Signalment:
1-year-old intact female Sprague-Dawley rat, (
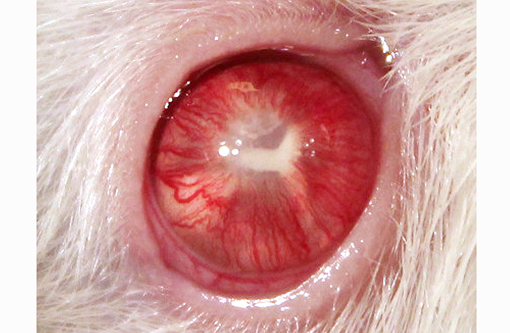
Rattus norvegicus).A one-year-old intact female experimentally na+�-�ve Sprague-Dawley rat (NTac:SD) used as a soiled bedding sentinel was noted to have buphthalmia of the right eye and had multiple foci of white tissue within the anterior chamber, in addition to a Y-shaped band of tissue (2mm in diameter) present within the posterior chamber. The vasculature surrounding the pupil was prominent and the anterior chamber was cloudy. Apart from the ocular lesion, the animal appeared to be in good health. The animal was monitored and the lesion showed no signs of progression. The animal was later euthanized for routine sentinel testing.
Gross Description:
Gross lesions were observed in the right eye, as described in the history. No other gross changes were observed on complete necropsy.Â
Histopathologic Description:
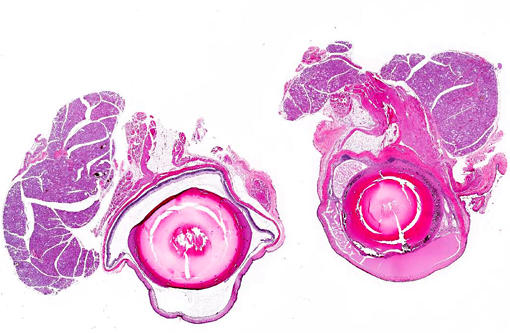
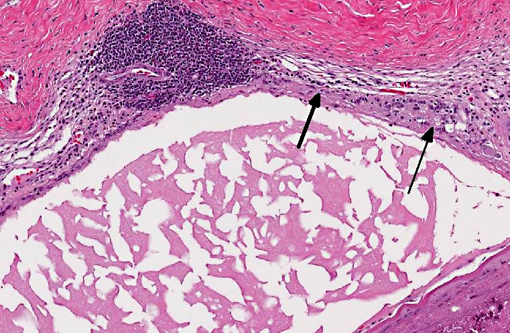
Right eye: The lens is misshapen and has a wrinkled capsule. Multifocally extensive areas of loss of sub-capsular lenticular fibers (both on anterior and posterior surfaces) and replacement by mineralized debris are evident. Lenticular epithelium is hyperplastic and there is migration of epithelium to the posterior lens surface. Several lenticular epithelial cells are spindle-shaped (fibrous metaplasia).Â
Multifocal adhesions between the lens and posterior surface of the iris (posterior synechiae) are present. Extensive fibrovascular membranes are present and are adhered to the ciliary body, posterior lens capsule, and choroid (cyclitic membranes). The retina shows full thickness atrophy. The iris shows mild vascular congestion. The iris, ciliary body and choroid are thickened by fibroplasia and an inflammatory infiltrate composed of numerous lymphocytes, and moderate numbers of plasma cells, eosinophils, neutrophils and hemosiderin-laden macrophages, with multifocal formation of lymphocytic aggregates in the choroid. Remnant spaces in the posterior chamber that are bordered by cyclitic membranes are filled with eosinophilic homogenous proteinaceous fluid.
The anterior chamber is filled with eosinophilic homogenous proteinaceous fluid (plasmoid aqueous).Â
Left eye (section not provided): Within normal limits.
Morphologic Diagnosis:
Right eye:
1. Marked panuveitis, lymphocytic, with posterior synechiae and cyclitic membranes, chronic.
2. Hypermature cataract, subcapsular, chronic.
3. Retina: Marked retinal atrophy, chronic.
Condition:
Cataract formation, spontaneous
Contributor Comment:
Histopathological changes in the right eye are consistent with advanced cataract, and other changes within the eye suggest phacolysis (lens protein leakage) leading to secondary uveitis (phacolytic uveitis). The pathogenesis is suspected to be leakage of lenticular protein from the lens (even with intact lens capsule) leading to uveal inflammation. Predominant lymphocytic uveitis present in this case is also consistent with phacolysis leading to secondary uveitis. The Y-shaped macroscopic appearance of the aforementioned cataract is similar to the congenital sutural cataract that occurs along the suture lines of the lens in humans and may also be observed as a late change in advanced cataracts of other etiologies. Although suture lines are present in both anterior and posterior lens capsules, it is the posterior capsular suture lines that manifest a characteristic Y-shape grossly when cataract forms along its margins. Some forms of non-congenital mature cataracts can progress along suture lines.
Cataracts are commonly associated with aging in various strains, including Sprague-Dawley (SD) rats. No sex predilection for the development of cataracts has been reported in the literature. Capsular cataracts, specifically posterior sub-capsular cataracts, are noted with increasing incidence starting at week 57 of age in SD rats (Week 57- 2.7%, Week 83- 8.5% and Week 110- 13.4%).(1) Diffuse opacification of the lens along the posterior suture lines has also been reported in this strain.1 Anterior cortical striations that can eventually progress to senile cataracts are commonly seen in SD rats between 83 and 110 weeks.(1) It is interesting to note that this case has hypermature cataract in both anterior and posterior subcapsular regions. It is difficult to ascertain the region where the cataract first started. However, based on the reported age-based incidence, it is speculated that the posterior subcapsular region is the first site of cataract formation. It is also possible that both anterior and posterior regions co-evolved the cataractous change independently of one another.
Approximately 3-4% of Sprague-Dawley rats have minor lens lesions comprised of foci of swollen or degenerate lens fibers starting as early as 19-20 days of gestation and these may represent early changes that may eventually lead to cataract.(2) Glaucoma has been described as a complication of advanced cataracts in rats, and the proposed pathogenesis was lens-induced uveitis.(3)
JPC Diagnosis:
Eye: Uveitis, lymphoplasmacytic, chronic, diffuse, severe, with cataract formation, drainage angle occlusion, and posterior synechiae.
Conference Comment:
Cataract is the most common lens disorder in any domestic species; it can be distinguished microscopically from fixation/sectioning artifact by (in order of occurrence) detection of Morgagnian globules (eosinophilic globules of denatured lens protein), bladder cells (large, foamy nucleated cells that may represent abortive epithelial attempts at new lens fiber formation), lens epithelial hyperplasia and/or posterior migration of lens epithelium (occasionally followed by fibrous metaplasia), and mineralization. Hypermature cataracts often exhibit residual nuclei within lakes of proteinaceous fluid, surrounded by a wrinkled capsule. Cataracts can be inherited (i.e. familial cataracts in dogs) or secondary to anatomic anterior segment anomalies; formation may also be induced by a variety of stimuli, including solar (or other) irradiation, cold, increased intraocular pressure, toxins, nutritional derangements, local inflammation and direct trauma. Most cataracts encountered within veterinary medicine are classified as inherited, post-inflammatory or idiopathic; however, cataract formation due to canine diabetes, galactose accumulation in kangaroos and wallabies raised on cows milk, arginine deficiency in puppies, wolf cubs, or kittens on milk replacer, dietary deficiencies (in sulfur-containing amino acids, zinc or vitamin C) or solar irradiation in farmed fish and administration of the aminoglycoside hygromycin B in sows, have also been described, although the pathogenesis of these examples is not necessarily fully elucidated.(4) Additionally, as noted by the contributor, cataract formation is a common age-related change in several strains of rats.
In this case, we concur with the contributors comprehensive histological description as well as the proposed pathogenesis. The formation of age-induced cataracts likely resulted in lens protein leakage (phacolysis), with subsequent phacolytic uveitis, occlusion of the drainage angle, increased intraocular pressure, glaucoma and eventually, full thickness retinal atrophy.Â
References:
1. Taradach C, Regnier B, Perraud J. Eye lesions in Sprague-Dawley rats: type and incidence in relation to age. Lab Anim. 1981;15(3):285-287.
2. Taradach C, Greaves P. Spontaneous eye lesions in laboratory animals: incidence in relation to age. Crit Rev Toxicol. 1984;12(2):121-147.
3. Wegener A, Kaegler M, Stinn W. Frequency and nature of spontaneous age-related eye lesions observed in a 2-year inhalation toxicity study in rats. Ophthalmic Res. 2002; 4:281-287.Â
4. Wilcock BP. Eye and ear. In: Maxie MG, ed. Jubb, Kennedy and Palmers Pathology of Domestic Animals. Vol 1. 5th ed. Philadelphia, PA: Elsevier; 2007:494-497.