CASE II: 15-185 (JPC 4090739)
Signalment:
Adult female koala (Phascolarctos cinereus)
History:
Hyperkeratotic dermatitis.
Gross Pathology:
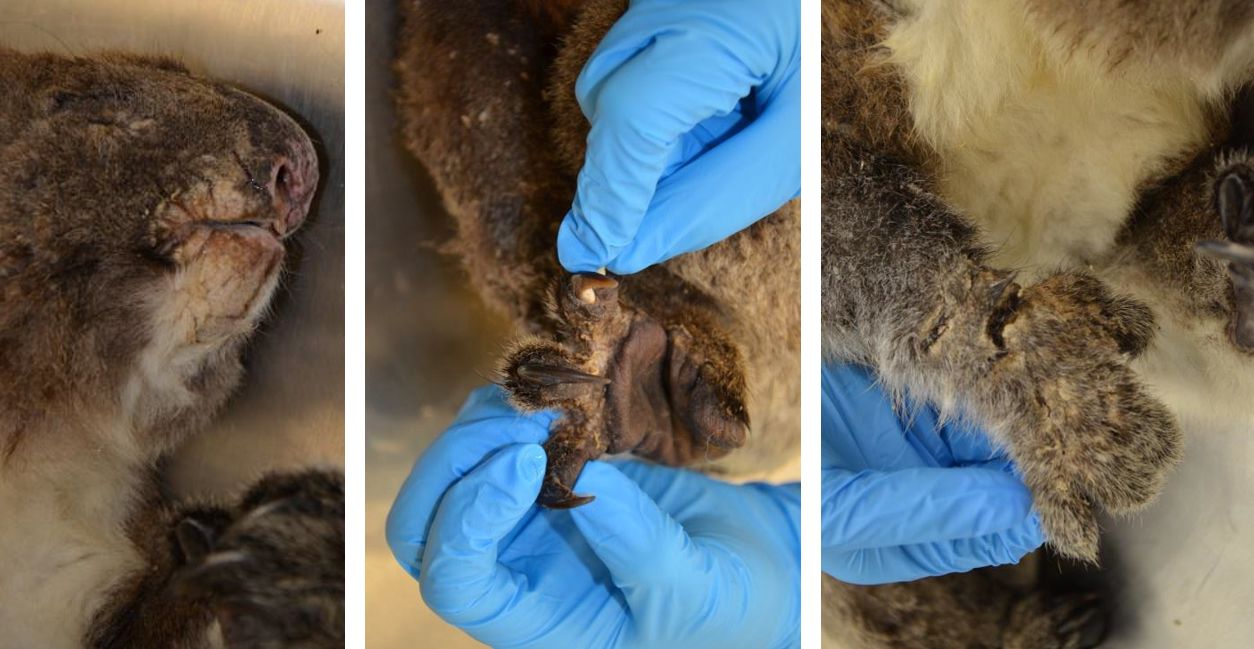
The haired skin over the ventro-medial aspects of the forelimbs, the dorsomedial aspects of the hindlimbs, and between the digits of all four feet is markedly thickened with alopecia and crusting (marked hyperplastic and hyperkeratotic dermatitis). Similar thickening and crusting of the skin with fissuring is noted in the perioral and perinasal haired skin.
Laboratory results:
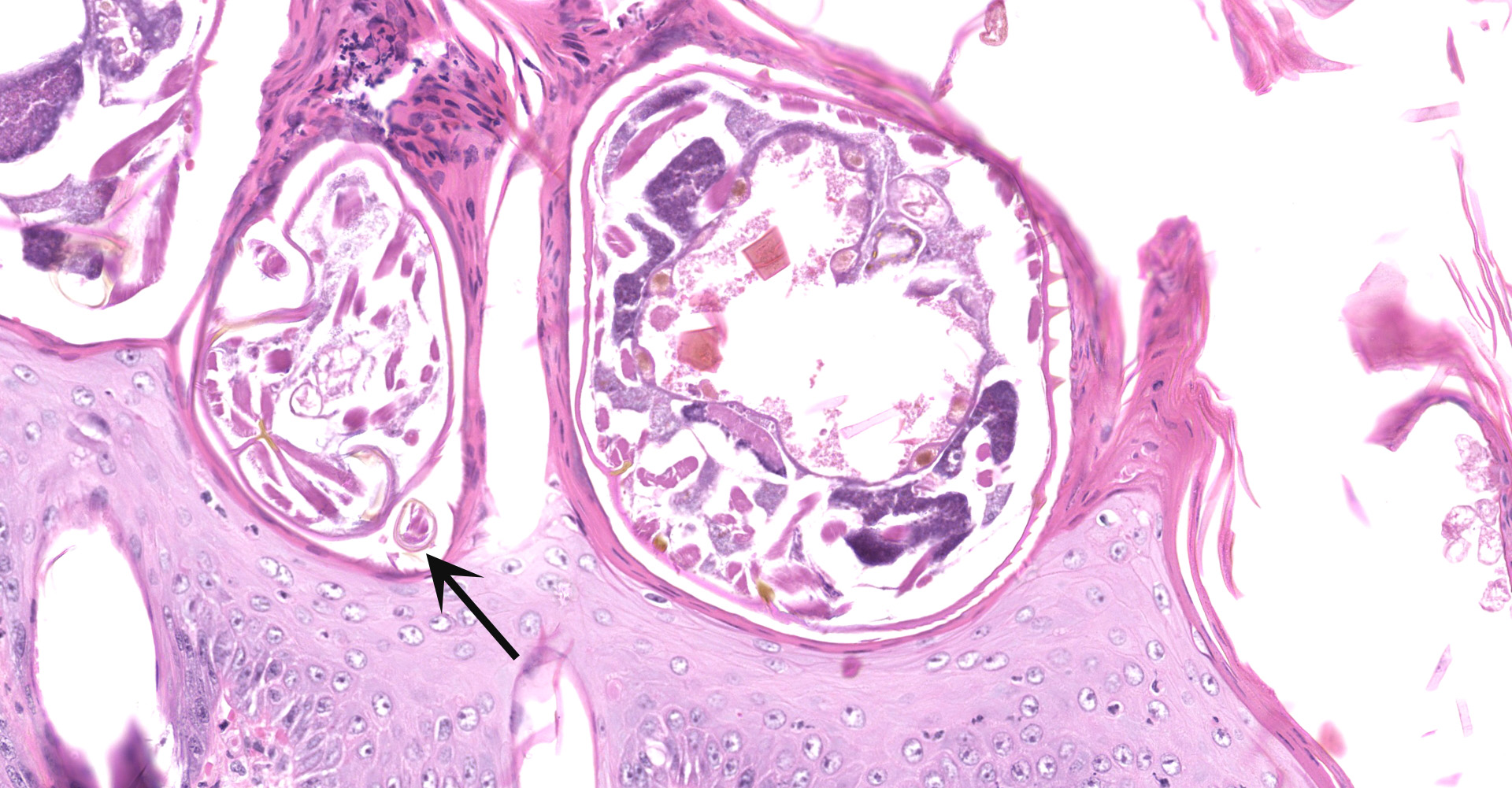
Skin scrapings revealed numerous mites with morphologic features consistent with Sarcoptes scabiei. The adult mites measured up to 450µm in length with round or globoid bodies, very short jointed appendages, a posteriorly positioned anus, and numerous dorsal triangular spines.
Microscopic Description:
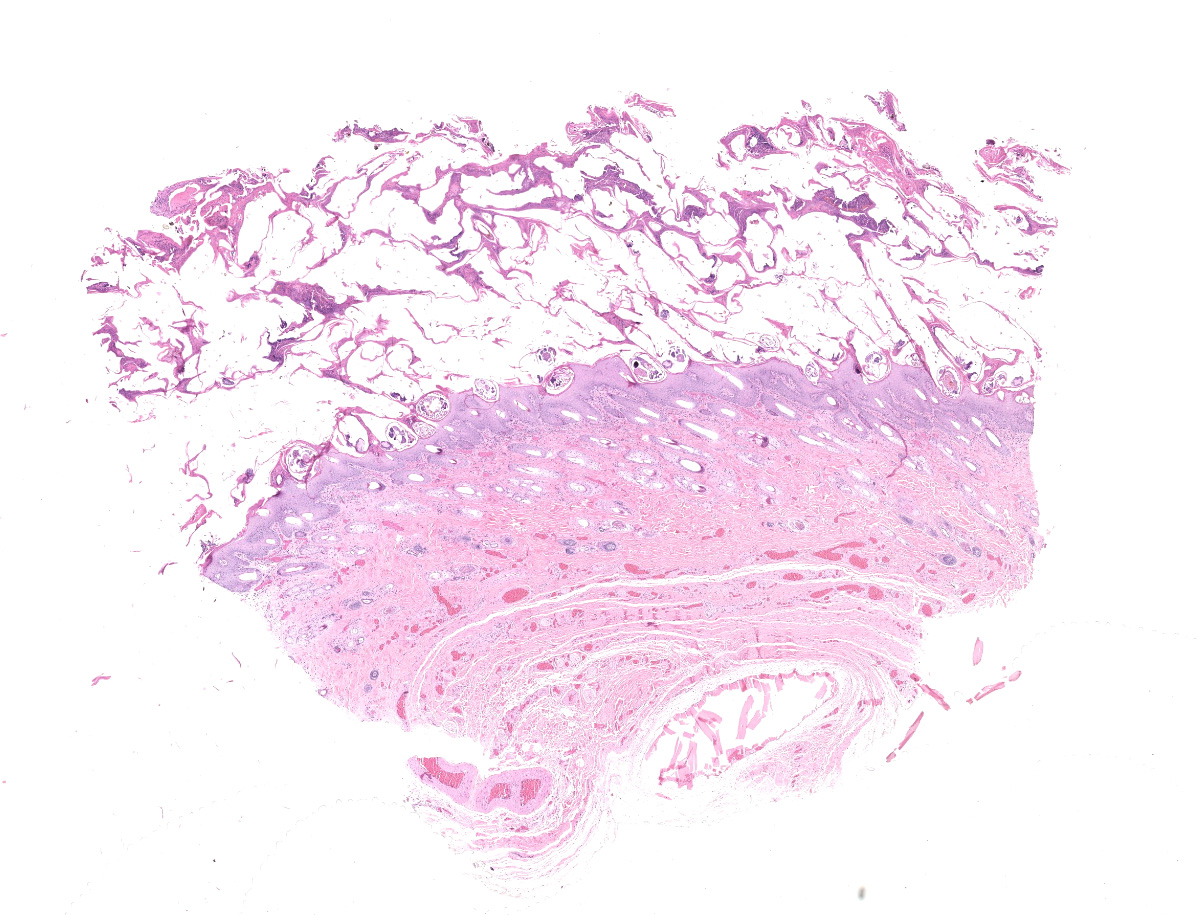
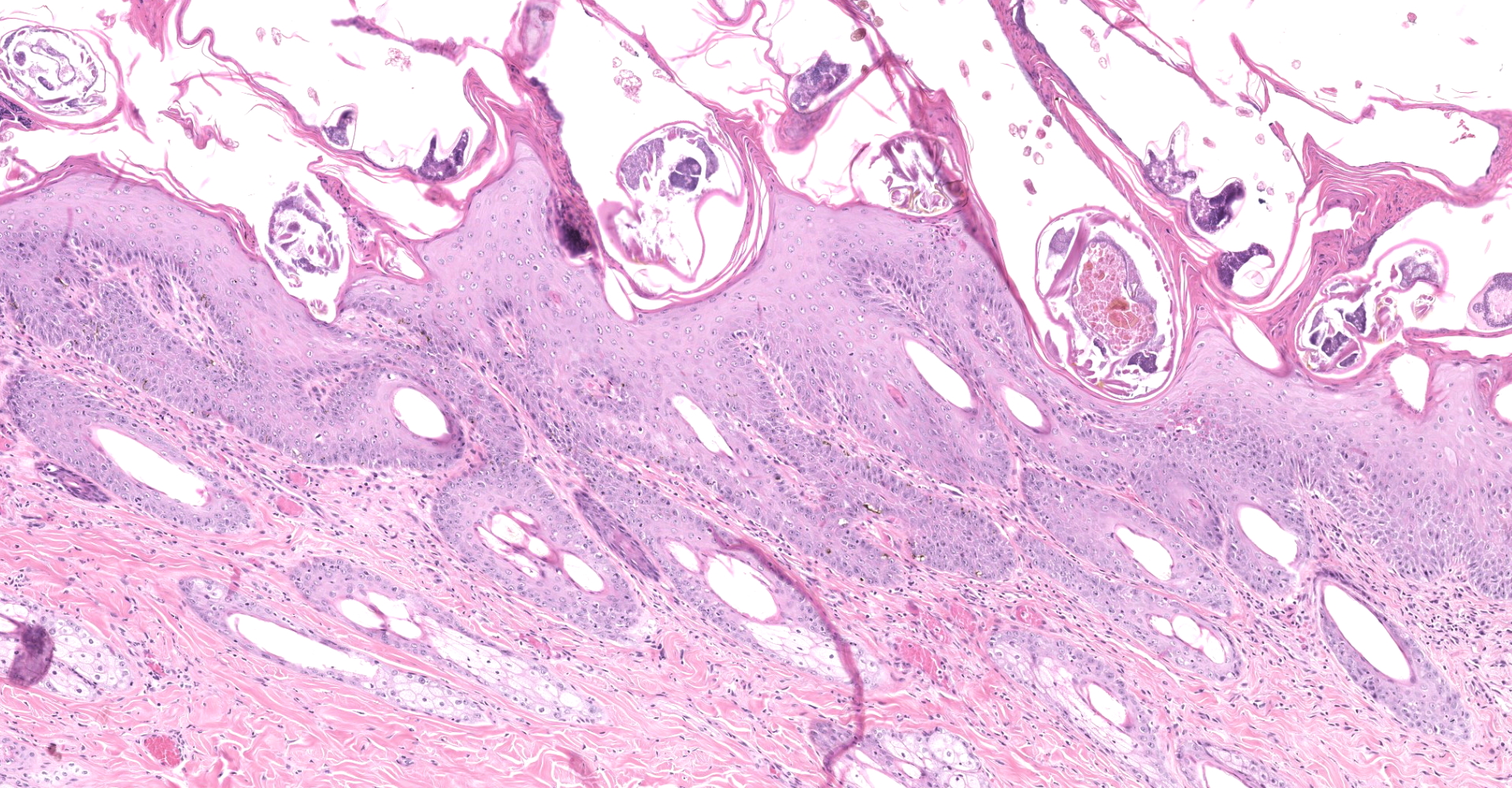
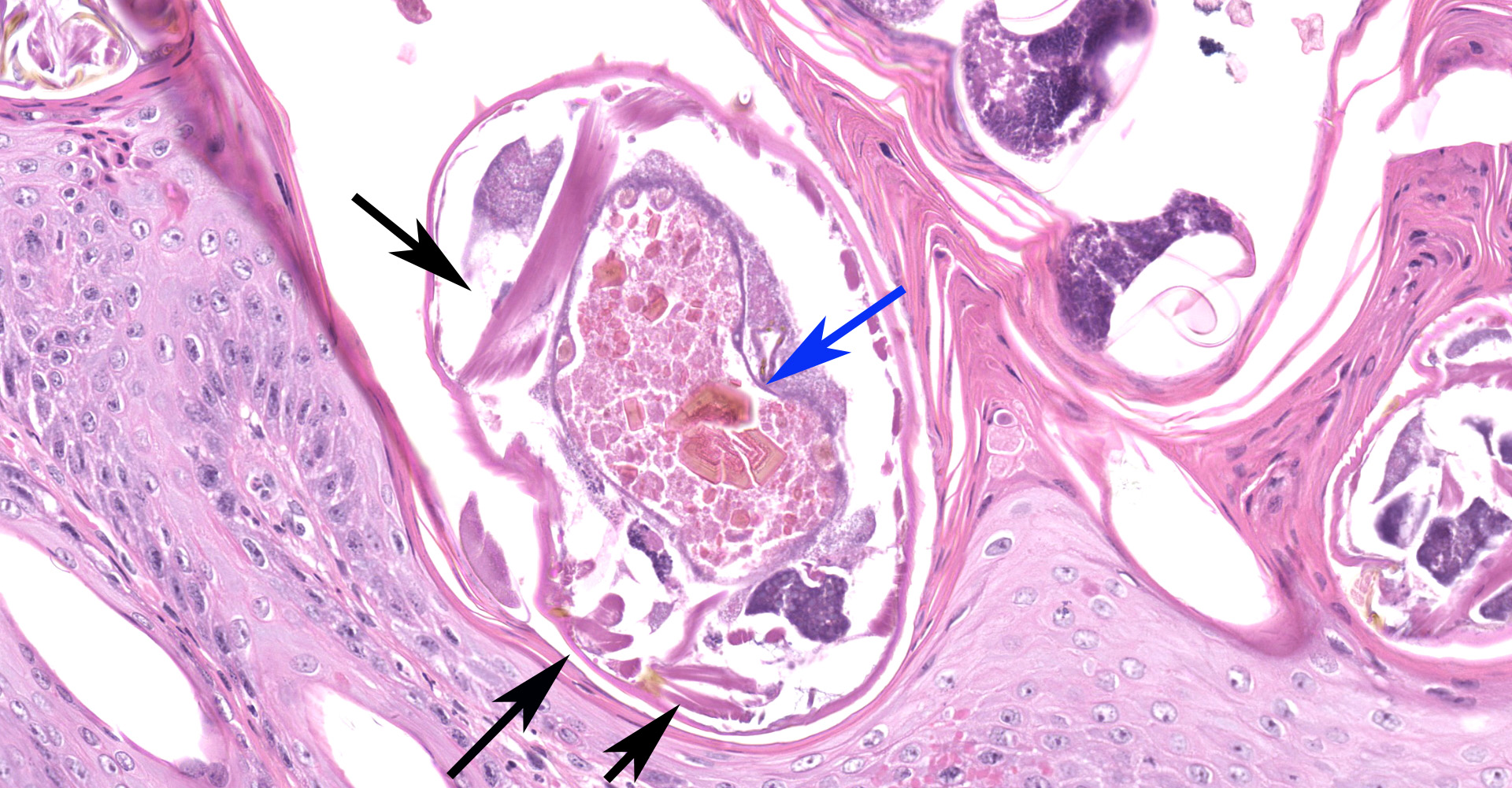
Haired skin: The epidermis is diffusely moderately to markedly thickened by irregular epidermal hyperplasia with mild intercellular edema (spongiosis), intracellular edema, and marked diffuse parakeratotic to orthokeratotic hyperkeratosis. Scattered within the thickened stratum corneum are numerous intracorneal arthropod mites, small to moderate numbers of coccoid bacteria, and multifocal to coalescing areas of neutrophilic and eosinophilic exudation and crusting. The arthropod mites (up to ~350 µm in greatest dimension) have a chitinous exoskeleton with dorsal spines, short jointed appendages, striated skeletal muscles, and internal gastrointestinal and reproductive tracts. The underlying superficial perivascular dermis is infiltrated by small numbers of neutrophils admixed with very small numbers of lymphocytes, plasma cells, macrophages, mast cells, and rare eosinophils. The superficial dermal and mid-dermal blood vessels are diffusely congested. In some sections (facial skin), the epidermis contains areas of erosion and ulceration (possible excoriations) with marked local fibrinosuppurative exudation and crusting and underlying superficial dermal edema.
Contributor's Morphologic Diagnoses:
Haired skin: Severe, diffuse, subacute to chronic, neutrophilic and hyperplastic dermatitis, with marked parakeratotic to orthokeratotic hyperkeratosis, neutrophilic exudation and crusting, moderate to large numbers of superficial coccoid bacteria, and numerous intracorneal mites (Sarcoptes scabiei, presumed).
Contributor's Comment:
Sarcoptic mange (referred to as scabies in humans) is a highly contagious skin disease caused by the parasitic mite, Sarcoptes scabiei. Sarcoptic mange has a broad host range and has been reported in over 100 species of animals including domestic dogs, livestock (e.g., cattle, pigs, goats, camelids), and wildlife (e.g., red foxes, wild dogs, coyotes, wolves, dingoes, deer, ibex, cheetahs, lions, bobcats, gorillas, wombats, koalas and wallabies).2,6 Sarcoptic mange is a major disease of Australian wildlife, particularly in wombats (bare-nosed/common and southern hairy-nosed), which also impacts humans, domestic animals, and other Australian wildlife.2 Reports of the disease were previously rare in koalas but more recently several outbreaks of sarcoptic mange have been observed in Australia (Dr. Lucy Woolford, personal obs.).7
Sarcoptic mites parasitizing the various host species are largely morphologically indistinguishable, thus it is unclear if the mites parasitizing different mammalian host species are different species or if they are strains or variants (pathovars) of one species.2,6 Some strains appear to parasitize a single host species while others parasitize multiple host species (e.g., dogs and rabbits), and infestations from prey-to-predator appear to occur in some ecosystems.1,6 Cross-infestations between different host species have been shown to occur, but these documented spillover events have typically been self-limiting.2
Concurrent pyoderma is often observed in cases of sarcoptic mange, as seen in this case. Sarcoptes scabiei infestation results in injury to the epidermal layer caused by a com-bination of host scratching and the mechanical damage by the burrowing mites and provides an entry point for the invading bacteria. Localized complement inhibition by excreted mite products in the mite burrow may provide a protected microenvironment in which bacteria can initiate colonization without immediate attack by the host immune system and thus play a role in the development of pyoderma in scabies mite infested skin.1,4,8
Contributing Institution:
University of Adelaide, School of Animal and Veterinary Sciences
http://www.adelaide.edu.au/vetsci/
JPC
Diagnosis:
Haired skin: Epidermal hyperplasia and hyperkeratosis, diffuse, marked, with mild eosinophilic dermatitis and numerous intracorneal mites and eggs, etiology consistent with Sarcoptes scabiei.
JPC Comment:
Sarcoptes scabiei is a submacroscopic burrowing mite in which male and female adults, larvae, protonymphs, tritonymphs, and eggs are found within the epidermis, extending to the level of the stratum granulosum, and consume living cells and tissue fluid.5 As noted by the contributor, there are no definitive means to taxonomically distinguish the various strains that tend to demonstrate a high degree of host specificity. Thus S. scabiei as a species is divided into morphologically indistin-guishable variants with a high degree of host specificity and low degree of cross infectivity. As a result, varieties are named for their predominant host species, such as S. scabiei var. canis, S. scabiei var suis, etc.5
Infection occurs either via direct or indirect transmission. Direct transmission occurs via contact between an infected host and an uninfected animal since larvae and nymphs leave their burrows and wander the skin's surface. Alternatively, indirect transmission may occur since S. scabiei are capable of surviving off the host for potentially up to three weeks in low temperature and high relative humidity environments, such as former dens of infected animals.2,5 Host species population density also tends to influence the parasite's prevalence since transmission is dependent on direct and indirect contact.5
Following exposure, S. scabiei burrows into the skin, usually to the level of the stratum granulosum, using a combination of its mouthparts (chelicerae and gnathosoma) and cutting hooks on its legs. Digestive enzymes may also play a role in burrowing and feeding but have not yet been described.5
A significant component of S. scabiei's pathogenesis is due to host hypersensitization as a result of mechanical disruption of the skin, excretions and secretions of the mite, and the massive amount of antigenic material produced (including dead mites, molted skins of adults and immature life stages, and egg shells). Both type I and type IV hyper-sensitivity reactions have been demonstrated in domestic animals such as the dog and pig; however, only type I hypersensitivity reactions were demonstrated in experi-mentally infected red foxes, which was also seems to occur in coyotes, even in advanced cases. Initial lesions in many hosts are localized and nonpruritic. After a period of several weeks an allergic response develops, resulting in generalized urticarial eruptions and severe pruritus. Depending on the host, this may or may not be accompanied by a delayed hypersensitivity response, which is associated with low numbers of mites within the lesion. In contrast, recently exposed, previously naïve new hosts as well as anergic and malnourished animals may develop non-pruritic, severe crusting dermatitis with severe hyperkeratosis, serosanginous exudate, and large numbers of mites.5
Numerous epizootics of sarcoptic mange in wildlife have been documented, frequently resulting in devastatingly high losses, such as a 70% of the southern Texas coyote population in one epizootic. During the 1960s and 1970s an epizootic in Fenno-scandia red foxes resulted in a 50% population decline overall and up to 90% mortality in some areas. Similar epizootics have been reported in multiple species across the globe and seem to cycle every 30-45 years in some areas. Despite very high losses, many populations recover over time. However, remnant or isolated populations, even species not listed by the Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES), can be severely affected by sarcoptic mange. As an example, naïve red foxes on the island of Borrnholm in Denmark completely disappeared following introduction of sarcoptic mange. Remnant populations of endangered species identified to be at particular risk include the Bonobo chimpanzee (Pan panisci), Ugandan mountain gorillas, cheetahs, wild dogs, chamois, Iberian Ibex, and the highly endangered European mouflon (Ovis orientialis musimoon).5
References:
1. Bergström FC, Reynolds S, Johnstone M, Pike RN, Buckle AM, Kemp DJ, Fischer K, Blom AM. Scabies mite inactivated serine protease paralogs inhibit the human complement system. J Immunol. 2009;182(12):7809-17.
2. Fraser TA, Charleston M, Martin A, Polkinghorne A, Carver S. The emergence of sarcoptic mange in Australian wildlife: an unresolved debate. Parasit Vectors. 2016;9(1):1.
3. Gakuya F, Rossi L, Ombui J, Maingi N, Muchemi G, Ogara W, Soriguer RC, Alasaad S. The curse of the prey: Sarcoptes mite molecular analysis reveals potential prey-to-predator parasitic infestation in wild animals from Masai Mara, Kenya. Parasit Vectors. 2011;4(1):1.
4. Holt DC, Fischer K. Novel insights into an old disease: recent developments in scabies mite biology. Curr Opin Infect Dis. 2013;26(2):110-5.
5. Pence DB, Ueckermann E. Sarcoptic manage in wildlife. Rev Sci Tech. 2002;21(2):385-398. (Rearranged contributor's, which were initially in order of use?not alphabetized)
6. Rider SD, Morgan MS, Arlian LG. Draft genome of the scabies mite. Parasit Vectors. 2015;8(1):1.
7. Speight K, Whiteley PL, Woolfod L, Duigan PJ, Bacci B, Lathe S, Boardman W, Scheelings TF, Funnell O, Underwood G, Stevenson MA. Outbreaks of sarcoptic mange in free-ranging koala populations in Victoria ans South Australia: A case series. Aus Vet J (in press).
8. Swe PM, Reynolds SL, Fischer K. Parasitic scabies mites and associated bacteria joining forces against host complement defence. Parasit Immunol. 2014;36(11):585-93.