Joint Pathology Center
Veterinary Pathology Services
Wednesday Slide Conference
2018-2019
Conference 23
24 April, 2019
CASE III: 66619 (JPC 4066455)
Signalment: 12 year old, Male Neutered, Italian Greyhound, Canis lupus familiaris, Canine
History: One-week history of anxiety, nervousness, intermittent anorexia, and excessive vocalization. The vocalization was especially pronounced with regards to interactions involving the head or neck. The patient was referred to a neurologist for examination and further workup. On the morning of neurological consultation, an episode of ataxia, with a partial seizure occurred, followed by several hours of mental disorientation, the inability to sit up, and repetitive paddling activities impacting the entire left side of the body. When presented to the neurologist, the dog exhibited postural deficits on the right side. Magnetic resonance imaging (MRI) showed a multifocal disease process impacting the falx cerebri rostral to the lateral ventricles and the ventrolateral right cerebrum adjacent to the meningeal surface. The ventrolateral lesion was approximately 1 cm in diameter with the falx cerebral lesions being smaller in size. Lesions coalesced at the cribiform plate.
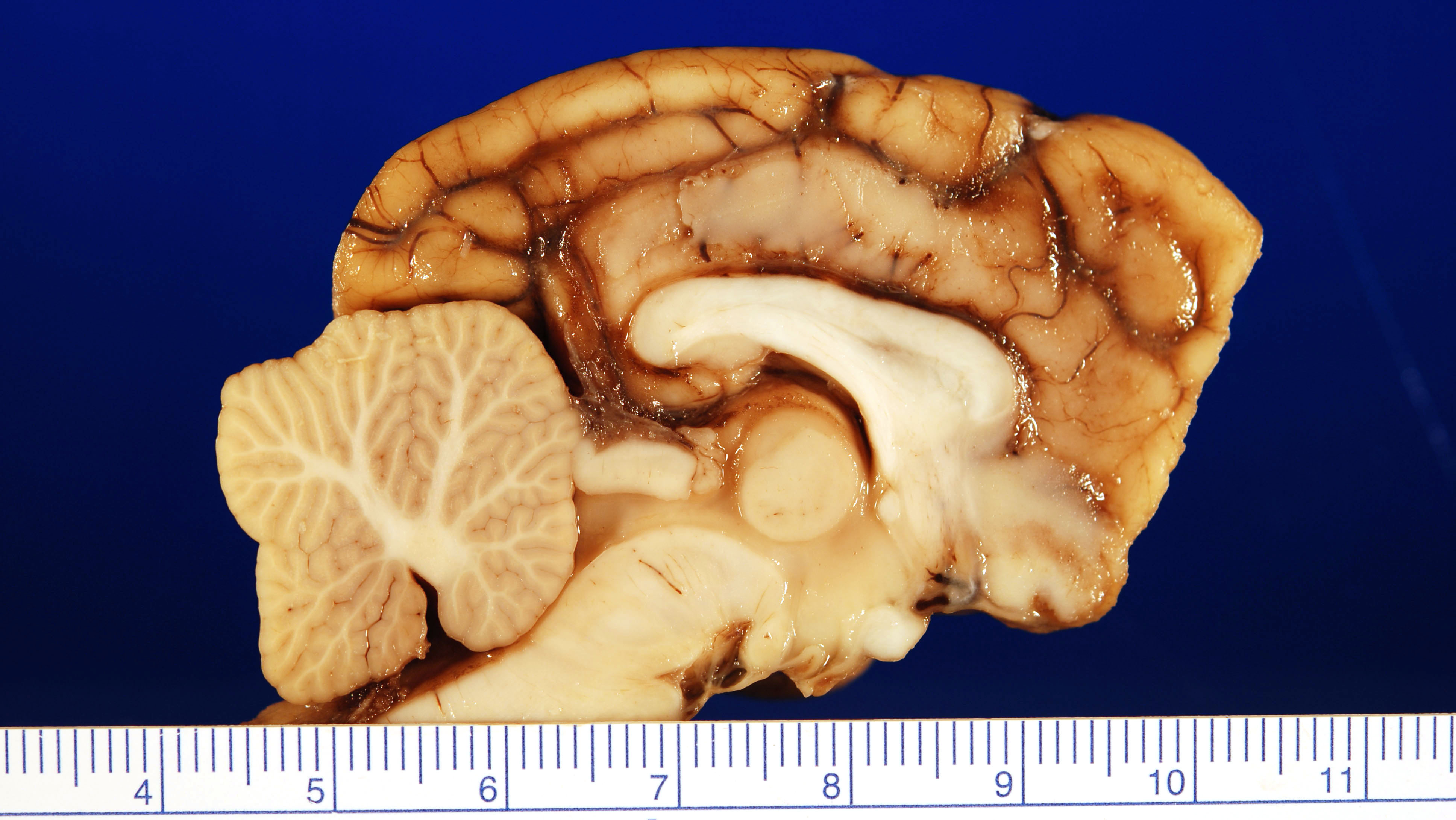
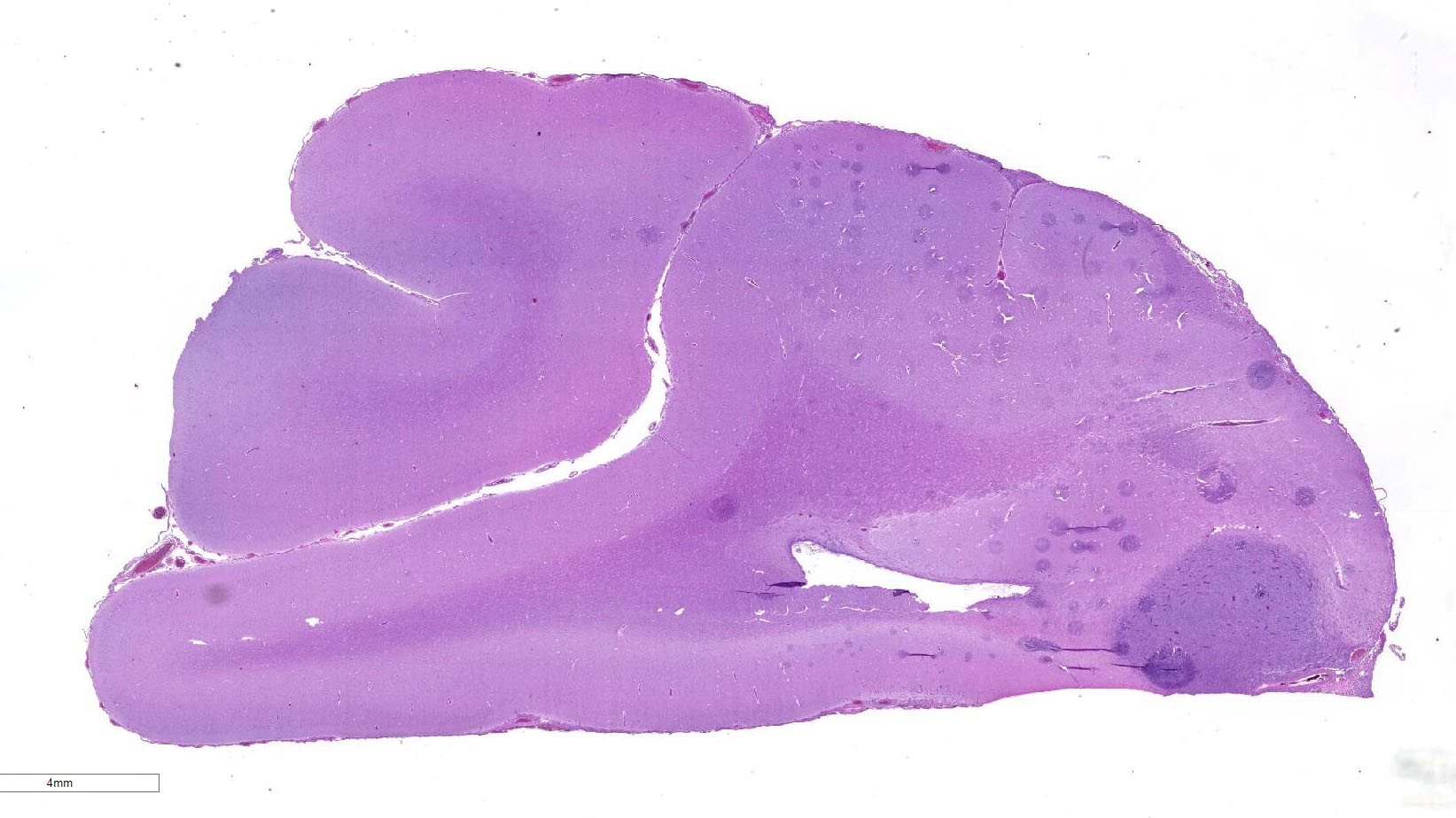
Gross Pathology: Specimens submitted for histopathologic examination included brain (in entirety), nasal turbinate, lung, liver, spleen, and kidney. Gross examination of the brain revealed a focal extensive, 1.6 cm by 1 cm tan, firm, non-encapsulated, invasive mass in the gray matter of the frontal lobe abutting the corpus callosum, centered with an approximate midline – midline right orientation. Multifocally within the mass there are areas of malacia with a tan/red coloration (necrosis). The remainder of the submitted tissues are grossly unremarkable.
Laboratory results: NA
Microscopic Description:
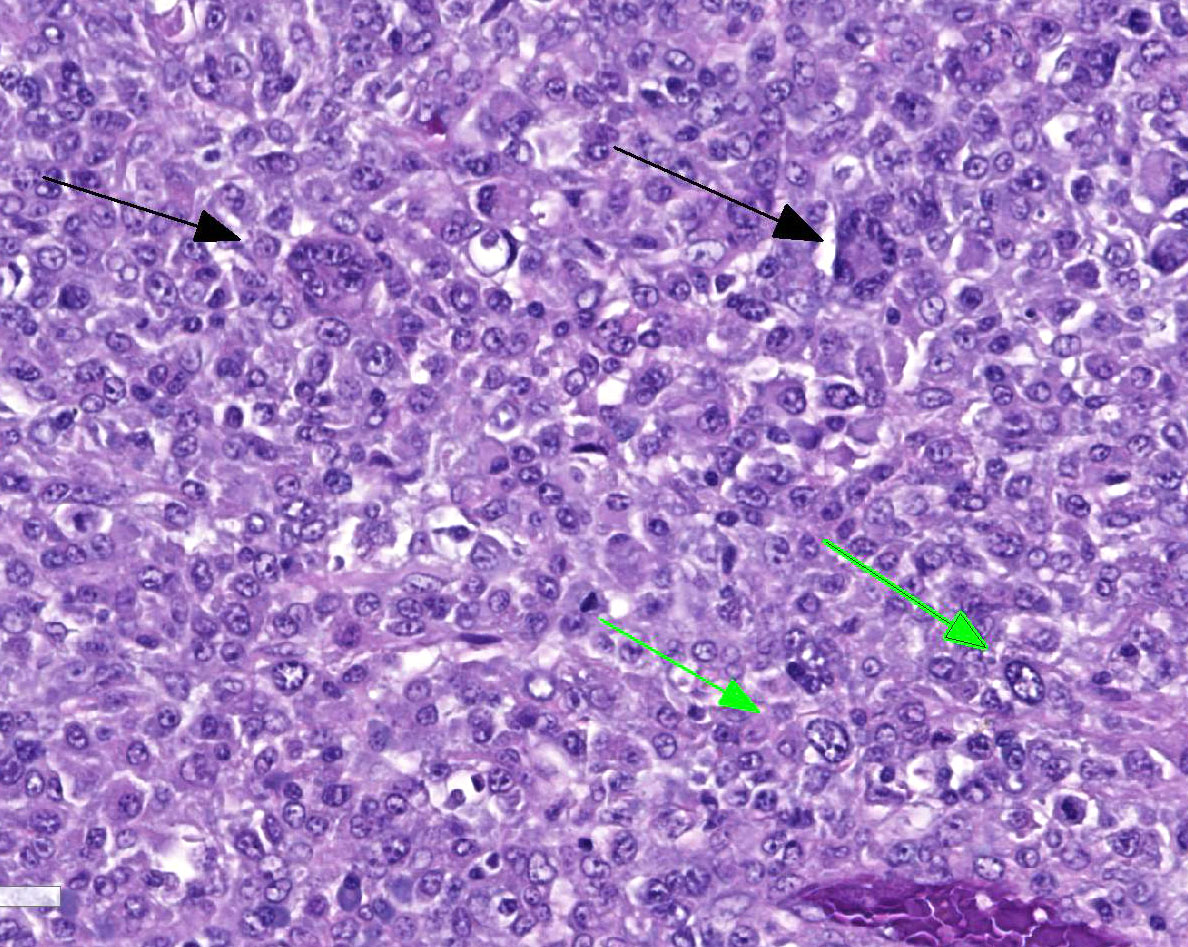
Located in the grey matter of the frontal lobe is a non-encapsulated, poorly demarcated, multicentric, invasive mass of neoplastic round cells arranged in sheets and individual cells and supported on a pre-existing fibrovascular stroma. Neoplastic cells are round to oval with moderate eosinophilic finely vacuolated cytoplasm. Nuclei are generally eccentrically located and round to reniform with coarsely stippled chromatin and up to three prominent nucleoli. There is marked anisocytosis and anisokaryosis, and mitoses number 1-3 per 40x field. Neoplastic cells are often multinucleate, with up to eight nuclei per cell. Neoplastic cells also frequently exhibit erythrophagocytosis. Throughout the mass, there are multifocally moderate numbers of infiltrating lymphocytes and plasma cells, and multifocal areas of necrosis. Multifocally, neoplastic cells invade and expand the meninges. No evidence of vascular or lymphatic invasion is appreciated.
Immunohistochemistry:
Kappa and Lambda Light Chain - Negative
CD3 – Negative
CD20 – Negative
GFAP – Negative
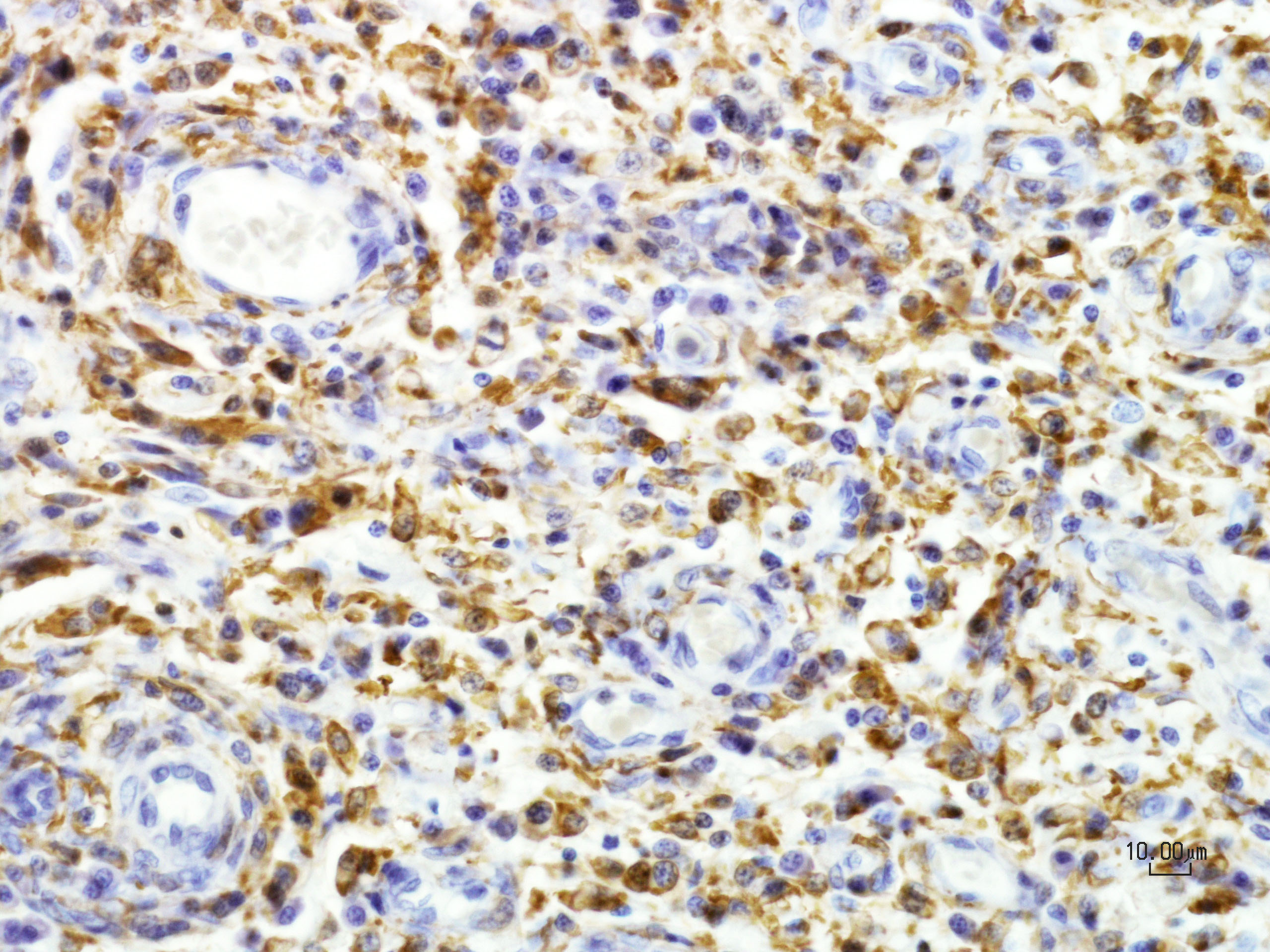
CD163 – Neoplastic cells exhibit variable cytoplasmic and membranous immunoreactivity
Iba1 – Neoplastic cells exhibit strong diffuse cytoplasmic immunoreactivity
Contributor’s Morphologic Diagnoses:
Brain (frontal lobe), primary malignant histiocytosis
Contributor’s Comment: Histologically, the mass in this dog’s brain consisted of aggressive and highly invasive round cells with multicentric extension into the meninges. On immunohistochemistry, neoplastic cells were negative for T, B, and glial cell markers and positive for macrophage/monocyte markers (Iba-1, CD-163). Overall, immunohistochemistry results along with histopathological morphology are consistent with the diagnosis of malignant histiocytosis.
Histiocytic proliferative disorders can be subdivided into local and disseminated forms. Localized forms are referred to as histiocytic sarcomas and are common primary tumors of the liver, spleen, tongue, and stomach wall in the canine patient.7 Malignant histiocytosis is the term most commonly used to describe disseminated neoplastic cells of histiocytic origin. The term “diffuse histiocytic sarcoma” has also been used to designate a localized histiocytic sarcoma that has become systemic. Additionally, benign aspects of histiocytic proliferative disorders include the cutaneous histiocytoma and cutaneous and systemic histiocytosis.
In the canine patient, malignant histiocytosis most commonly occurs in middle age to older male dogs with increased incidence in the Bernese Mountain Dog, Rottweiler, Golden Retriever, and Labrador Retriever.5 Overall, the spleen and liver are the most commonly impacted tissues, but other organs including the lungs, lymph nodes, bone marrow, and central nervous system can also be impacted. Primary cases involving the brain are considered rare, with an overall poor to grave prognosis due to the invasive/aggressive nature of the neoplasia.7
As this case documents, definitive diagnosis of histiocytic tumors can be challenging with histopathology alone, and the use of immunohistochemistry can be advantageous. Overall, malignant histiocytic tumor histopathology is characterized by sheets of highly pleomorphic cells that exhibit severe atypia along with abundant mitotic figures. Noted throughout the neoplastic cells often are multinucleated giant cells, erythophagcytosis, and scattered populations of lymphocytes and plasma cells. Malignant histiocytic tumors exhibit positive staining with CD1c, CD11c, CD45, lysozyme, CD18, and Iba1, and do not exhibit stain uptake for B cell, T cell, and glial cell markers.4,7 Iba1 is a 17-kDa EF hand protein that is specifically expressed in microglia and is not reactive with neurons or astrocytes. Iba1 has been recently recognized as a macrophage marker, expressed by all subpopulations of cells of the monocyte/macrophage lineage with efficacious used in the characterization of canine and feline histiocytic tumors.4
Differentials for this case included: a tumor of neuroglial origin, lymphoma, plasma cell tumor, and granulomatous disease.
Contributing Institution:
http://www.hopkinsmedicine.org/mcp/index.html
JPC Diagnosis: Cerebral cortex: Histiocytic sarcoma
JPC Comment: The contributor has done an excellent job describing this rare entity in the dog. Disseminated histiocytic sarcomas (also referred to malignant histiocytosis) is not an uncommon neoplasm in the dog, but primary lesions in a single organ are.
A diagnosis of primary intracerebral histiocytic sarcoma not only requires immunophenotyping to establish the histiocytic lineage of the neoplastic cells, but also to establish that no other similar lesions are present throughout the other tissues of the body. Recent studies in dogs have identified abnormalities in certain tumor suppressor genes (RB1, PTEN, and CDKN2A/B).3
No gender or age predispositions have been identified for the development of primary intracerebral histiocytic sarcoma, and Pembroke Welsh Corgis have been identified as a predisposed breed. 3
This particular case demonstrates the traditional appearance of a primary CNS histiocytic sarcoma – a single whitish subdural lesion within the cerebrum with a broad meningeal base. Secondary or metastatic lesions are usually multiple, and often arise within the meninges.2 The subdural or meningeal location of these tumors may reflect the restriction of dendritic cells (the presumptive cell of origin for this neoplasm) to the meninges and choroid plexus.2
The moderater reviewed the maturation of cells of the macrophage/monocyte lineage as well as common manifestations of histiocytic diseases in dogs and cats including canine cutaneous histiocytosis, reactive and systemic histiocytosis, feline progressive pulmonary histiocytosis, histiocytic sarcoma and the very rare dendritic cell leukemia.
References:
- Chandra AM, Ginn PE. Primary malignant histiocytosis of the brain in a dog. J Comp Pathol. 1999;121:77–82.
- Higgins RJ, Bollen AW, Dickinson PJ, Siso-Llonch S. Tumors of the nervous system. In: Meuten DJ, ed. Tumors in Domestic Animals. Ames, IA, Wiley Blackwell, 2017, pp. 877, 878.
- Moore, PF. A review of histiocytic diseases of dogs and cats. Vet Pathol 201451:167-184.
- Pierezan F, Mansell J, Ambrus A and Rodrigues Hoffmann A. Immunohistochemical Expression of Ionized Calcium Binding Adapter Molecule 1 in Cutaneous Histiocytic Proliferative, Neoplastic and Inflammatory Disorders of Dogs and Cats. J. Comp. Pathol. 2014, Vol. 151
- Thio T, Hilbe M, Grest P and Pospischil A. Malignant Histiocytosis of the Brain in Three Dogs. J. Comp. Path. 2006, Vol. 134, 241–244
- Uchida K, Morozumi M, Yamaguchi R, and Tateyama S. Diffuse Leptomeningeal Malignant Histiocytosis in the Brain and Spinal Cord of a Tibetan Terrier. Vet Pathol 38:219–222 (2001)
- Zimmerman K, Almy F, Carter L, Higgins M, Rossmeisl J, Inzana K, Duncan R. Cerebrospinal fluid from a 10-year-old dog with a single seizure episode. Veterinary Clinical Pathology Vol. 35 / No. 1 / 2006.