Signalment:
5-month-old, female, mixed-breed dog (
Canis familiaris).This puppy, purchased in Texas, but living in Indiana at presentation, was evaluated for chronic respiratory
distress (present since purchase at 8 weeks of age) and a 2-month history of progressive neurologic dysfunction.
Littermates had responded to antibiotic therapy for
Bordetella infection, but this puppys respiratory condition had
not improved. Serum titers were positive for canine distemper virus. Radiographically, the lungs had a diffuse
interstitial pattern. A cerebral mass was detected in the frontal/parietal lobes by magnetic resonance imaging.
Histologic evaluation of a biopsy specimen of the cerebral mass resulted in a diagnosis of granulomatous amebic
encephalitis. The dog died about 36 hours after the cerebral biopsy procedure and was presented for necropsy
examination.
Gross Description:
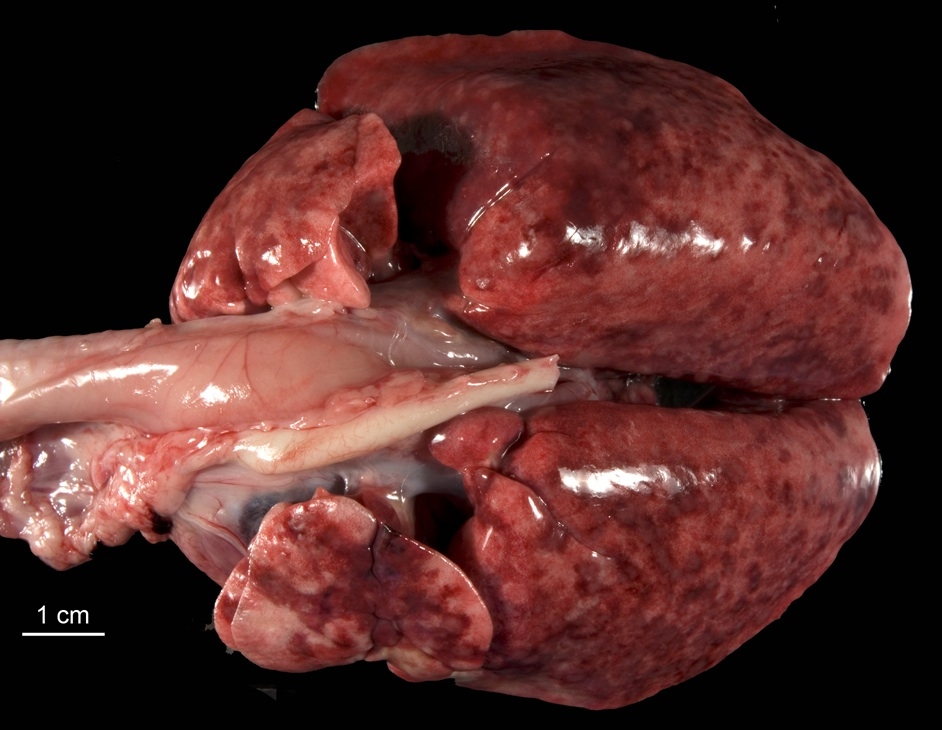
Gross lesions were found in the brain and thoracic cavity. A friable, dark-brown to tan, 1.3 cm in
diameter, roughly spherical mass extended from the dorsal aspect of the right frontal lobe of the cerebrum into the
thalamus. The thymus was atrophied. Both lungs were over-expanded, firm, and mottled tan to red-brown.
Indistinct, pale gray to tan, coalescing nodules, 1 to 3 mm in diameter, were evident in cross-section.
Histopathologic Description:
The submitted sections of lung are representative of the pulmonary lesions.
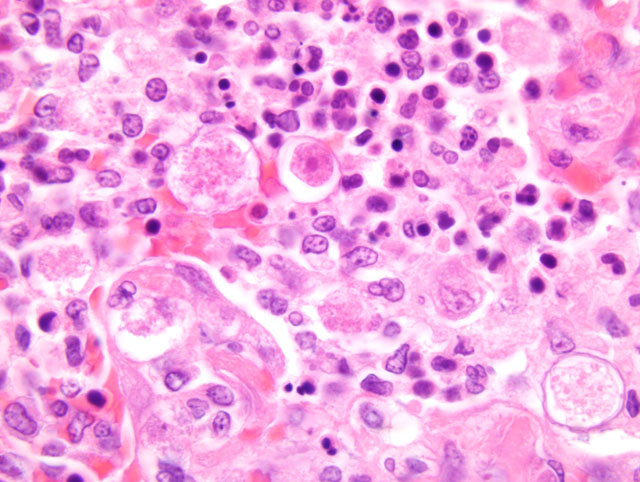
Individual and coalescing, poorly demarcated nodules (up to 2 mm in diameter) of granulomatous inflammation
were scattered through the pulmonary parenchyma. The centers of the nodules had undergone amorphous to
fibrinoid necrosis with scanty hemorrhage. Alveolar spaces in necrotic (central) and viable peripheral zones of the
nodules were partially filled with neutrophils, macrophages, fibrin, and numerous amoebic trophozoites with fewer
cysts. Trophozoites were spherical to ovoid, ranged from 7 to 19 μm in maximal dimension (mean, 14 μm), and had
a large nucleus with a prominent karyosome and abundant vacuolated cytoplasm. Encysted amoebae had
amphophilic cyst walls, 1-2 μm in thickness, with space between the exocyst and endocyst.
Where granulomatous inflammation extended to the visceral pleura, overlying mesothelial cells were hypertrophied.
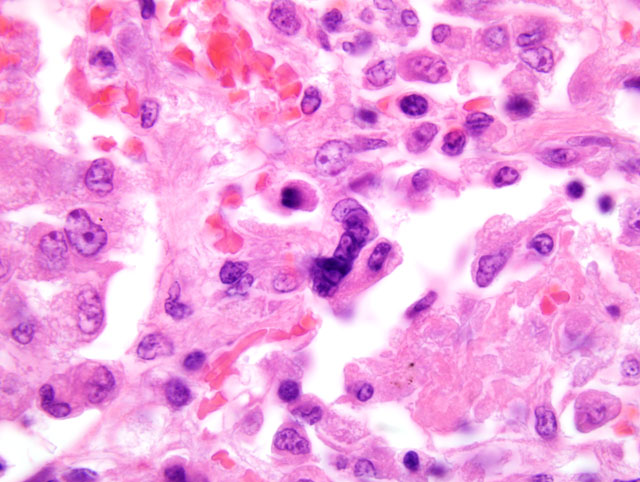
Between nodules of granulomatous inflammation, the lung was congested and edematous with increased numbers of
alveolar macrophages. Eosinophilic inclusions were observed in the cytoplasm (and less commonly in the nucleus)
of bronchiolar epithelial cells and alveolar macrophages; these were labeled immunohistochemically with antibody
to canine distemper virus. In other sections (not a feature in the submitted section), sloughed bronchiolar epithelial
cells had large amphophilic intranuclear inclusions that were labeled immunohistochemically with antibody to
canine adenovirus.
Morphologic Diagnosis:
1. Multifocal granulomatous pneumonia with intralesional amebic
trophozoites and cysts.
2. Histiocytic alveolitis with eosinophilic cytoplasmic and intranuclear inclusions.
Lab Results:
Frozen lung specimens were submitted to Dr. Visvesvara at the Centers for Disease Control
and Prevention (CDC) for identification of the amoebae. The lung was positive by immunofluorescence and realtime
PCR for
Acanthamoeba spp., and negative by both tests for
Balamuthia mandrillaris and for
Naegleria fowleri.
Pseudomonas aeruginosa and
Escherichia coli were isolated from the lung by bacterial culture. The lung and spleen
were positive by immunofluorescence for canine distemper virus, and negative for canine adenovirus, herpesvirus,
and parvovirus. No virus was isolated from the lung, spleen or brain.
Condition:
Canine distemper; Acanthamoeba sp.
Contributor Comment:
Canine distemper viral infection may have resulted in immunosuppression that
predisposed this puppy to other infections. Acanthamoebiasis was considered the most important of these and the
cause for granulomatous encephalitis and pneumonia. Amoebae were not detected in tissues other than brain and
lung. However, the puppy also had histologic evidence of pulmonary infection with canine adenovirus and oral
candidiasis.
Acanthamoeba, Balamuthia, and
Naegleria species are the free-living amoebae that have been associated with
encephalitis and disseminated infection in dogs and humans.(2) In histologic sections, recognition of nuclear
features, such as the prominent karyosome and lack of peripheral chromatin, is useful in distinguishing amebic
trophozoites from macrophages. In this case, the presence of cysts in addition to trophozoites in infected tissues
tended to eliminate
Naegleria from the differential diagnosis, but definitive diagnosis of
Acanthamoeba infection
was based on immunofluorescence and PCR results (performed on lung specimens). Some reported canine cases of
acanthamoebiasis have had granulomatous encephalitis and pneumonia (1), and it has been proposed that pulmonary
infection is the result of inhalation or aspiration of the organism from water with hematogenous extension to the
brain. However, amebic encephalitis has also been recognized in a dog with widely disseminated acanthamoebiasis
in which no organisms were detected in the lungs.(2)
JPC Diagnosis:
1. Lung: Pneumonia, pyogranulomatous, multifocal, severe, with necrosis and many amebic
trophozoites and few amebic cysts.
2. Lung: Pneumonia, bronchointerstitial, diffuse, moderate, with alveolar histiocytosis, type II pneumocyte
hyperplasia, viral syncytia, and few bronchiolar and histiocytic intranuclear and intracytoplasmic viral inclusion
bodies.
Conference Comment:
Like several others evaluated in recent WSC sessions, this case demonstrates the
importance of searching for an underlying cause of immunosuppression upon the identification of an opportunistic
pathogen. In this case, the extensive pyogranulomatous nodules in the lung were clearly evident to conference
participants as the predominant lesion. Following description of the most striking lesions, the conference moderator
encouraged participants to carefully examine the remainder of the lung; closer examination, beyond cursory
subgross perusal, reveals diffuse bronchointerstitial pneumonia attributed to canine distemper virus infection. Slide
variation is present, and the characteristic intranuclear and intracytoplasmic eosinophilic viral inclusions are rare in
some sections, underscoring the utility of molecular diagnostics (e.g. immunohistochemistry) as employed in this
case. Additional microscopic findings noted by conference participants include multifocal hypertrophy of the
pleural mesothelium, and in some sections, small aggregates of extracellular and intrahistiocytic coccobacilli
(consistent with the bacterial culture results reported by the contributor).
Conference attendees compared and contrasted cases I and II of this conference, and continued the discussion of
pathogenic free-living amoebae, as summarized in the conference comment for case I. This case differs from case I
by the presence of numerous cyst forms, the walls of which are Periodic Acid-Schiff (PAS)-positive. Additionally,
systemic acanthamoebiasis is associated almost exclusively with immunosuppression, as noted above, whereas
balamuthiasis occurs in both immunocompromised and immunocompetent hosts. Primary amebic encephalitis
caused by
Naegleria fowleri is not associated with immunocompromise. Interestingly,
Acanthamoeba keratitis is a
disease of immunocompetent humans associated with corneal trauma or improper contact lens hygiene, and carries a
far better prognosis than does
Acanthamoeba encephalitis.(3)
We thank Dr. Christopher Gardiner, Consulting Parasitologist for the AFIPs Department of Veterinary Pathology,
for reviewing this case.
References:
1. Bauer RW, Harrison LR, Watson CW, Styer EL, Chapman WL: Isolation of
Acanthamoeba sp. from a
greyhound with pneumonia and granulomatous amebic encephalitis. J Vet Diagn Invest
5:386-391, 1993
2. Dubey JP, Benson JE, Blakely KT, Booton GC, Visvesvara GS: Disseminated
Acanthamoeba sp. infection in a
dog. Vet Parasitol
128:183-187, 2005
3. Schuster FL, Visvesvara GS: Free-living amoebae as opportunistic and non-opportunistic pathogens of humans
and animals. Int J Parasitol
34:1001-1027, 2004