Signalment:
18-month-old female American bison (
Bison bison)The animal was sick for 10 days, gradually lost condition, developed diarrhea, and exhibited swelling in both carpal joints. Lameness led to difficulty walking. The animal eventually became recumbent and was euthanized.
Gross Description:
Both lungs contained randomly distributed irregular, sometimes raised, variably sized foci of caseous necrosis. Both stifle joints were swollen, fluctuant on palpation, and, upon incision, oozed purulent to caseous exudate. There was marked inflammation of the joint capsules, synovial tissue, and tendon sheaths. There was a localized area of necrosis in the cervical musculature on the right side of the neck (injection site).
Histopathologic Description:
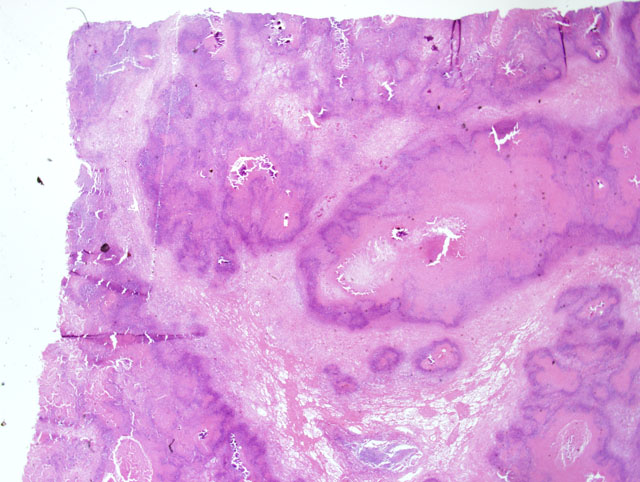
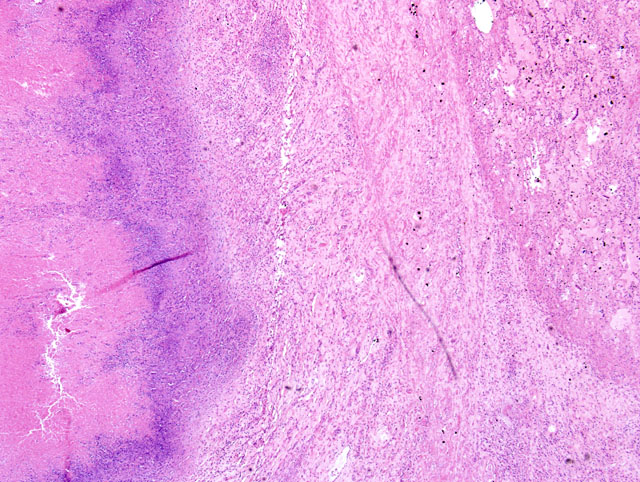
Large areas of caseous necrosis and inflammation were noted in the pulmonary parenchyma (
Fig. 4-1). Affected areas were characterized by a distinct outer zone of variably dense fibrous connective tissue infiltrated with a mixture of inflammatory cells, a thinner middle zone consisting of a bilayer of activated macrophages and plasma cells (outer portion) and necrotic neutrophils (inner portion), and an expansive interior zone of caseous necrosis in which necrotic alveolar septa and bronchioles were seen. Normal lung adjacent to the outer fibrous connective tissue capsule had several areas of atelectasis, some accumulation of proteinic material within alveoli, and increased numbers of alveolar macrophages. Sections of synovium, joint capsule and tendon sheath had a marked necropurulent synovitis, arthritis, and tenosynovitis respectively.
Morphologic Diagnosis:
Severe, multifocal to coalescing, caseonecrotic pneumonia with bronchiectasis and atelectasis.
Lab Results:
Mycoplasma spp. was cultured from lung tissue and joint exudate. This isolate was then identified as
Mycoplasma bovis by PCR and immunohistochemistry. No other ruminant respiratory pathogens were identified in these tissues.
Condition:
Mycoplasma bovis
Contributor Comment:
Mycoplasma bovis-associated disease manifests itself in a variety of ways in cattle including pneumonia and tenosynovitis, arthritis, keratoconjunctivitis, otitis media, and mastitis.(2) A condition caused by
M. bovis characterized by chronic pneumonia and polyarthritis has been recognized in feedlot cattle.(3) Coinfection with Bovine viral diarrhea virus (BVDV), and common bovine respiratory viruses appears to occur with some frequency. Previous studies utilizing IHC to examine the pattern of bacterial colonization in
M. bovis infected lungs described staining in bronchiolar epithelial cells, inflammatory cells, and abscessed airways (naturally infected animals), random staining in areas of both coagulative and caseous necrosis with peripheral zones of purulent to pyogranulomatous inflammation (naturally infected animals), and staining in areas of coagulative necrosis and bronchiolar epithelium (naturally infected animals) and in inflammatory cells in alveoli and septal walls (experimentally infected animals).(3) Staining patterns in naturally infected animals were consistent with the type of staining seen in this animal. The most intense staining was observed in bronchiolar epithelial cells. This pattern of staining supports the proposed pathogenesis for this condition, which is early bronchitis leading to bronchiectasis that eventually coalesces into large zones of caseous necrosis.(1) Characteristics of this case indicate that bison are susceptible to severe infections with this pathogen, and that
M. bovis is capable of causing primary disease in this species.
JPC Diagnosis:
Lung: Pneumonia, necrotizing, fibrinosuppurative, diffuse, severe with interlobular edema and fibrosis.
Conference Comment:
Mycoplasma bovis is of significant economic importance in both the United States and Europe. It is the cause of enzootic pneumonia in young calves and a cause of chronic pneumonia and polyarthritis in adult cattle. This disease in feedlot cattle mimics shipping fever, but infected cattle are often times refractory to antimicrobial treatment with continued clinical decline. M. bovis is spread via genital, nasal, and mammary secretions.(1)
M. bovis is very effective at evading the host immune response by a variety of mechanisms too in-depth to discuss here, and subsequently this organism causes chronic infections despite valiant clinical intervention.Â
Grossly,
M. bovis can cause striking lesions within the respiratory tract characterized by multiple, well demarcated, caseonecrotic nodules up to a few centimeters in diameter dispersed throughout the cranioventral lung lobes. Joint lesions are characterized by a reddened and reactive synovium and serofibrinous exudate within the joint capsule.(1) Otitis media is an additional sequelae in calves to
M. bovis infection.(1)
Histologically, acute lesions begin in the airways and progress to areas of multifocal to coalescing caseonecrotic debris often containing mineral. Early in the progression of
M. bovis infections, leukocytes are necrotic but retain their cellular architecture and contain hypereosinophilic cytoplasm and karyorrhectic nuclei. This is characteristic for respiratory lesions caused by
M. bovis.(1) Foci of necrosis with this distinctive gross and histologic appearance are pathognomonic for
M. bovis in cattle.(1)
References:
1. Caswell JL, Williams KJ: Respiratory system.Â
In: Jubb, Kennedy and Palmers Pathology of Domestic Animals, ed. Maxie MG, 5th ed., pp. 611-614. Elsevier Limited, Edinburgh, UK, 2007
2. Dyer N, Hansen-Lardy L, Krogh D, Schaan L, Schamber E: An outbreak of chronic pneumonia and
polyarthritis syndrome caused by
Mycoplasma bovis in feedlot bison (
Bison bison). J Vet Diagn Invest
20:369-371, 2008
3. Gagea MI, Bateman KG, Shanahan RA, van Dreumel T, McEwen BJ, Carman S, Archambault M,
Caswell JL: Naturally occurring
Mycoplasma bovis-associated pneumonia and polyarthritis in feedlot beef calves. J Vet Diagn Invest
18:29-40, 2006