Signalment:
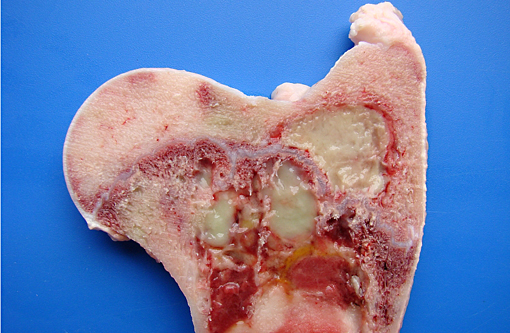
Gross Description:
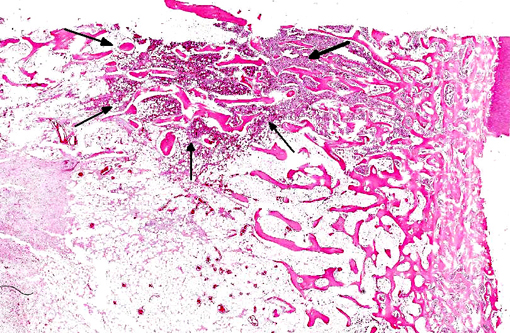
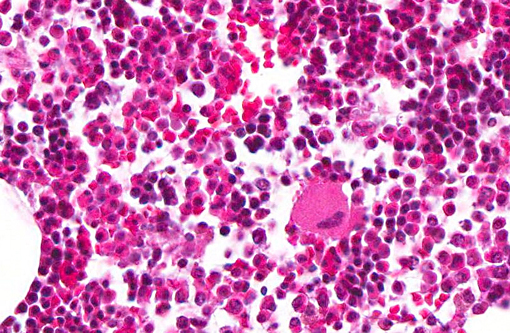
Histopathologic Description:
Morphologic Diagnosis:
Lab Results:
Condition:
Contributor Comment:
In humans, granulocytic sarcomas precede or occur concomitantly with acute myeloid leukemia, chronic myeloproliferative disorders, or myelodysplastic syndromes but are also described without association with any other hematologic disturbance. In the veterinary literature, granulocytic sarcomas are mentioned affecting dogs, cats, cattle,(12) a rabbit,(9) and a pig.(5) In dogs and cats, the more consistently involved organs are the lungs, intestine, skin, lymph nodes and liver. In one of the few reports found in the veterinary literature, this tumor is mentioned as a mass in the neck of a Bull Terrier dog. In cattle, skeletal muscle is characteristically affected. In the case reported in a rabbit, the granulocytic sarcoma involved the skin, subcutaneous tissue, and skeletal muscle of the perineum.(9) In the reported case in the pig, the tumor involved liver, kidneys, and mesenteric lymph nodes.(5) Differently from what occurs in human patients, animals affected by granulocytic sarcomas are almost always aleukemic; however, progression to a leukemia may occur. The current report describes gross findings, cytology, histopathology, histochemistry, and immunohistochemistry of a multicentric eosinophilic granulocytic sarcoma affecting a pig.Â
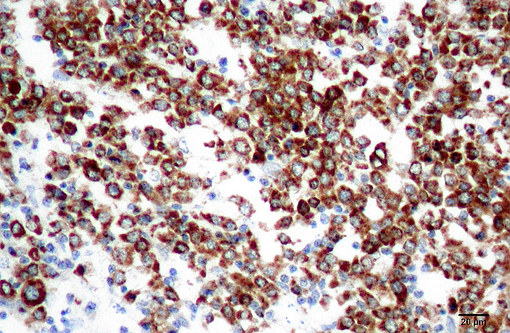
Granulocytic sarcoma was suspected in the current case based on the presence of light green masses in the gross inspection of the carcass and viscera, a typical aspect of this tumor. The anatomical distribution of the lesions observed in the current case is quite similar to that described for human granulocytic sarcomas, in which the occurrence is primarily subperiosteal and involves mainly the ribs, sternum, and pelvis. The microscopic presentation pattern observed both at cytological and histological examination consisting predominantly of precursor cells with myelocyte differentiation, allowed the presumptive diagnosis of well-differentiated granulocytic sarcoma. Immunohistochemistry results established the cell origin as of the granulocytic lineage, and histochemistry determined that the cells were possibly of eosinophil lineage, definitively confirming the diagnosis suspected at gross examination.Â
The definitive diagnosis of myeloid sarcomas is based on the association of phenotypic (cytology, histology, cytochemistry, and histochemistry) and immunophenotypic (immunocytochemistry and immunohistochemistry) aspects. An immune phenotype of neoplastic cells positive for myeloperoxidase is the hallmark for granulocytic sarcoma. Other antibody markers have reportedly yielded positive results when applied to human cases of granulocytic sarcoma, including CD13, CD33, CD117, CDl5, CD68, CD43,(7) and lysozyme.(8) However, lysozyme and CD68 are also marked in cases of monoblastic sarcoma, a less common form of myeloid sarcoma.
In the porcine tumor described here, the negative staining for CD117 could be explained by both the predominance of myelocytes and absence of myeloblasts, which are precursor cells that express this antigen. The negative staining for lysozyme was somewhat expected because swine granulocytes have been described as negative for this marker,(3) whereas porcine monocytes and/or macrophages are strongly positive,(4) similar to what is described in humans, a species in which lysozyme is the choice marker for monocytes and/or macrophages. Based on this species-specific feature, the negative reaction to lysozyme further helps to differentiate granulocytic sarcoma from monoblastic sarcoma.
In human patients, several different forms of lymphoma presentation are often confused with myeloid sarcoma(7) and thus should be the main tumors to be included in the differential diagnosis. In the current case, the differentiation was made based on the following aspects: histological evidence of cytoplasmic eosinophilia, fine granularity observed in the cytoplasm of neoplastic cells when examined in cytological preparations, occurrence within the neoplasm of more mature eosinophil precursors amidst myeloblasts, and the immunohistochemistry results. Furthermore, the observation of light green masses that partially obliterated the bone marrow of several flat and long bones prompted the suspicion of granulocytic sarcoma, as this is the only hematopoietic neoplasm that expresses this typical color grossly. The green discoloration observed in fresh tissue specimens is due to the presence of myeloperoxidase and substantially helps in the diagnosis of granulocytic sarcoma.
Few lesions could grossly resemble granulocytic sarcoma. Such lesions include chlorellosis, a granulomatous algal infection observed in human beings and animals,(6) and eosinophilic myositis, which is frequently associated with Sarcocystis spp. infection in cattle(11) and has been described in a pig. The association of gross examination, cytology, histology, histochemistry, and immunohistochemistry findings is consistent with a diagnosis of eosinophilic granulocytic sarcoma.Â
JPC Diagnosis:
Conference Comment:
References:
2. Brum JS, Lucena RB, Martins TB, Fighera RA, Barros CSL. Eosinophilic granulocytic sarcoma in a pig. J Vet Diagn Invest. 2012;24:807-811.
3. Chianini F, Maj³ N, Segal+�-�s J, Domingu+�-�z J, Domingo M. Immunohistological study of the immune system cell in paraffin-embedded tissues of conventional pigs. Vet Immunol Immunopathol. 2001;82:245-255.
4. Evensen O. An immunohistochemical study on the cytogenetic origin pulmonary multinucleate giant cells in porcine dermatosis vegetans. Vet Pathol , 1993;30:162-170.Â
5. Fisher LF, Olander HL. Spontaneous neoplasms of pigs-a study of 31 cases. J Comp Pathol. 1978;88:505-517.Â
6. Haenichen T, Facher E, Wanner G, Hermanns W. Cutaneous ch1orellosis in a gazelle (Gazella dorcas). Vet Pathol. 2002;39:386-389.Â
7. Menasce LP, Banerjee SS, Beckett E, Harris M. Extramedullary myeloid tumour (granulocytic sarcoma) is often misdiagnosed: a study of 26 cases. Histopathology. 1999;34:391-398.Â
8. Neiman RS, Barcos M, Berard C, Bonner H. Mann R, Rydell RE, et al. Granulocytic sarcoma: a clinicopathologic study of 61 biopsied cases. Cancer. 1981;48:1426-1437.Â
9. Perkins SE, Murphy JC, Alroy J. Eosinophil granulocytic sarcoma in a New Zealand white rabbit. Vet Pathol. 1996;33:89-91.Â
10. Valli VE. The hematopoietic system. In: Maxie MG. ed. Jubb, Kennedy, and Palmers Pathology of Domestic Animals. 5th ed. Vol. 3. Philadelphia, PA: Elsevier; 2007:107-324.Â
11. Vangeel L, Houf K, Geldhof P, Nollet H, Vercruysse J, Ducattle R, et al. lntramuscular inoculation of cattle with Sarcocystis antigen results in focal eosinophilic myositis. Vet Parasitol. 2012;183:224-230.Â
12. Webb JL, Latimer KS. Leukocytes. In: Latimer KS, ed. Duncan and Prasses Veterinary Laboratory Medicine Clinical Pathology. 5th ed. Ames, IA: John Wiley and Sons. 2011:45-58.