Signalment:
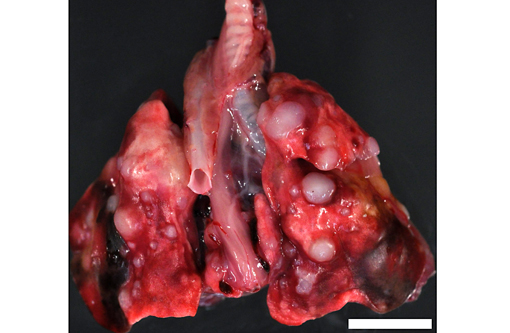
Gross Description:
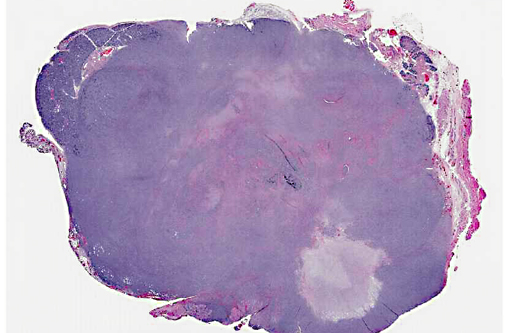
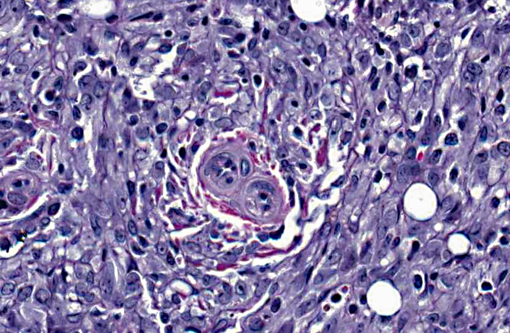
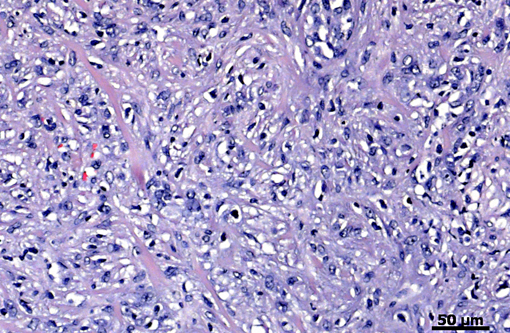
Histopathologic Description:
Review of a Massons trichrome-stained section of tumor revealed that the neoplastic epithelial cells were negative for collagen while the desmoplastic tissue response was positive for collagen.
Morphologic Diagnosis:
Other significant microscopic findings (not submitted): Haired Skin, Incision Site of Tumor Removal: There are numerous small clusters to large foci (0.4 CM) of neoplastic cells similar in appearance to the primary tumor described in the biopsy report above scattered within the dermis and subcutis.
Lung, Thymus (none visible): There is a metastatic neoplastic process admixed with hemorrhage and inflammation that effaces and replaces at least 50% of the lung. The neoplastic cells are similar in appearance to those described for the salivary gland mass above, with the exception of a greater epithelial cell component. Clusters and islands of neoplastic cells are frequent within alveoli. Hemorrhage, fibrin and inflammatory cells (macrophages) predominate within adjacent alveolar spaces.
Liver, Kidney, Adrenal Gland, Spleen, Pancreas: Within the one visible adrenal gland, 75% of the cortex and medulla are effaced and replaced by neoplastic cells similar in appearance to those described for the salivary gland above. There is bilateral mild to moderate chronic progressive glomerulonephropathy.
Condition:
Contributor Comment:
Benign salivary gland tumors tend to occur more commonly in females, whereas malignant tumors are more prevalent in males.(10) Malignant salivary gland tumors can be diagnosed using the World Health Organization (WHO) classification system.(22) In general, mucoepidermoid carcinomas account for over 50% of malignant salivary gland tumors in humans, while adenocarcinomas were reported in only 12% of the cases. Tumors have been documented in all three major salivary glands, as well as in minor salivary glands; however, the most common site in humans is the parotid gland and the second most common being the submaxillary gland.(2,4,10,20,22)
In humans, submandibular gland tumors account for 5-15% of all salivary gland tumors and less than 1% of all malignant head and neck tumors, however, over 50% are malignant.(1) Malignant submandibular adenocarcinomas in humans tend to have areas of necrosis and hemorrhage with irregular and ill-defined margins that infiltrate growth into surrounding tissues. They tend to form nests or islands of tumor cells and have a widespread ductal differentiation. Clinically, over 20% of patients have reported pain and facial weakness associated with the mass.(14) Most tumors have progressed to high grade by the time they are diagnosed; therefore, these malignant submandibular salivary adenocarcinomas have a poor prognosis in humans.
Primary salivary gland tumors are also rare in domestic animals and have been reported in dogs(9,17), cats(13), lions(5), and cows.(3) In dogs, the most common salivary gland neoplasms are adenocarcinomas and carcinomas. There are two reports that describe rare cases in dogs, a malignant fibrous histiocytoma in a boxer(9) and another report of an osteosarcoma of the mandibular salivary gland in a collie.(17) Primary salivary gland tumors in cats are commonly carcinomas found in the minor salivary glands, rather than the parotid gland.(13) A single case of a high-grade mucoepidermoid carcinoma of the mandibular salivary gland has been reported in a lion.(5) There have also been three cases of parotid gland carcinomas reported in cows, two of which were pleomorphic carcinomas, and one was a squamous cell carcinoma.(3) Primary salivary gland tumors are also rare in rodents.(6,7,12,15,16,18,19,23) There has also been a single report of a Mongolian Gerbil developing a pleomorphic sarcoma of the salivary gland(18), and a ground squirrel with an adenocarcinoma of a minor salivary gland.(23) Mice very rarely develop salivary gland tumors, but myoepitheliomas have been reported to occur spontaneously in certain inbred strains of mice.
Spontaneous primary salivary gland tumors are also rare in rats, but may be chemically induced.(15,25) A study during carcinogenesis in rat submandibular glands showed that histopathology and immunohistochemistry is a useful tool for tumor histiogenesis.(15) Submandibular salivary gland adenocarcinomas in rats are poorly differentiated, show local invasion, and, in most cases, incite a significant desmoplastic response.(7) They should also exhibit the same characteristics seen in human salivary glands, including nests or islands of tumor cells, and tumor-grade specific nuclear and mitoses characteristics. In this case, the rat demonstrated a significant amount of irregular epithelial cells, marked nuclear pleomorphism, prominent nucleoli, large area of necrosis, scattered foci of hemorrhage, a variable number of mitotic figures, with a desmoplastic tissue response. All of these characteristics are consistent with a diagnosis of a high grade submandibular salivary gland adenocarcinoma. Metastasis to the lungs and adrenal gland also indicates that this was a high-grade tumor with a poor prognosis.
JPC Diagnosis:
Conference Comment:
References:
2. Bjorndal K, Krogdahl A, Therkildsen MH, Overgaard J, Johansen J, Kristensen CA, et al. Salivary gland carcinoma in Denmark 1990-2005: a national study of incidence, site and histology. Results of the Danish Head and Neck Cancer Group (DAHANCA). Oral Oncol. 2011;(47):677-682.
3. Bundza A. Primary salivary gland neoplasia in three cows. J Comp Pathol. 1983;93:629-632.
4. Cho KJ, Ro JY, Choi J, Choi SH, Nam SY, Kim SY. Mesenchymal neoplasms of the major salivary glands: clinicopathological features of 18 cases. Eur Arch Otorhinolaryngol. 2008;(265 Suppl 1): S47-56.
5. Dorso L, Risi E, Triau S, Labrut S, Nguyen F, et al. High-grade mucoepidermoid carcinoma of the mandibular salivary gland in a lion (Panthera leo). Vet Pathol. 2008;45:104-108.
6. Ishikawa Y, Nishimori K, Tanaka K, Kadota K. Naturally occurring mucoepidermoid carcinoma in the submandibular salivary gland of two mice. J Comp Pathol. 1998;118:145-149.
7. Nishikawa S, Sano F, Takagi K, Okada M, Sugimoto J, Takagi S. Spontaneous poorly differentiated carcinoma with cells positive for vimentin in a salivary gland of a young rat. Toxicol Pathol. 2010;38:315-318.
8. Pang B, Leong CC, Salto-Tellez M, Petersson F. Desmoplastic small round cell tumor of major salivary glands: report of 1 case and a review of the literature. Appl Immunohistochem Mol Morphol. 2011;19:70-75.
9. Perez-Martinez C, Garcia Fernandez RA, Reyes Avila LE, Perez-Perez V, Gonzalez N, Garcia-Iglesias MJ. Malignant fibrous histiocytoma (giant cell type) associated with a malignant mixed tumor in the salivary gland of a dog. Vet Pathol. 2000;37:350-353.
10. Pinkston JA, Cole P. Incidence rates of salivary gland tumors: results from a population-based study. Otolaryngol Head Neck Surg. 1999;120:834-840.
11. Schaefer O, Hildes JA, Medd LM, Cameron DG. The changing pattern of neoplastic disease in Canadian Eskimos. Can Med Assoc J. 1975;112:1399-1404.
12. Slavin BG, Paule WJ, Bernick S. Morphological changes in the submandibular gland of aging rats. Gerodontology. 1989;8:53-58.
13. Sozmen M, Brown PJ, Eveson JW. Salivary duct carcinoma in five cats. J Comp Pathol. 1999;121:311-319.
14. Spiro RH, Huvos AG, Strong EW. Adenocarcinoma of salivary origin. Clinicopathologic study of 204 patients. Am J Surg. 1982;144:423-431.
15. Sumitomo S, Hashimura K, Mori M. Growth pattern of experimental squamous cell carcinoma in rat submandibular glands--an immunohistochemical evaluation. Eur J Cancer B Oral Oncol. 1996;32B:97-105.
16. Sundberg JP, Hanson CA, Roop DR, Brown KS, Bedigian HG. Myoepitheliomas in inbred laboratory mice. Vet Pathol. 1991;28:313-323.
17. Thomsen BV, Myers RK. Extraskeletal osteosarcoma of the mandibular salivary gland in a dog. Vet Pathol. 1999;36:71-73.
18. Toyoda T, Tsukamoto T, Cho YM, Onami S, Takasu S, Shi L, et al. Undifferentiated sarcoma of the salivary gland in a Mongolian gerbil (Meriones unguiculatus). J Toxicol Pathol. 2011;24:173-177.
19. Tsunenari I, Yamate J, Sakuma S. Poorly differentiated carcinoma of the parotid gland in a six-week-old Sprague-Dawley rat. Toxicol Pathol. 1997;25:225-228.
20. Vargas PA, Gerhard R, Araujo Filho VJ, de Castro IV. Salivary gland tumors in a Brazilian population: a retrospective study of 124 cases. Rev Hosp Clin Fac Med Sao Paulo. 2002;57:271-276.
21. Wallace AC, Macdougall JT, Hildes JA, Lederman JM. Salivary gland tumors in Canadian Eskimos. Cancer. 1963;16:1338-1353.
22. Barnes L. Tumors of the salivary glands. In: Barnes L, Eveson JW, Reichart P, Sidransky D, eds. World Health Organization (WHO): The WHO Classification of Head and Neck Tumours. Pathology and Genetics of Head and Neck Tumours. Lyon, France: International Agency for Research on Cancer (IARC) Press. 2005:209-281.
23. Yamate J, Yamamoto E, Nabe M, Kuwamura M, Fujita D, Sasai H. Spontaneous adenocarcinoma immunoreactive to cyclooxygenase-2 and transforming growth factor-beta1 in the buccal salivary gland of a Richardson's ground squirrel (Spermophilus richardsonii). Exp Anim. 2007;56:379-384.
24. Yin WH, Guo SP, Yang HY, Chan JK. Desmoplastic small round cell tumor of the submandibular gland--a rare but distinctive primary salivary gland neoplasm. Hum Pathol. 2010;41:438-442.
25. Zaman A, Kohgo T, Shindoh M, Iizuka T, Amemiya A. Induction of adenocarcinomas in the submandibular salivary glands of female Wistar rats treated with 7,12-dimethylbenz(a)anthracene. Arch Oral Biol. 1996;41:221-224.