Signalment:
Gross Description:
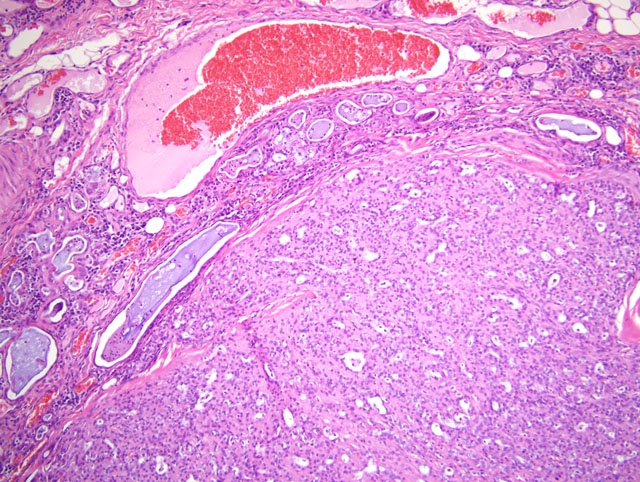
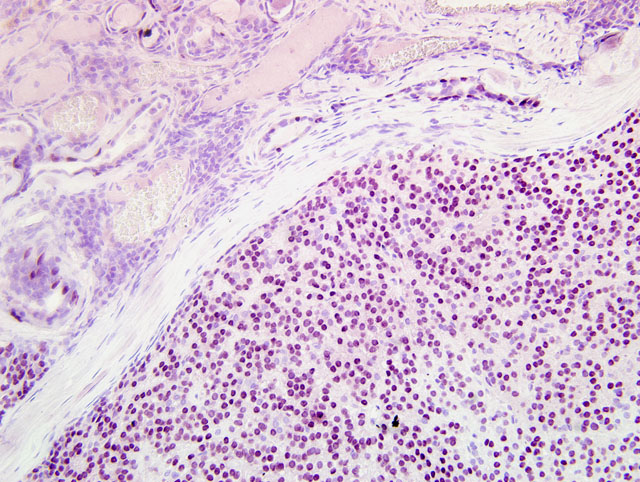
Histopathologic Description:
The remaining portions of the gland of the third eyelid located adjacent to the mass are compressed and markedly infiltrated by varying numbers and combinations of lymphocytes, plasma cells, histiocytes, hemosiderin laden macrophages, and neutrophils. Lymphocytes form dense sheets and few lymphofollicular aggregates that separate glandular structures. Glandular structures are often tortuous and are occasionally hyperplastic, with hypertrophied plump cuboidal epithelium lining. Glandular structures are variably distended by variable amounts of eosinophilic to amphophilic proteinaceous fluid and occasional neutrophils. Severely distended glandular structures are lined by markedly attenuated epithelium. Occasionally, glandular tubules are surrounded by thin rings of fibrosis. Blood vessels within the gland and the surrounding stroma are prominently congested.
The conjunctival epithelium overlying the mass is variably eroded and ulcerated. There is prominent exocytosis of neutrophils through areas of intact epithelium. In areas of severe erosion, the epithelium is reduced to a thin layer of attenuated cells. In areas of ulceration, there is mild hemorrhage and prominent exudation of fibrin and neutrophils into the conjunctival sac. There is segmental squamous metaplasia of the mucosal epithelium lining the remainder of the conjunctiva, with focal erosion and ulceration. The normal pseudostratified columnar conjunctival epithelium in this area is variably replaced by thick nonkeratinizing squamous epithelium. Goblet cells are rare. In many areas, the superficial surface of the squamous epithelium is eroded and tattered. There is prominent exocytosis of neutrophils through the epithelium, with neutrophils rarely forming small aggregates in the superficial epithelial layers. A variably thick band of primarily lymphocytes and plasma cells, and fewer neutrophils and histiocytes parallels the junction of the mucosal epithelium and the underlying substantia propria of the third eyelid, the palpebrae, and the globe. Blood vessels in the superficial substantia propria of the entire conjunctiva are prominently congested.
The corneal epithelium of the globe is diffusely hyperplastic. The basal border of the epithelium is irregularly undulated with the epithelium rarely forming short thick rete pegs that extend into the underlying stroma. Rare basal epithelial cells contain finely granular, intracytoplasmic, dark brown pigment consistent with melanin. Exocytosis of neutrophils and, to a lesser degree, lymphocytes through the corneal epithelium is focally prominent. The corneal stroma contains small blood vessels lined by plump endothelial cells that are often surrounded by infiltrates of lymphocytes and neutrophils. Ingrowth of blood vessels is most prominent in the superficial corneal stroma, but is also present in the mid and deep stroma. The normal regularly arranged, pale staining corneal stroma is superficially replaced by dense, more haphazardly arranged collagenous stroma. Blood vessels at the limbus are surrounded by aggregates of lymphocytes and plasma cells.
The anterior chamber of the globe is moderately collapsed and contains scant amounts of fibrin and proteinaceous flocculent material. There is widespread synechiation between the axial posterior cornea, the iris, the ciliary body and the lens. In this region, there is focally extensive loss of the corneal endothelium, and the pupillary margins of the iris and the anterior capsule of the lens are adhered to Descemets membrane by a thick band of fibrosis containing occasional melanin-containing round to spindle cells. This band of fibrosis is continuous with a thin periridal fibrovascular membrane composed of fibroblasts, small blood vessels, and scant collagenous stroma. In some sections, a dense retrocorneal band of fibrosis extends from areas of synechiation over the inferior portions of Descemets membrane. The pupillary margins of the iris are reflected into the posterior chamber of the anterior compartment by bands of fibrovascular stroma that extend onto the posterior aspect of the iris creating entropion uveae. The pigmented epithelium that normally lines the posterior iris is widely lost and there are numerous loosely arranged fibrous adhesions between the posterior iris, the pars plicata of the ciliary body, and the lens capsule. The superior aspect of the iris is bowed into the anterior chamber (iris bomb+�-�) and the associated ciliary cleft is collapsed. The anterior uvea (iris and ciliary body) is moderately infiltrated by lymphocytes, plasma cells and fewer histiocytes that aggregate around blood vessels. The stroma of the pars plicata of the ciliary body is expanded by edematous, myxomatous matrix containing melanin-laden round cells. A band of dense fibrovascular stroma extending from the ciliary body lines the posterior lens capsule. Within this cyclitic membrane, there are few variably sized, spherical, dull gray bodies consistent with asteroid hyalosis, and many melanin-laden cells.
There is severe cataractous change of the lens. Lens epithelial cells are widely lost. There is marked liquefaction of cortical lens material. Amorphous, eosinophilic pools of liquefied lens material contain numerous granular foci of dense mineralization. There are occasional granular to globular foci of mineralization distributed throughout the remaining lens fibers. The posterior lens capsule is focally wrinkled. There is marked plasmoid degeneration of the vitreous characterized by replacement of normal vitreous with pale eosinophilic flocculent material admixed with dependent aggregates of neutrophils, scant hemorrhage, and occasional pigment-laden cells.
The tapetal (superior) retina is widely detached. Peripheral portions of the tapetal retina are markedly atrophied showing marked loss of neurons throughout all layers, vesiculation of plexiform layers, blending of the inner and outer nuclear layers, and loss of photoreceptors. More centrally within this portion of retina, ganglion cells and inner nuclear layer neurons remain intact, but many neurons in the outer nuclear layer and nearly all photoreceptors are lost. There are rare foci of interretinal hemorrhage. The subretinal space contains mild accumulation of flocculent basophilic material and scant hemorrhage. Retinal pigmented epithelial cells in the superior fundus are markedly hypertrophied, creating rounding and tomb-stoning of the apical border of these cells. The nontapetal (inferior) retina is nondetached, but is severely atrophied. Atrophy within this portion of the retina is often diffuse, but is generally most severe within the inner most retinal layers. In the most severely affected areas, the retina is reduced to a thin layer of glial cells that contains occasional round to spindle shaped pigmented cells. In segmental areas of the nontapetal fundus, there is loss of retinal pigmented epithelial cells with adherence of the atrophied retina to Bruchs membrane. In one area, the nontapetal retina is torn and the anterior portion of the torn retina is detached (not present in all sections). The free end of the torn retina is rounded while the end that remains adherent to the RPE is attenuated. There is mild accumulation of hemorrhage in the subretinal space. Vessels in the choroid and retina are lined by plump endothelial cells and often contain prominent numbers of neutrophils. The neuropil of the optic nerve is mildly vesiculated.
Morphologic Diagnosis:
1. Third eyelid:
- Complex adenocarcinoma of the gland of the third eyelid
- Chronic lymphoplasmacytic adenitis of the gland of the third eyelid
3. Eye:
- Severe chronic superficial keratitis with corneal epidermalization
- Retrocorneal fibrous membrane
- Preiridal fibrovascular membrane
- Anterior and posterior synechiae, lenticular-corneal synechia, and iridocyclitic synechia
- Retrolenticular cyclitic membrane
- Entropion uveae and mild iris bomb+�-�
- Moderate chronic lymphoplasmacytic anterior uveitis
- Mature cataract (loss of lens epithelium, lens fiber liquefaction, and mineralization)
- Plasmoid degeneration of the vitreous and asteroid hyalosis
- Chronic diffuse tapetal retinal detachment with moderate outer retinal atrophy
- Severe diffuse nontapetal inner retinal atrophy with focal retinal tear and detachment
Condition:
Contributor Comment:
A diagnosis of adenocarcinoma rather than adenoma was based on the moderate cellular atypia of neoplastic epithelial cells and the focal infiltration of neoplastic cells into the partial capsule. Carcinomas of the gland of the third eyelid may be locally invasive, but metastasis is generally considered rare. In a report by Wilcock and Peiffer, local recurrence occurred in three of four dogs in which a carcinoma of the gland of the third eyelid was excised.(4)
In our case, the neoplastic mass was diagnosed as a complex adenocarcinoma based on the prominence of myoepithelial cells within the mass. On immunohistochemistry, the prominent spindle cell populations within some areas of the mass were immunoreactive for muscle specific actin and a pancytokeratin marker (MNF116), but were not labeled for vimentin. This pattern of immunoreactivity supports the designation of these cells as myoepithelial cells. To our knowledge, complex adenocarcinoma of the gland of the third eyelid has not previously been reported. In the report by Wilcock and Peiffer, spindle-cell and squamous metaplasia was described in a proportion of the tumors.(3) It is possible that the spindle-cell metaplasia described by Wilcock and Peiffer actually represented myoepithelium, but was not addressed as such in their report.
There was marked inflammation within the remaining portion of the gland of the third eyelid and throughout the conjunctiva. Such inflammation may be secondary to the mass itself and local irritation, but a secondary bacterial infection cannot be ruled out. The changes within the globe are numerous, and all are likely secondary to the presence of the mass in the third eyelid and associated trauma.
Corneal epidermalization (cutaneous metaplasia) is an adaptive change to chronic superficial irritation/trauma. Such change in this case was characterized by epithelial hyperplasia with mild rete peg formation, rare epithelial pigmentation, and superficial stromal fibrosis and vascularization. Keratinization of the corneal epithelium is another feature that is commonly seen with corneal epidermalization, but it was not prominent in this case.
Chronic uveitis likely led to the formation of a preiridal fibrovascular membrane, a retrocorneal fibrous membrane, a retrocorneal cyclitic membrane, and multiple synechiae between the cornea, iris, lens and ciliary body. The widespread synechiae between the iris, lens, and cornea resulted in obstruction of the pupil and glaucoma. Changes consistent with glaucoma are most prominent in the nontapetal retina. There is some degree of sparing of the detached tapetal retina in terms of atrophy in comparison to other segments of the retina. Such differences between the degree of atrophy between the tapetal and nontapetal retina and between detached and nondetached portions of the retina are common. The pathophysiology explaining the differences in susceptibility between various segments of the retina is unclear.
The cataractous changes within the lens may have occurred secondary to uveitis and the proliferation of fibrovascular stroma surrounding the lens or may be a degenerative age related change as cataracts were reported in both eyes of this dog. Clear evidence of the reported prolapse of the eye and rupture was not observed in the examined sections.
JPC Diagnosis:
1. Eye, lacrimal gland: Epithelial-myoepithelial carcinoma of lacrimal gland (low-grade complex adenocarcinoma of the lacrimal gland).
2. Eye, conjunctiva, third eyelid and palpebrae: Conjunctivitis, lymphoplasmacytic and neutrophilic, chronic, diffuse, severe, with squamous metaplasia.
3. Eye, cornea: Keratitis, superficial, diffuse, chronic, severe, with epidermalization.
4. Eye, uvea: Anterior uveitis, lymphoplasmacytic, moderate, diffuse, with anterior and posterior syncehiae, lenticular-corneal synechia, iridocyclitic synechia, and preiridal fibrovascular membrane formation.
5. Eye, lens: Lens fiber degeneration and liquefaction, diffuse, severe, with mineralization (mature cataract) and retrolenticular cyclitic membrane formation.
6. Eye, retina: Detachment, focally extensive, with marked atrophy.
Conference Comment:
Histologically, the neoplasm resembles epithelial-myoepithelial carcinoma of salivary gland, i.e., wellcircumscribed, partially encapsulated tumor of both luminal epithelium and myoepithelium, in which the myoepithelium predominantes and myoepithelial cells are multifocally polygonal with clear cytoplasm. Using the unstained serial histologic sections submitted with this case, immunohistochemistry was performed for cytokeratin AE1/AE3, vimentin, smooth muscle actin, and the myoepithelial markers calponin and p63. Most neoplastic cells display strong cytoplasmic immunoreactivity for cytokeratin, and many particularly the spindled neoplastic cells are strongly immunoreactive for smooth muscle actin. Rare vimentin-positive cells are scattered throughout the tumor; these are interpreted as non-neoplastic supporting stromal cells. While neoplastic cells are negative for calponin by immunohistochemistry performed at the AFIP, over 80% of the neoplastic cells exhibit positive, specific nuclear immunohistochemical staining for p63, which is a sensitive and specific marker for myoepithelium in canine tissue.(2)
The immunohistochemistry results generally agree with those reported by the contributor, and support the presence of myoepithelial proliferation; and although not officially recognized in the current World Health Organization classification scheme, participants agreed with the contributors diagnosis of complex adenocarcinoma. This case was also studied in consultation with the AFIP Department of Ophthalmic Pathology, which concurred with the diagnosis. Dubielzig and coworkers at the Comparative Ocular Pathology Laboratory of Wisconsin reportedly have collected 109 cases of epithelial tumors of the gland of the third eyelid in dogs, which includes adenocarcinoma, adenoma, and complex and mixed tumors that collectively represent 1.9% of canine tumor submissions to that laboratory.(1)
Conference participants briefly reviewed neoplasia in the nictitating membrane among animals, a rarity in the dog, a species more prone to the development of protrusion of the gland of the third eyelid (cherry eye). Squamous cell carcinoma is the most common neoplasm in domestic animal species, particularly in cattle and horses; its prevalence is correlated with high altitude and sunlight exposure. Conjunctival squamous cell carcinoma (SCC) develops via progression from precancerous changes to malignancy, akin to those described for cutaneous SCC: squamous plaque, squamous papilloma, squamous cell carcinoma in situ, and invasive SCC. Less common tumors of the nictitans include vascular tumors (e.g. hemangioma, hemangiosarcoma, and angiokeratoma), lymphoma, melanoma, and mast cell tumor.(3)
References:
2. Gama A, Alves A, Gartner F, Schmitt F: p63: a novel myoepithelial cell marker in canine mammary tissues. Vet Pathol 40:412-420, 2003
3. Wilcock B, Dubielzig RR, Render JA: Histological Classification of Ocular and Otic Tumors of Domestic Animals, 2nd series, vol. IX, ed. Schulman FY, pp. 18-22. Armed Forces Institute of Pathology (in cooperation with the American Registry of Pathology and the World Health Organization Collaborating Center for Worldwide Reference on Comparative Oncology), Washington, DC, 2002
4. Wilcock B, Peiffer R Jr. Adenocarcinoma of the third eyelid in seven dogs. J Am Vet Med Assoc 193:1549-1550, 1988