Signalment:
Five-month-old,
female, Coton de Tulear, dog (
Canis familiaris).The dog
presented with ataxia evolving since the age of four months, with a rapid onset
of clinical signs. Neurological examination oriented towards a cerebellar
origin of the ataxia. Magnetic Resonance Imaging revealed a decreased size of
the cerebellum, without signs of inflammation. The dog was euthanized after
ataxia had worsened.
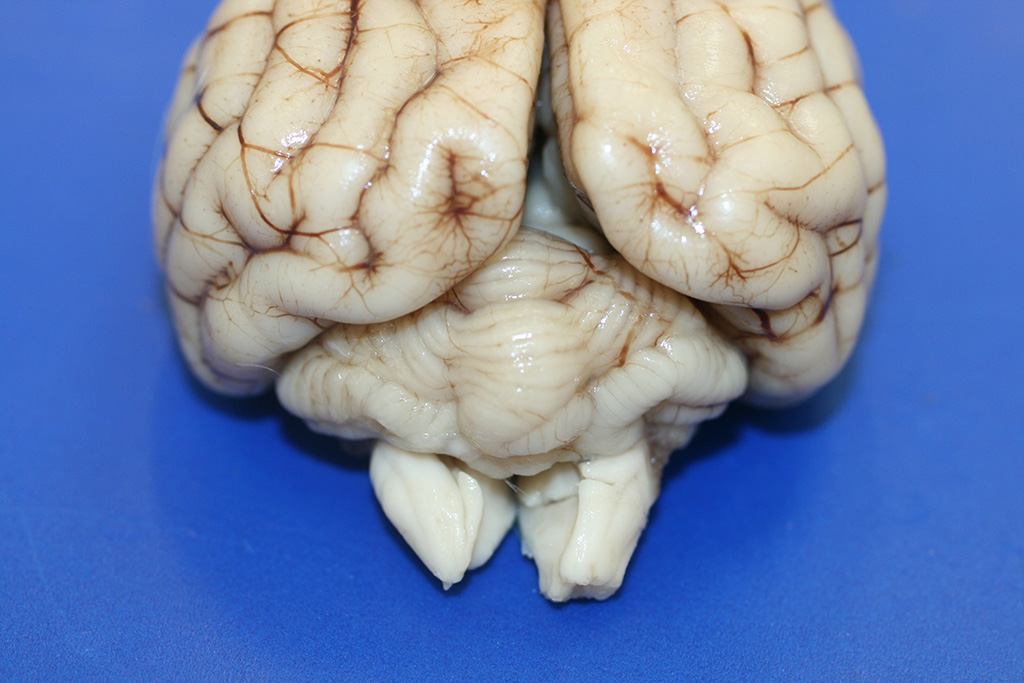
Gross Description:
The
only significant gross lesion at necropsy was a reduction in size of the
cerebellum, with slight asymmetry between the two hemispheres. The gyri were
diffusely sharply delineated and shrunken.
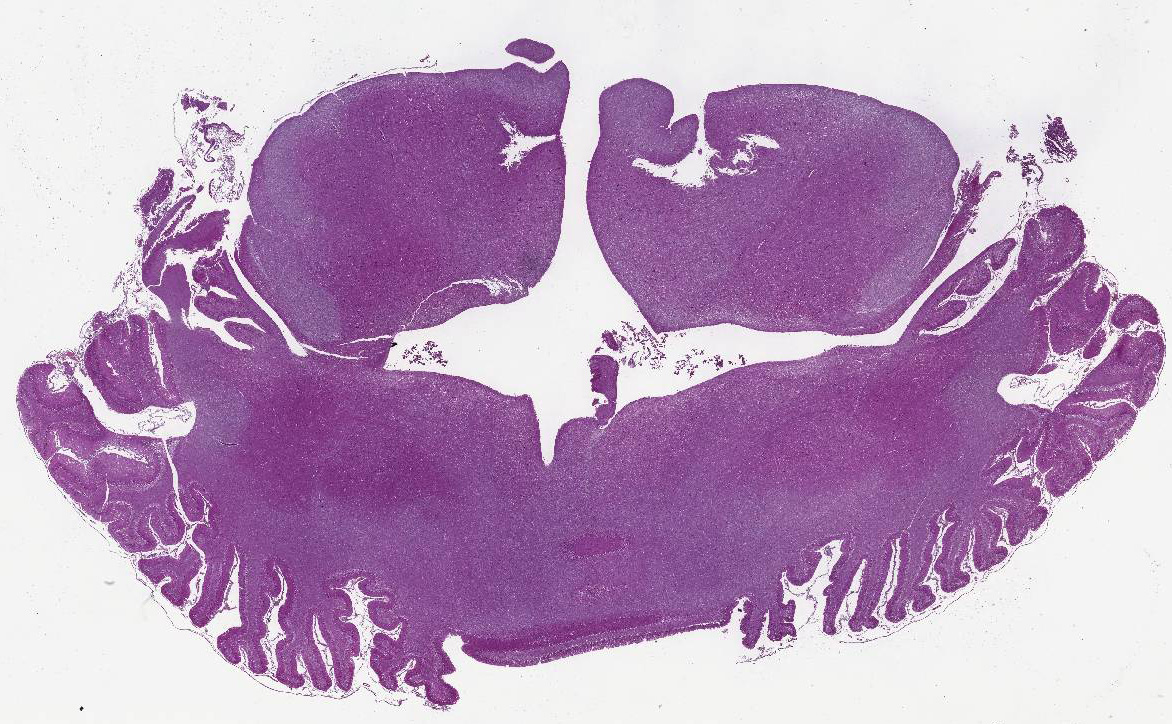
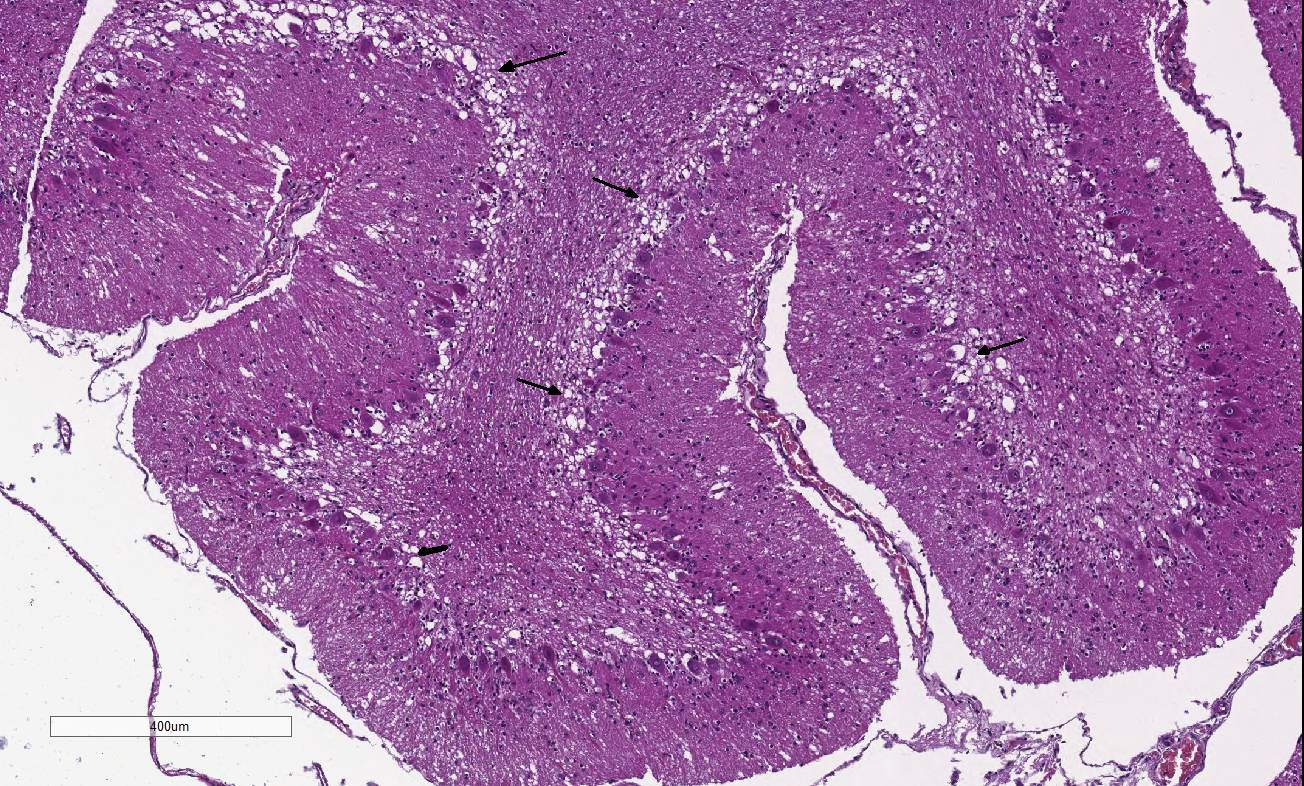
Histopathologic Description:
Diffusely,
the cerebellar folia are slightly flattened. Severe loss of cells within the
granular layer is present, multifocally leading to complete absence of this
layer. Rarely, granular cells are swollen and vacuolated (degeneration),
multifocally associated with empty baskets. Diffusely replacing this layer are
glial cells (mainly astrocytes). A discrete population of microglial cells is
also present. The molecular layer is mostly normal in thickness, or more rarely
thinner. Purkinje cells do not show remarkable degenerative changes.
Morphologic Diagnosis:
Cerebellum, Granular cell degeneration and loss,
diffuse, severe, Coton de Tulear, canine.
Lab Results:
N/A
Condition:
Cerebellar abiotrophy
Contributor Comment:
Cerebellar
cortical abiotrophy is a spontaneous, premature and progressive degeneration
and death of neurons without an intrinsically identifiable defect; it is well
characterized in the dog and is described in several breeds. This condition is
characterized by ongoing Purkinje neuronal cell degeneration and loss with
reactive gliosis. Mostly, affected animals are healthy at the time of birth and
develop clinical signs at several months of age, which worsen with time. In
some breeds, a possible inherited genetic defect in the metabolism of the
neurotransmitter glutamic acid has been proposed (or established).
2,11
This case is an
unusual form of a cerebellar degeneration in the Coton de Tuléar breed,
characterized by a severe depletion in the granular cell layer, hence the name
cerebellar granuloprival degeneration for this condition.
9 Rare
cases of this condition in this breed have been published to date.
9
Similar to this case, all differ from the Purkinje cell atrophy reported in
many canine breeds.
Some similarity
between this Coton de Tuléar and cerebellar granuloprival hypo-plasia in cats
caused by intrauterine parvovirus infection has been proposed. However, in
Coton de Tuléars, there is no disorganization of the cerebellar cortex and no
lesions in the Purkinje cell layer. Parvovirus infection in dogs is not known
to induce cerebellar changes.
9,12
Contrary to what
has been published, in this case there is no significant inflammatory change in
the cerebellum.
9 The restriction of the disease specifically to
Coton de Tuléar breed is favors a genetic basis for the lesions, but this
hypothesis needs further analysis.
JPC Diagnosis:
Cerebellum:
Granular cell degeneration and loss, diffuse, severe, with spongiosis, and
minimal multifocal Purkinje cell loss, Coton de Tulear, canine.
Conference Comment:
The contributor provides a compelling example of an atypical form
of cerebellar abiotrophy in the canine. Cerebellar abiotrophy, also known as
cerebellar cortical degeneration,
has been
described as a hereditary defect in several breeds of dogs,1,3,9
Arabian horses,8 rabbits,7 an alpaca,6 and
recently in goats.5 Histologically, the characteristic distribution
of lesions includes the marked loss and degeneration of the Purkinje cell
neurons, often with retrograde degeneration in granular cells due to failure of
synaptogenesis between parallel nerve fibers of the granular cell layer and
Purkinje cells.1,3,5 In this Coton de Tuléar dog, there is diffuse and severe degeneration and loss of the
granular cell layer, with only scattered loss of Purkinje cells. This
histomorphology has been rarely reported in the veterinary literature as cerebellar
granuloprival de-generation in a number of different canine breeds, including
the Coton de Tuléar, as discussed by the
contributor.3,4,9 Neonatal cerebellar ataxia in Coton de Tuléar dogs
has also been reported as Banderas syndrome, suggesting a breed-related
hereditary disease.3
Abiotrophy is a spontaneous cerebellar degenerative disease
process characterized by premature loss of neurons in the cerebellum.2
Conference participants discussed how this differs from cerebellar hypoplasia,
a condition in which the cerebellum does not completely form during
embryogenesis due to in-utero viral infections from parvoviruses or
pestiviruses. Examples include feline parvovirus (pan-leukopenia), bovine
pestivirus (bovine viral diarrhea virus), classical swine fever (hog
cholera/pestivirus), sheep and goat pestivirus (Border disease), and rat
parvovirus (Kilham rat virus).2 Additionally, certain toxicities,
such as organophosphates, and malnutrition can also cause cerebellar
hypoplasia.3 In contrast to animals born with cerebellar hypoplasia,
those affected with abiotrophy are neurologically normal at birth and develop
early-onset progressive cerebellar proprioceptive deficits during the
post-natal period, in the case of this dog at four to five months.1,3,5
Typical neurologic deficits include ataxia, head tremor, intention tremors,
symmetrical hypermetria, broad-based stance, and loss of balance.2
In addition to the diffuse and severe degeneration and loss
of the cerebellar molecular cell layer, the conference moderator noted an
increase in the number of hypertrophic astrocytes with large
vesicular nuclei within the Purkinje cell layer, interpreted as Bergmann
gliosis. This astrocytic reaction occurs predominantly in areas where Purkinje
cells are lost, described by several conference participants as empty baskets.4
Bergmann glial cells are astrocytes with cell bodies located in the Purkinje
cell layer with long radial processes that surround the synapses on Purkinje
cell dendrites and extend to the molecular layer, terminating on the pial
surface of the cerebellum;10 they are essential for the normal
differentiation, migration and maturation of Purkinje cell and granular cell
neurons. The immunohistochemical stain, glial fibrillary acidic protein (GFAP),
is useful in demonstrating the empty baskets surrounded by Bergmann gliosis in
cases of cerebellar abiotrophy.10
The confounding aspect of this case is the severe selective
depletion of granular cells with only scattered loss of Purkinje cells. The
pathogenesis of cerebellar granuloprival degeneration in this breed has not yet
been elucidated; it is hypothesized to be the result of an inherited disorder
of granular cell development, but most Purkinje cells survive since their main
excitatory input is from the olivary nucleus.4 However, some authors
suggest that Purkinje cells can be lost as result of granular cell depletion in
chronically affected dogs.4
References:
1. Berry ML, Machado UB. Cerebellar abiotrophy in a miniature schnauzer. Can
Vet J. 2003; 44:657-659.
2. Cantile C, Youssef S. Nervous system. Maxie MG ed. In: Jubb
Kennedy and Palmer's Pathology of Domestic Animals.
Vol 1. 6th ed. Philadelphia, PA: Elsevier Saunders; 2016:275-276.
3. Coates JR, OBrien DP, Kline KL, et al. Neonatal cerebellar ataxia in
Coton de Tulear dogs. J Vet Intern Med. 2002; 680-689.
4. Huska J, Gaitero L, Heindrich SN, et al. Cerebellar granuloprival
degeneration in an Australian kelpie and a Labrador retriever dog. Can Vet J.
2013; 54:55-60.
5. Koehler JW, Newcomer BW, Holland M, Caldwell JM. A novel inherited
cerebellar abiotrophy in a cohort of goats. J Comp Path. 153:135-139.
6. Mouser P,
Levy M, Sojka JE, Ramos-Vara JA. Cerebellar abiotrophy in an alpaca (Lama
pacos). Vet Pathol. 2009; 46:1133-1137.
7. Sato J, Yamada N, Kobayashi R, et al. Morphometric analysis of
progressive changes in hereditary cerebellar cortical degenerative disease
(abiotrophy) in rabbits caused by abnormal synap-togenesis. J Toxicol Pathol.
2015; 28:73-78.
8. Scott EY, Penedo MC, Murray JD, Finno CJ. Defining trends in
global gene expression in Arabian horses with cerebellar abiotrophy. Cerebellum.
2016; Oct 5. [Epub ahead of print].
9. Tipold A, Fatzer R, Jaggy A, Moore P, Vanevelde M. Presumed
immune-mediated cerebellar granuloprival de-generation
in the Coton de Tuléar breed. J neuroimmunol. 2010; 110:130-133.
10. Yamada K, Watanabe
M. Cyto-differentiation of Bergmann glia and its relationship with
Purkinje cells. Anat Sci Int.
11. Zachary JF, McGavin DM. Nervous system. In: Pathologic Basis
of Veterinary Disease. 5th ed. St. Louis, MO: Mosby; 2012:816, 856-857.