Signalment:
One approximately 3-month-old female Coopers hawk (
Accipiter cooperi)This animal was admitted to the Raptor Center of the University of Minnesota with neurologic signs in
late August. It was emaciated and had evidence of head trauma. Funduscopic investigation revealed an exudative chorioretinitis. Due to the deterioration of the clinical state of the animal, and based on the funduscopic finding indicating WN disease, the animal was euthanatized two days after admission.
Gross Description:
The vitreous of the left eye was slightly opaque after fixation. In addition, the bird had acute
bilateral leptomeningeal hemorrhages at the base of both cerebral lobes. The spleen was moderately enlarged, soft and diffusely dark red.
Histopathologic Description:
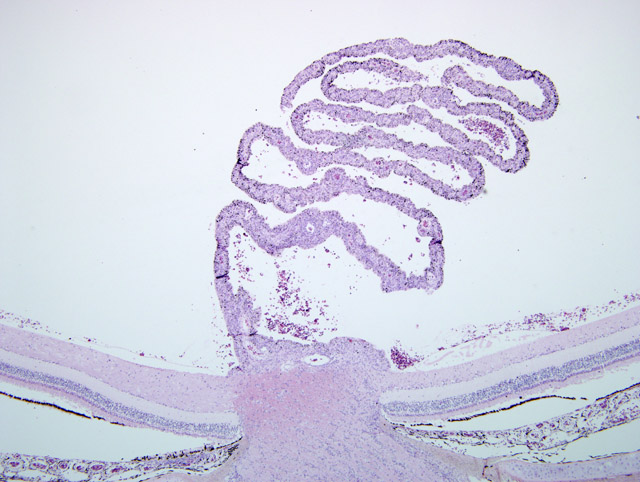
Sections of two blocks representing both eyes were submitted (slide A = left eye;
slide F = right eye): Right eye (slide F): Numerous capillaries of the pecten were internally lined or surrounded by
predominantly plasma cells and lymphocytes and fewer macrophages. The capillaries had hyperplastic endothelial
cells. Clusters of inflammatory cells were also present between lamellae of the pecten. A similar infiltrate was
present in the optic disc. The retina and choroid on one side of the pecten appeared to be largely unaffected except
for the deposition of a small amount of faint basophilic stringy material on the nerve fiber layer that entrapped a
small number of degenerate inflammatory cells and a minimal infiltration of the nerve fiber layer by plasma cells.
The choroid and retina on the other side of the pecten was significantly altered. The choroid of this segment was
markedly infiltrated by plasma cells and lymphocytes. The retinal pigmented epithelial (RPE) cells overlying
inflamed choroidea were degenerate with loss of their delicate processes that support the photoreceptor cells. More
severely affected RPE cells were detached and individualized and assumed a plump shape. The retina was detached
in these areas. Occasional small aggregates of inflammatory cells (lymphocytes and plasma cells) were present
within the nerve fiber layer of this retinal segment particularly close to the pecten. Swollen axons were occasionally
present within the nerve fiber layer. The iris was infiltrated by few individual plasma cells and lymphocytes. The
lens was absent in some of the sections either due to a processing artifact or due to an oblique angle of the initial
sectioning of the eye globe. Some sections contained adipose tissue within the area of the vitreous due to a
processing artifact.
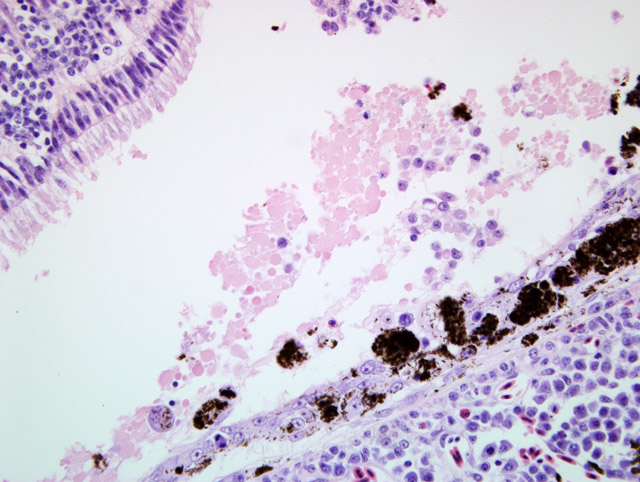
Left eye (slide A): The lesions were similar to the lesions described in the right eye. The degenerate changes of the
retinal pigmented epithelial cells were more widespread so that non-degenerate RPE cells were essentially absent.
In addition, there was multifocal acute hemorrhage into the choroid, retina and vitreous. A group of foamy
macrophages (gitter cell morphology) were present at the margin of disrupted retina in one location.
Morphologic Diagnosis:
Pectenitis and choroiditis, lymphoplasmacytic, chronic, marked with
retinal degeneration and detachment
Acute intravitreal hemorrhage (only left eye, slide A)
Lab Results:
A tissue pool containing kidney, heart and brain was positive for West Nile virus (WNV) by
PCR but virus isolation using Vero cells failed to detect WNV (Animal Health Diagnostic Center of Cornell
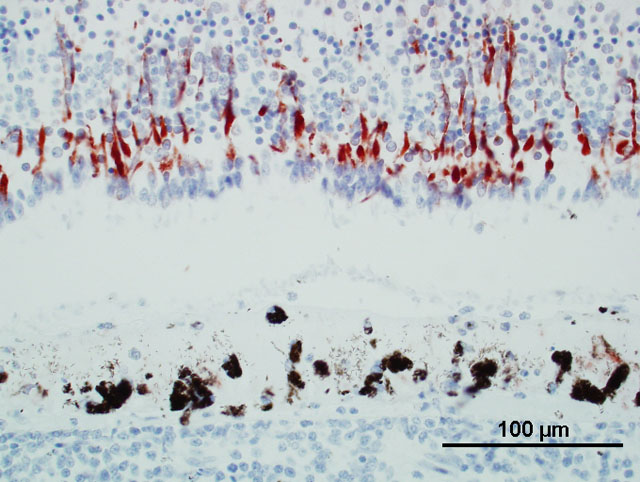
University). WNV was not detected in the aqueous humor of the right eye. WNV antigen was present in the retina
of the left eye (slide A), mainly in the outer nuclear layer (Fig. 1; monoclonal antibody against envelope protein
epitope of WNV; clone 7H2; BioReliance, Rockville, Maryland, USA). The animal had a high WNV specific
antibody titer (> 1:320) while antibodies against Saint Louis encephalitis virus were undetectable (< 1:10).
Condition:
West Nile virus
Contributor Comment:
West Nile virus (WNV) infection is common in Coopers hawks (and red tailed
hawks).(7) The disease may be transmitted to Coopers hawks by mosquito bites but oral infection is also plausible
since Coopers hawks tend to prey on songbirds some of which may constitute an avian reservoir for WNV. In a
subgroup of the infected Coopers hawks, e.g., many hatch year birds, the infection results in WN disease while the
majority of infected Coopers hawks (and red tailed hawks) likely eliminate the virus rapidly without having clinical
signs, similar to the situation in humans. This is evidenced by a fairly high WNV seroprevalence in apparently
healthy Coopers hawk and red tailed hawk populations.(1,6) WN disease in Coopers hawks (and red tailed hawks)
is frequently characterized by neurologic signs (e.g., depression and tremors) and visual impairment ultimately
potentially progressing to blindness.(5) Ophthalmologic examination of hawks with WN disease frequently
demonstrates opacities in the fundus of one or both eyes that is interpreted as chorioretinitis by clinicians.(5) The
presence of funduscopic lesions in hawks may aid in establishing a diagnosis of WN disease in the living patient.
Gross lesions other than emaciation are uncommon in hawks with WN disease, but may include opacities of the
fundus (chorioretinitis) of one or both eyes, myocarditis and unilateral or bilateral cerebral malacia.
Histologically, pectenitis is one of the most consistent findings in Coopers hawks with WN disease.(5,7) As in the
present case, chorioditis is frequently also present and occasionally, retinal degeneration and necrosis are additional
ocular lesions. Retinal pigmented epithelial cells overlying areas of chorioditis are usually degenerate as evidenced
by individualization and clumping of these cells possibly leading to detachment of the retina in the respective
segment. The funduscopically or grossly visible opacities in the fundus likely represent fibrin deposition on the
nerve fiber layer of the retina. Lymphoplasmacytic iridocyclitis may in some cases be a minor component of the
ocular lesions.
WNV may be detectable by PCR, immunohistochemistry or virus isolation in the eyes (e.g., aqueous humor) even
when other organs are negative.(4,5,7) The retina may harbor only a few individual WNV antigen positive cells
(e.g., ganglion cells), or in some cases entire full thickness segments of the retina overlying areas of chorioditis may
be strongly WNV antigen positive. In addition, WNV-specific antibodies are usually present in the aqueous humor
of infected Coopers hawks (and red tailed hawks) with WN disease although plasma/serum titers are usually
higher.(5) Hence aqueous humor may be used as substrate for detection of antibodies when plasma or serum is not
available (e.g. in carcasses submitted to veterinary diagnostic laboratories).
Besides the inflammatory and degenerative lesions that were present in both eyes, one eye had evidence of trauma in
the form of intravitreal hemorrhage in the present case. Trauma likely was also the cause of the macroscopically
observed leptomeningeal hemorrhage.
JPC Diagnosis:
Eye: Pectenitis, lymphoplasmacytic, diffuse, moderate with multifocal, mild choroiditis and
retinal degeneration and detachment
Conference Comment:
West Nile virus (WNV) is a single-stranded, icosohedral, enveloped RNA virus in the
family
Flaviridae, genus
Flavirus. WNV is mainly transmitted by mosquitoes and circulates in the environment
often through a sylvatic cycle from bird to mosquito to bird. West Nile derives its name from the West Nile district
of Uganda, where the virus was first discovered. WNV was first recognized in the United States in 1999 in New
York after an outbreak in the local bird population.(2)
West Nile virus has a very wide host range with the major natural amplifier being crows and corvids. Birds develop
a severe viremia with virus detectable in many organs in the body. This is in contrast to horses, where the virus is
only found in the CNS.(3) Horses and some birds are exquisitely sensitive to WNV, and disease outbreaks are
usually seen during peak mosquito season, which is the summer and fall. Mortality rate in horses can reach 50%.
Gross lesions are usually absent or unspectacular, but spinal cord lesions do occur and manifest as hemorrhage and
malacia of the thoracic and lumbar spinal cord. Histologically, lesions are usually present in the brainstem and
thoracolumbar spinal cord and consist of a nonsuppurative encephalomyelitis, gliosis, and glial nodule formation
with or without necrosis. The gray matter usually contains more severe lesions. Birds often have lesions in the
heart and liver. WNV infection is much more of a systemic disease in affected birds. Cats, dogs, cattle, and swine
are susceptible to WNV, but they often have subclinical disease.(3)
References:
1. Hull J, Hull A, Reisen W, Fang Y, Ernst H: Variation of West Nile virus antibody prevalence in migrating and
wintering hawks in central California. The Condor
108:435-439, 2006
2. Lichtensteiger CA, Greene CE: Arthropod-Borne viral infections.Â
In: Infectious Disease of the Dog and Cat, ed.
Greene, 3rd ed., pp. 192-195. Saunders, Elsevier, St. Louis, MO, 2006
3. Maxie MG, Youssef S: Nervous system.Â
In: Jubb, Kennedy and Palmers Pathology of Domestic Animals, ed.
Maxie MG, 5th ed., vol 1, pp.421-422. Elsevier Limited, Philadelphia, PA, 2007
4. Nemeth N, Gould D, Bowen R, Komar N: Natural and experimental West Nile virus infection in five raptor
species. Jour of Wil Dis
42:1-13, 2006
5. Pauli AM, Cruz-Martinez LA, Ponder J, Redig PT, Glaser A, Klauss G, Schoster JV, W+�-+nschmann A:
Ophthalmologic and oculopathologic findings in red-tailed hawks (
Buteo jamaicensis) and Coopers hawks
(
Accipiter cooperi) with naturally acquired West Nile virus infection. J Am Vet Med Assoc 231:1240-1249, 2007
6. Stout WE, Cassini AG, Meece JK, Papp JN, Rosenfield RN, Reed KD: Serologic evidence of West Nile virus
infection in three wild raptor populations. Avian Dis
49:371-375, 2005
7. W+�-+nschmann A, Shivers J, Bender J, Carroll L, Fuller S, Saggese M, van Wettere A, Redig P: Pathologic findings
in red tailed hawks (
Buteo jamaicensis) and Coopers hawks (
Accipiter cooperi) naturally infected with West Nile
virus. Avian Dis
48:570-580, 2004