. Filoviridae. In: Fenner’s Veterinary Virology. 5th ed. San Diego, CA: Elsevier; 2017:373-380.
- Prescott, J.B., et al., Immunobiology of Ebola and Lassa virus infections. Nat Rev Immunol, 2017. 17(3): p. 195-207.
- Younan P, Iampietro M, Bukreyev A. Disabling of lymphocyte immune response by Ebola virus. PLoS Pathog. 2018;14(4):e1006932.