Signalment:
5 year old, ovariohyster-ectomized
female Cavalier King Charles spaniel,
Canis familiaris.An adult spayed female Cavalier King Charles Spaniel reported to be 5 years old from a the household with multiple dogs exhibiting increased respiratory rate and cough. During thoracic radiographs the patient became agonal and arrested despite attempted cardiopulmonary resuscitation. Radiographs revealed pneumonia. The patient was presented for necropsy 1.5 hours following death.
Gross Description:
The trachea is diffusely mildly compressed dorsoventrally. Excluding a limited regional portion of the cranial aspect of the left cranial lung lobe, the lungs are diffusely dark red, heavy, and sink in formalin. The heart is mildly
enlarged with mild nodular thickening of the mitral valve. A small portion of the cerebellar vermis protrudes through a mildly narrowed and irregular foramen magnum.
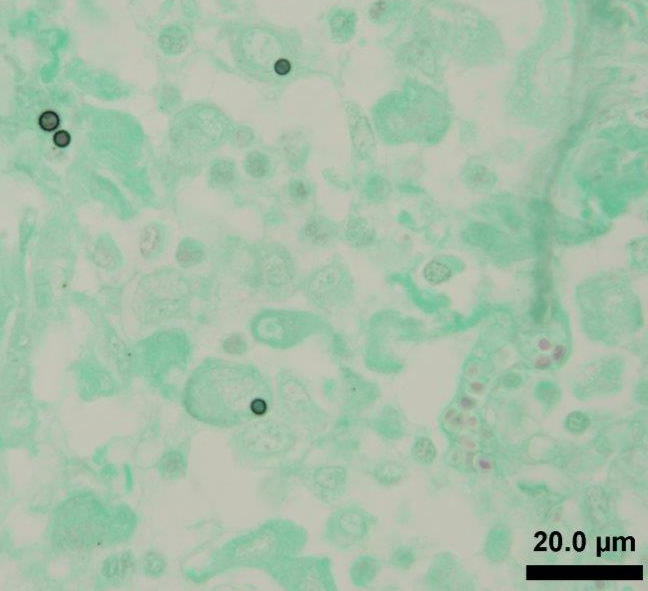
Histopathologic Description:
Lung:
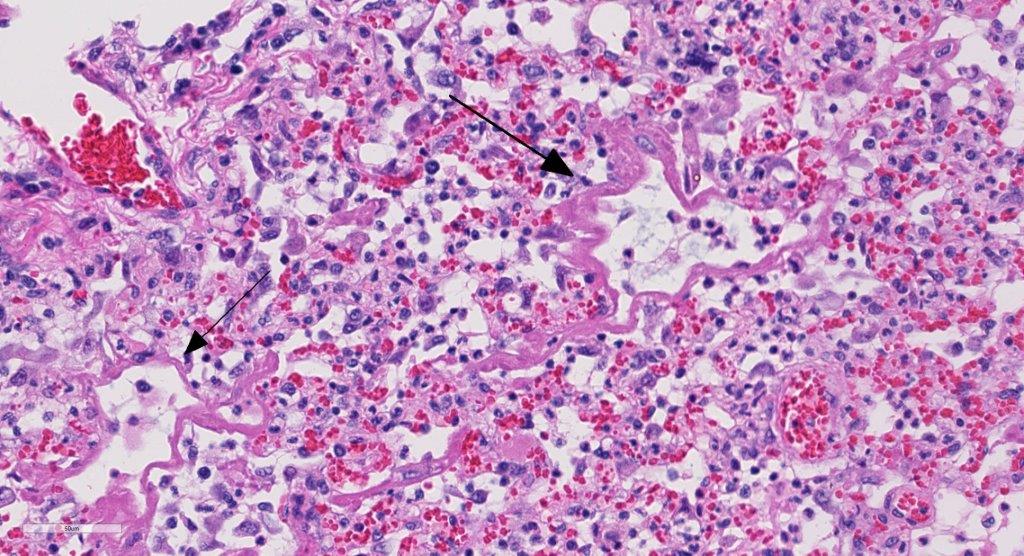
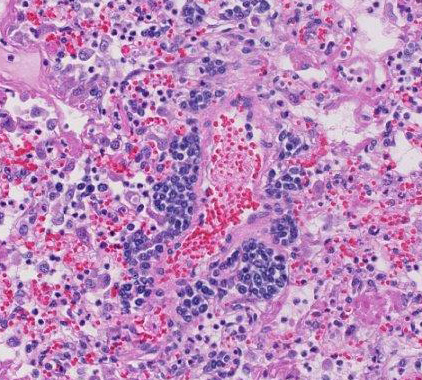
Alveoli and bronchioles frequently are filled with macrophages, neutrophils,
cellular debris, erythrocytes, multinucleate giant cells and edema. The
macrophages are frequently vesciulate, occasionally containing cellular and
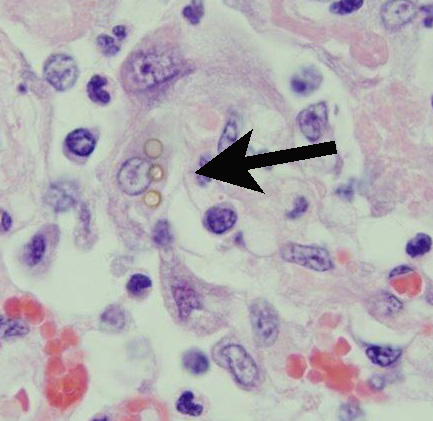
karyorrhectic debris. Random,
individual to multiple, golden-brown, 3-5Μm diameter fungal spores are present
both within macrophages and free within the alveolar lumens. Alveolar septa are
occasionally lined by finely fibrillar to glassy eosinophilic material (hyalin
mem-branes). Alveolar epithelial cells are multifocally cuboidal (type II
pneumocyte hyperplasia) with rare binucleate and multinucleate cells. There is
multifocal loss of the bronchiolar epithelium, with in-frequent epithelial
dysplasia. A few peri-bronchiolar macrophages contain black granular pigment
(anthracosis). Moderate numbers of plasma cells with fewer lymp-hocytes
surround pulmonary vessels. Vascular endothelial cells are frequently
hypertrophic. Pleural mesothelial cells are hypertrophic.
Morphologic Diagnosis:
Marked, diffuse, histiocytic interstitial pneumonia with fungal spores.
Lab Results:
Microbiology
Sample: Lung
Test: Aerobic Culture
Result: No growth seen in 3 days
PCR/DNA sequencing
99 % nucleotide identity with
Lycoperdon pyriforme (AY854075.1, strain AFTOL-ID48) over 682 bp.
Condition:
Lycoperdon spp. fungal pneumonia
Contributor Comment:
The intrahistiocytic and free
fungal spores within the lung were identified as spores of puffball mushrooms
by PCR and DNA sequencing. Eleven species of puffball mushrooms (
Lycoperdon
sp.) are indigenous to the area from which this case originated (greater
Smoky Mountains). Inhalation of spores has been reported infrequently in
association with pneumonia (pulmonary lyco-perdonosis) in dogs
1,8 and
humans
7,9.
JPC Diagnosis:
Lung:
Pneumonia, interstitial, necrotizing, fibrinosuppurative, and histiocytic,
diffuse, chronic, severe with hyaline membrane formation and rare intra-histiocytic
fungal spores.
Conference Comment:
Pulmonary lesions caused by the
inhalation of
Lycoperdon sp. spores are theorized to result from the
host hypersensitivity response to fungal spores acting as foreign bodies rather
than an infectious process.
2,7 The failure to culture
Lycoperdon
from a reported case and inability of this saprophytic fungus to germinate
under normal physiologic body temperatures supports this theory.
7
Hyp-ersensitivity pneumonitis (HP), also known as extrinsic allergic
alveolitis, is caused by intense and often prolonged exposure to inhaled
organic antigens such as fungal spores, but can also include bacterial products
and animal proteins. This disease is commonly diagnosed in dairy cows and
horses housed indoors and usually affect multiple animals within a group. It
results from chronic inhalation of spores of thermophilic actinomycetes (
Saccharo-physpora
rectivirgula) found in moldy hay.
5,6 This is followed by an
antibody response to inhaled antigen and local deposition of antigen-antibody
complexes (Arthus reaction) as well as the formation of multifocal granulomas
suggesting a T-cell-mediated response. This type of reaction suggests both type
III and type IV hypersensitivity response
2,5,7 (See WSC 2012-2013,
conference 22, case 4
for a review of hypersensitivity reactions).
Characteristic lesions in animal and human
HP include proliferation of type II pneumocytes and fibrosis of alveolar septa
and peribronchiolar tissue. In severe cases, hyaline membranes composed of
fibrin, serum proteins, and cell debris line the alveolar septa.
5
This hyaline membrane causes alveolar occlusion resulting in severe hypoxia and
death.
Conference
participants noted the severe necrotizing and inflammatory changes to the lung
interstitium, but had difficult time finding the rare fungal spores present in
the tissue. Interestingly, clinical disease of lycoperdonosis in humans and
dogs only occurs following a massive inhalation dose of puffball conidia
7;
however in this section there is a paucity of spores present. As a result,
there was discussion of differentials for hyaline membrane formation, which is
the most striking histologic feature in this case. Acute respiratory distress
syndrome (ARDS) in the dog was discussed as having a similar histopathologic
appearance to this case. In addition to hyaline membranes, type II pneumocyte
hyperplasia, and bronchiolar epithelial necrosis is a consistent feature of
ARDS. Inciting causes include: trauma, shock, disseminated intravascular coag-ulation,
septicemia, smoke inhalation, oxygen toxicity, viral infection, and
strangulation among others.
4
This
case was additionally studied in consultation with the Department of Pulmonary
and Mediastinal Pathology at the Joint Pathology Center, who disagreed with the
purported mechanisms discussed above, based on the morphologic changes noted in
this section. Their interpretation is that this section of lung is diagnostic
for diffuse alveolar damage (DAD) due to a toxic reaction to
Lycoperdon
spores rather than HP, published in a case report from this animal.
2 DAD
is the most commonly identified form of interstitial lung disease and is the
histologic correlate to the clinical condition of ARDS, discussed by conference
participants.
4 The pulmonary pathologists describe an intra-alveolar
inflammatory infiltrate composed predominantly of neutrophils and macrophages.
This is admixed with focal plasma cells and lymphocytes, hyaline membranes, and
acute inflammation of bronchiolar epithelium, along with multifocal fungal
spores. They see no histologic evidence to support the diagnosis of HP. They
also note the importance of recognizing this entity as DAD, and not HP, due to
the high mortality rate for DAD, even with intensive supportive treatment.
References:
1. Aleghat T,
Kellett-Gregory L, Van Winkle T: Lycoperdonosis in a dog,
Vet Pathol.
2009;46:1046.
2.
Alenghat T, Pillitteri C et al. Lycoperdonosis in two dogs.
J Vet Diagn
Invest. 2010;22:1002-1005.
3.
Buckeridge D, Torrance A, Daly M. Puffball mushroom toxicosis (lycoperd-onosis)
in a two-year-old dachshund.
Vet Rec. 2011;168:304.
4. Caswell J, Williams K. Respiratory
system. In: Maxie MG, ed.
Jubb, Kennedy, and Palmers Pathology of Domestic
Animals. Vol 2. 6
th ed. Philadelphia, PA: Elsevier Saunders;
2016: 514.
5. Husain
AN. The Lung. In: Kumar V, Abbas AK, Aster JC, eds.
Pathologic Basis of
Disease. 9th ed. Philadelphia, PA: Elsevier Saunders; 2015:694-695.
6. Lopez
A. Respiratory system, media-stinum, and pleurae. In: Zachary JF,
McGavin MD, eds.
Pathologic Basis of Veterinary Disease. 5th
ed. St. Louis, MO: Elsevier; 2012:513.
7. Munson
E, Panko D, Fink, J. Lycoperdonosis: Report of Two Cases and Discussion of the
Disease:
Clin Microbiol Newsl. 1997;19(3):17-20.
8.
Rubensohn M: Inhalation pneumonitis in a dog from spores of puffball mushrooms.
Can Vet Journal. 2009;50(1):93.
9. Taft T,
Cardillo R et al. Respiratory illness associated with inhalation of mushroom
spores-Wisconsin, 1994.
MMWR Morb Mortal Wkly Rep. 1994;43(29):525-526.