Joint Pathology Center
Veterinary Pathology Services
Wednesday Slide Conference
2018-2019
Conference 18
30 January, 2019
CASE IV: BA214/11 B1 (JPC 4035109)
Signalment: 2 year old, male entire, Pogona vitticeps, bearded dragon
History: An outbreak of fungal dermatitis occurred in a colony of bearded dragons. Seven responded successfully and six were euthanized with follow-up 3 months later apparently showing complete resolution of the disease in all animals. Several months later, this animal was presented with new skin lesions on the dorsum, left stifle and left hock which were treated with both topical preparations (F10, iodine and clotrimazole) and systemic antifungals (itraconazole). The animal’s condition failed to respond to therapy and due to continued deterioration (including development of coelomic distension) it was euthanized in March 2011.
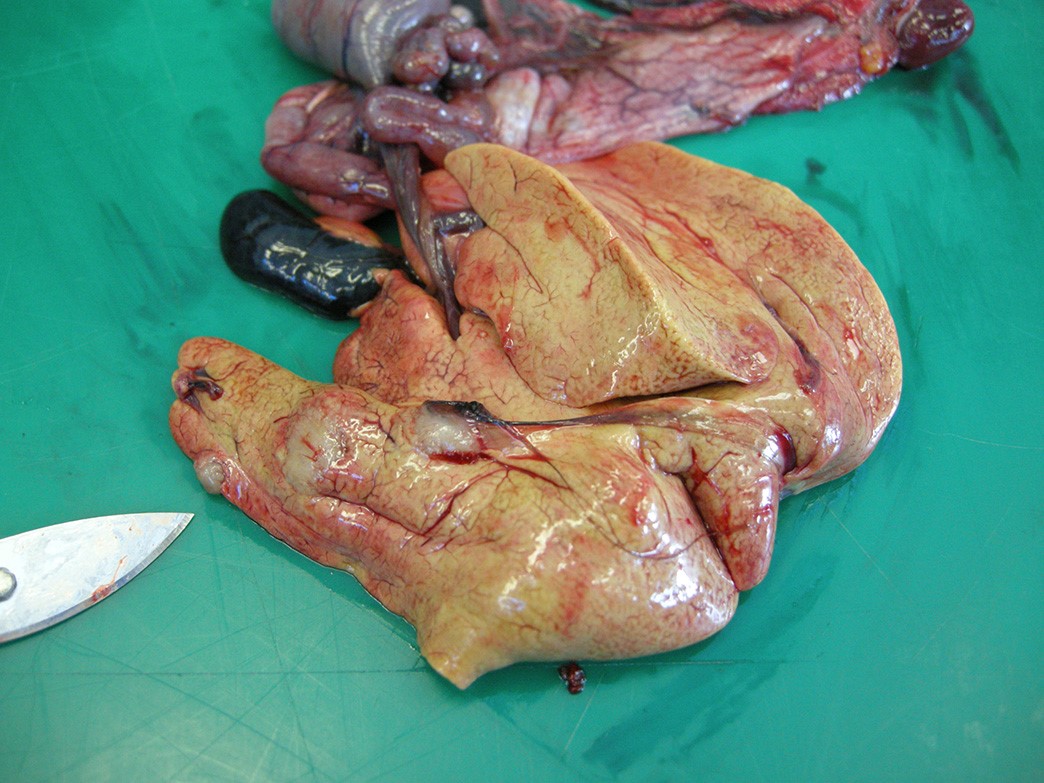
Gross Pathology: An adult (2 year old), male entire, bearded dragon, weighing 553g was presented for necropsy examination. On the midline of the dorsum, there was a large (1.27cm x 1.5cm), focal, well demarcated, raised, black, irregularly rounded midline plaque. There were similar smaller lesions, which were discoloured dark yellow to brown, located over the right elbow (1cm diameter), left elbow (8mm diameter) the lateral aspect of the right knee (5mm diameter), the plantar surface of the left foot (8mm), and the plantar surface of the right foot (1mm). The liver was mottled pink to yellow to red with three, well-demarcated, firm, raised, pale yellow, irregularly rounded nodules ranging from 8-20mm in diameter. One nodule was incised and contained yellow to brown, granular material. There was a similar 5mm diameter lesion present within the omentum, near the spleen.
Laboratory Results: 3 months prior to euthanasia blood was evaluated to assess liver function and white cell count while on treatment. Total protein=74 g/l (Range=52-72); AST=103 U/l (Range=0-80); CK=5830 (Range=0-550); Leukocytes =0.4 cells x109/L (Range=1.5-8.5). The owner was given a guarded prognosis due to the low leukocyte count. Cultures submitted previously from this colony from similar lesions were identified as Chrysosporium anamorph of Nannizziopsis vriesii (CANV).
Microscopic Description:
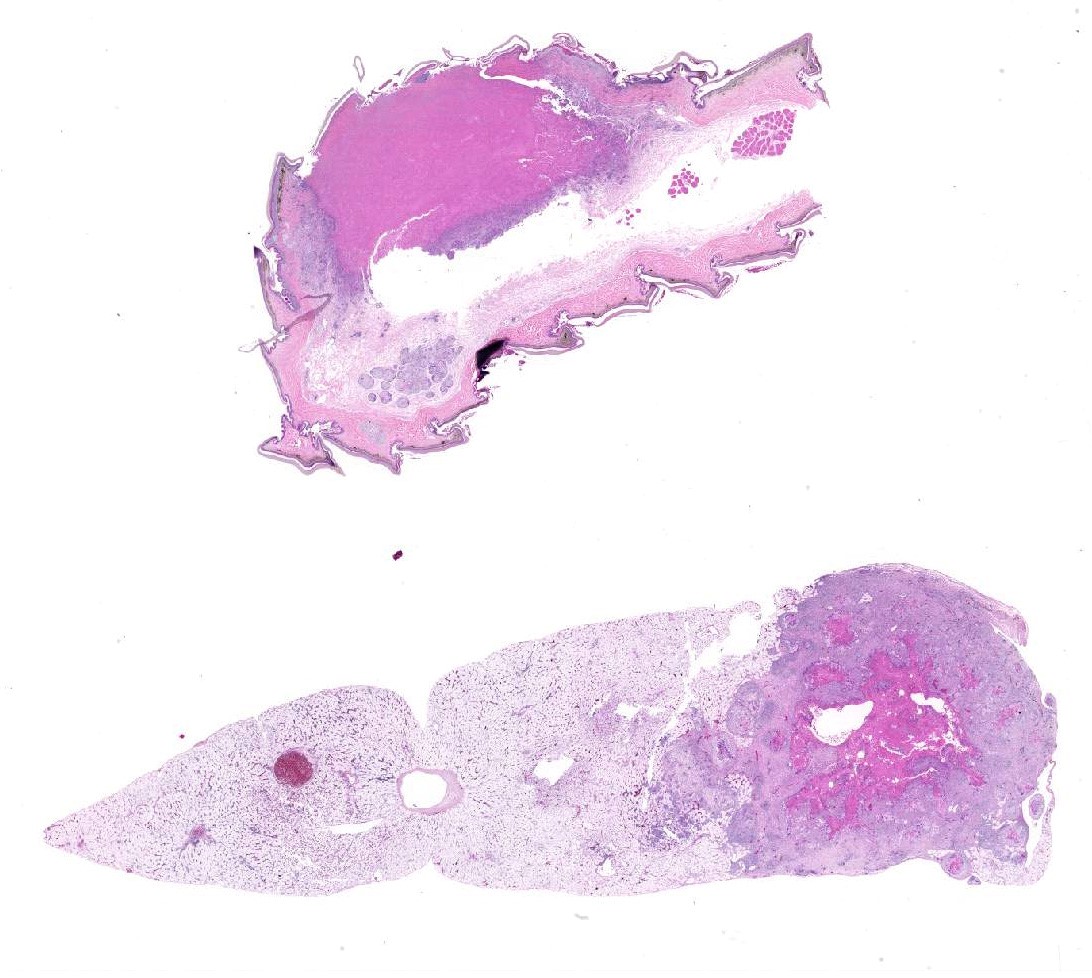
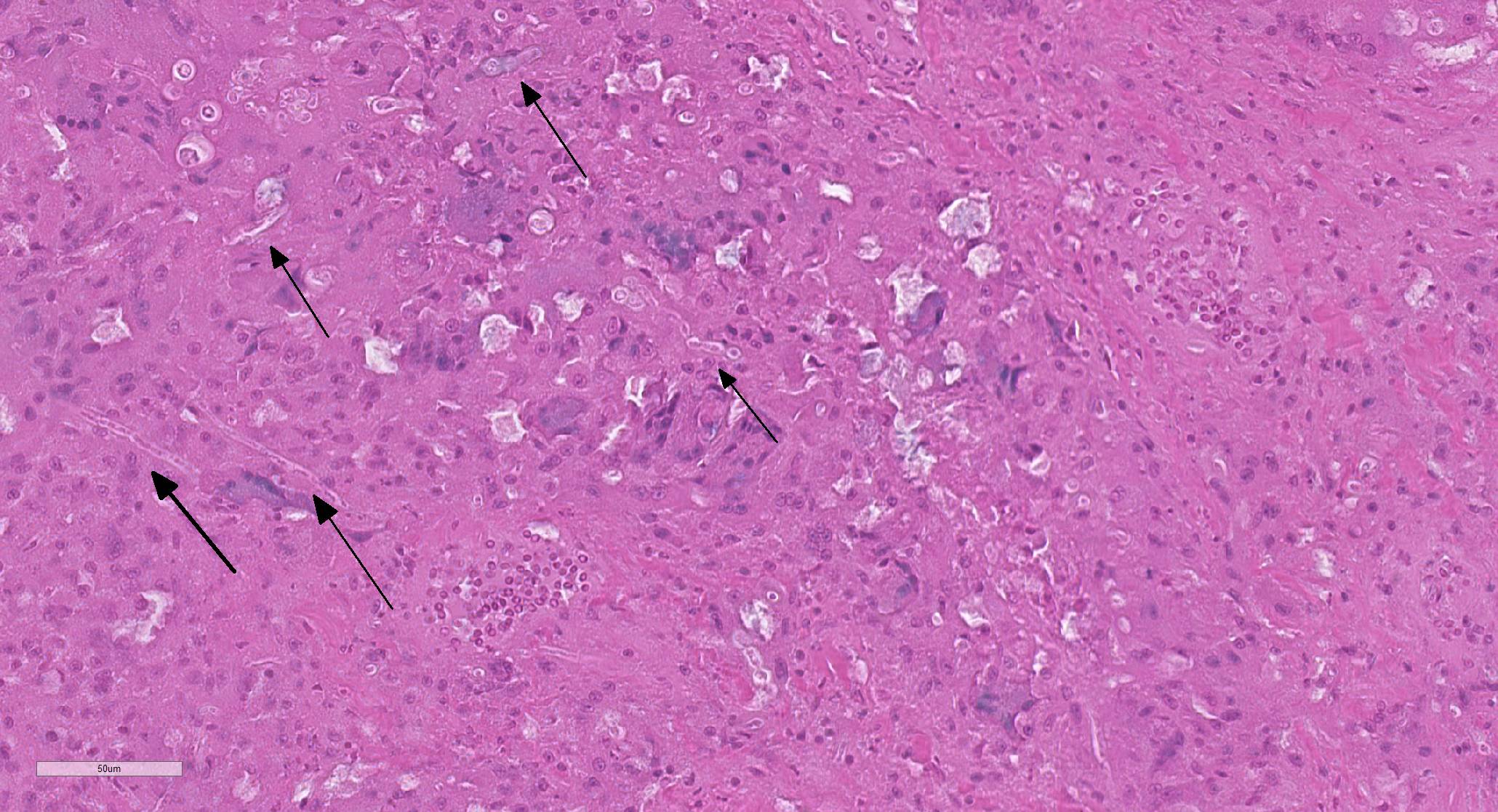
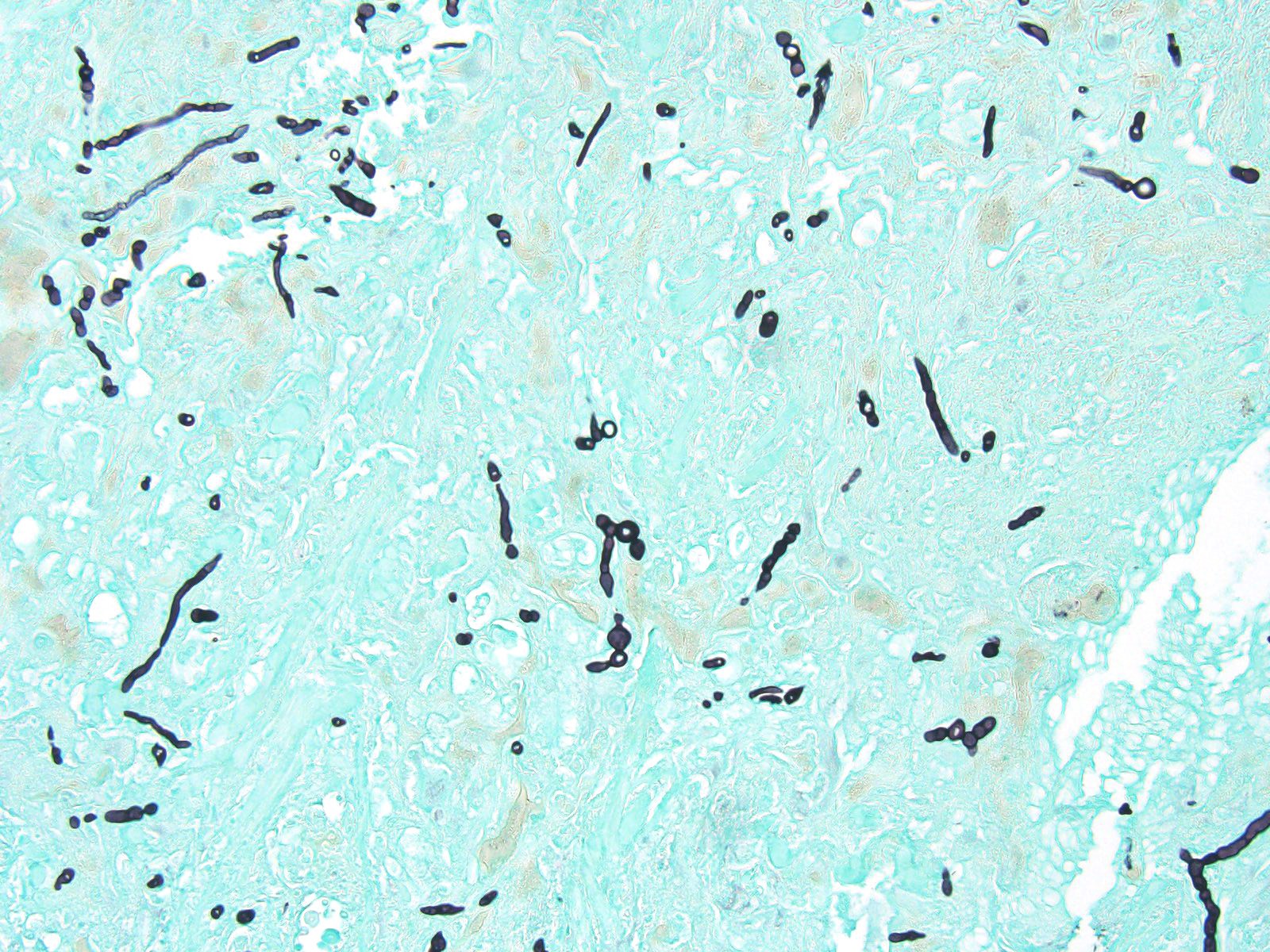
Elbow skin (1 section) –There is full thickness loss of cellular and nuclear detail extending into the dermis (necrosis). In this area, multifocal remnants of the overlying stratum corneum are present and there are scattered subcorneal accumulations of palely basophilic 1um round to oval organisms (yeasts-presumptive). Multifocally throughout the necrotic tissue there are negatively stained outlines of fungal hyphae (approx 2-4um diameter with occasional septae, branching and terminal buds) with parallel walls and occasional segmentation. At the edges of the necrosis there are large numbers of epithelioid macrophages with multinucleated giant cells, heterophils and scattered lymphocytes with mild to moderate vascular congestion. Scattered haemorrhage is present. Smaller granulomas which are centrally necrotic and contain fungal organisms (as described above) are present around the edges of the necrotic region. Intact skin is mildly to moderately hyperplastic with multifocal, mild heterophilic and mononuclear inflammation within the superficial dermis.
Liver– There is a focal, large centrally necrotic area composed within the hepatic parenchyma which contains fungal organisms as previously described. Histiocytic, lymphocytic and heterophilic inflammation is present around the periphery with moderate to large numbers of multinucleated giant cells with scattered admixed fibroblasts. Multifocal, small granulomas are present around the periphery of the large necrotic lesion. The surrounding hepatocytes have marked, diffuse, swelling with large numbers of small to moderately sized, well demarcated, clear intracytoplasmic vacuoles (consistent with lipid) which often displace the nucleus to the periphery.
Contributor’s Morphologic Diagnoses:
- Severe, granulomatous and necrotising, ulcerative dermatomycosis and cellulitis with intra-lesional fungal organisms – elbow skin
- Multifocal, severe, granulomatous hepatitis with intra-lesional fungal organisms – liver
- Diffuse, moderate, hepatic lipidosis – liver
NOTE: Similar inflammation was present in sections from the dorsal skin lesion, lung and omental nodule (not included in the submission). Given the previous isolation of CANV in this colony/animal, the progression of the disease and the histological features of the sections this was considered to be the most likely diagnosis.
Contributor’s Comment: Saprophytic fungi are often implicated in dermatomycoses of snakes and lizards and infections are often associated with poor husbandry, poor nutrition or immunosuppression.8,11 Chrysosporium species may be isolated from normal reptile skin but the most clinically important (and rarely isolated) is considered to be Chrysosporium anamorph of Nannizziopsis vriesii (CANV).8,11 This is a keratinophilic ascomycetous fungus reported to be the primary pathogen of “yellow fungus disease”, an emerging disease in reptiles. 1-12 CANV is a non-pigmented fungus (hyalohyphomycosis). CANV has been reported in a number of chameleon species, geckos, bearded dragons, leopard geckos, a girdled lizard, several snake species and saltwater crocodiles, among others.1-16 CANV f is difficult to identify on culture and may be mistaken for any of the following: Trichophyton, Geotrichum, Malbranchea and Trichosporon.6,12 Generally more than one animal is infected in an outbreak (suggesting that it is contagious) especially in bearded dragons. In this species it causes granulomatous dermatitis which often becomes systemic1,4,5, 9. This presents as multifocal, yellow-brown discoloured lesions on the body with crusting and/or hyperkeratotic plaques. These lesions tend to develop necrosis, leading to sloughing of the skin and ulceration. This is followed by invasion of the musculature and bones and may progress to lethal systemic infection.
Conference participants discussed the orthokeratotic hyperkeratosis and elevation of the scale overlying the granulomas as potentially a localized form of dysecdysis.
Contributing Institution:
Royal (Dick) School of Veterinary Studies
Easter Bush Campus
Roslin, Midlothian
EH25 9RG
UK.
JPC Diagnosis:
- Scaled skin: Dermatitis, granulomatous and necrotizing multifocal to coalescing, severe, with numerous intra- and extracellular fungal hyphae and epidermal arthroconidia
- Liver: Hepatitis, granulomatous, focally extensive, severe, with numerous intra- and extracellular fungal hyphae.
- Liver, hepatocytes: Lipidosis, diffuse, severe.
JPC Comment: Since the submission of this case in 2013, much has been written about the Chrysosporium anamorph of Nannizziopsis vriesii (CANV) and related pathogens (referred to as Chrysosporium anamorph of Nannizziopsis vriesii complex (CANVC) which cause disease in reptiles. Anamorphs, most often seen in the Ascomycota, are asexual reproductive states (i.e, these fungi do not produce a fruiting body).
CANV is a keratophilic species that lives primarily on fragments of feathers and hair in the soil. While most cases of CANV have been identified in reptiles, related species of Chrysosporium infection have been documented in other species – C. pannicola (dog, horse, human), C. tropicum (chickens), and C. keratinophilum and C. zonatum (human).5
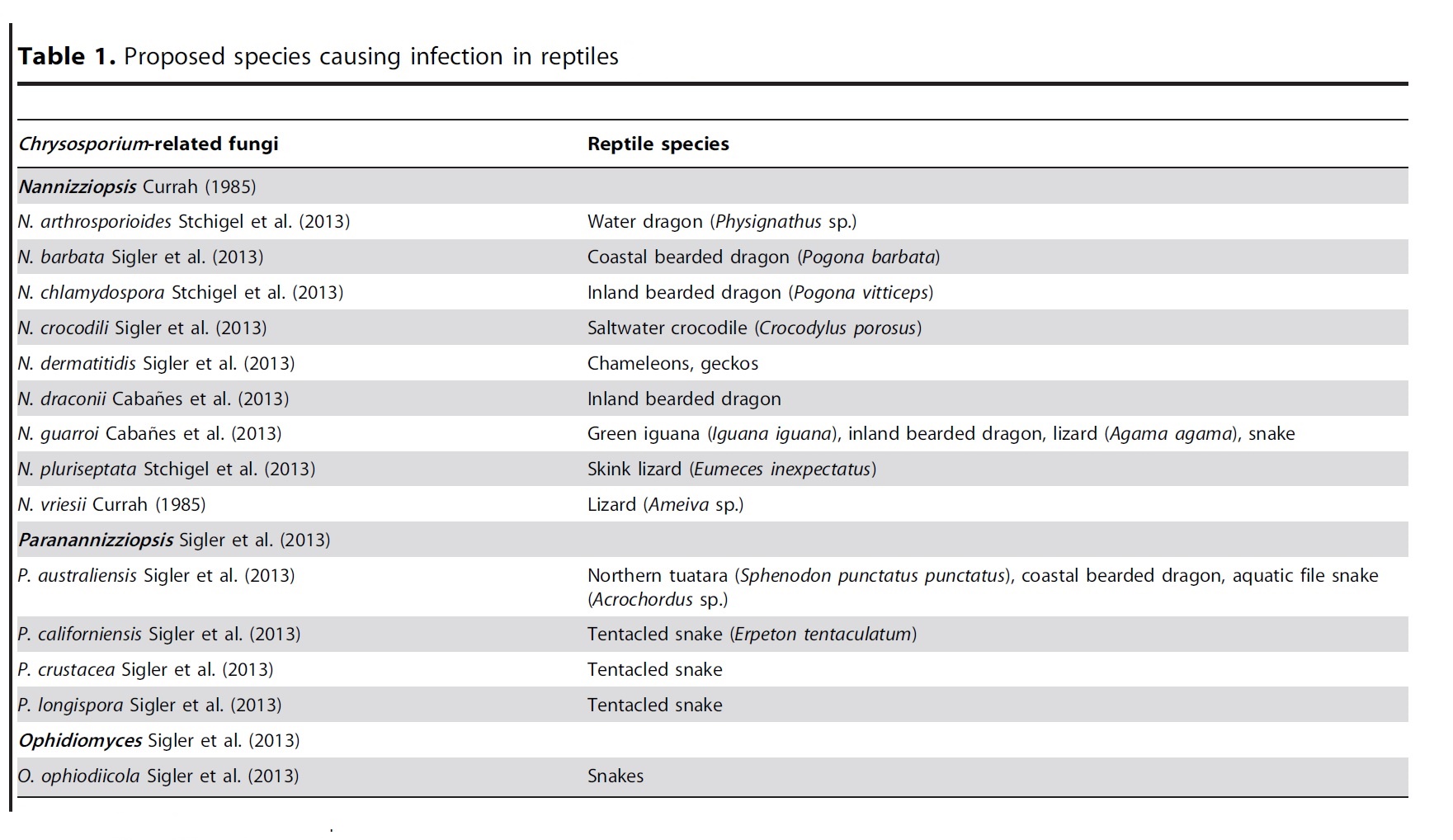
In 2013, Stchigel et al.14 and Sigler et al.15 published a taxonomic revision of Chrysosporium- related fungi and their likely host specificity (Table 1).5 In most hosts, the lesion begins in the skin, with dermal and subcutaneous heterophilic granulomas which eventually extend into underlying bone, muscle, and disseminate to viscera, as seen in this case.16 It is difficult, if not impossible, to differentiate members of the CANV in tissue section.5
A related fungus, Ophidomyces ophidiicola, which causes similar lesions in snakes was reassigned from the CANVC in 2013 to its own genus.15 A case of this was published in the WSC earlier this year (Conference 8, Case 4).
Conference participants discussed the uniqueness of the epidermal lesion in this case. While granuloma formation is the rule in this infection, some participants interpreted with large area of necrosis within the dermis extending to the overlying epidermis as an infarct, and one participant saw hyphae involving a cutaneous vessel. The moderate commented that this was a unique presentation in his experience. Conference participants also discussed the orthokeratotic hyperkeratosis and elevation of the scalse overlying the granulomas as potentially a localized form of dysecdysis.
References:
- Abarca LM, Martorell J, Castella G, Ramis A & Cabanes EJ. Dermatomycosis in a pet inland bearded dragon (Pogona vitticeps) caused by a Chrysosporium species related to Nannizziopsis vriesii. Veterinary Dermatology 2009; 20, 295-299
- Abarca ML, Martorell J, Castellá G, Ramis A & Cabanes EJ. Cutaneous hyalohyphomycosis caused by a Chrysosporium species related to Nannizziopsis vriesii in two green iguanas (Iguana iguana). Medical Mycology 2008; 46: 349–54.
- Bertlesen MF, Crawshaw GJ, Sigler L, Smith DA. Fatal cutaneous mycosis in tentacled snakes (Erpeton tentaculatum) caused by the Chrysosporium anamorph of Nannizziopsis vriesii. J Zoo Wildl Med 2005; 36, 82-87
- Bowman MR, Paré JA, Sigler L, Naeser JP, Sladky KK, Hanley CS, Helmer P, Phillips LA, Browers A & Porter R. Deep fungal dermatitis in three inland bearded dragons (Pogona vitticeps) caused by the Chrysosporium anamorph of Nannizziopsis vriesii. Medical Mycology 2007; 45, 371-376
- Cabanes FJ, Sutton DA, Guarro J. Chrysosporium-related fungi and reptiles: a fatal attraction. PLOS Pathogens, 2014; 10(10): 31004367.
- Hedley J, Eatwell K, Hume L. Necrotising fungal dermatitis in a group of bearded dragons (Pogona vitticeps). Veterinary Record 2010; 166, 464-465
- Hellebuyck T, Baert K, Pasmans F, Van Waeyenberghe L, Beernaert L, Chiers K, De Backer P, Haesebrouck F & Martel A. Cutaneous hyalohyphomycosis in a girdled lizard (Cordylus giganteus) caused by the Chrysosporium anamorph of Nannizziopsis vriesii and successful treatment with voriconaxole. Veterinary Dermatology 2010; 21, 429-433
- Jacobson ER, Cheatwood JL, Maxwell LK. Mycotic diseases of reptiles. Seminars in Avian and Exotic Pet Medicine 2000; 9: 94–101
- Johnson RSP, Sangster CR, Sigler L, Hambleton S & Paré JA. Deep fungal dermatitis caused by the Chrysosporium anamorph of Nannizziopsis vriesii in captive coastal bearded dragons (Pogona barbata). Australian Veterinary Journal 2011; 12; 515-519.
- Lorch JM, Knowles S, Lankton JS, Michell K, Edwards JL, Kapfer JM, Staffen RA, Wild ER, Schmidt KZ, Ballmann AE, Blodgett D, Farrell RM, Bloriosos BM, Last LA, Price SJ, Schular KL, Smith CE, Wellehan JFX, Blehert DS. Snake fungal disease; an emerging threat to wild snakes. Phil Trans R Soc B 2015; 371:20150457.
- Paré JA, Coyle KA, Sigler L, Maas AK III & Mitchell RL. Pathogenicity of the Chrysosporium anamorph of Nannizziopsis vriesii for veiled chameleons (Chamaeleo calyptratus). Medical mycology 2006; 44:25-31
- Paré JA, Sigler L, Rosenthal KL et al. Microbiology. Fungal and bacterial diseases of reptiles. In: Mader DR, ed. Reptile Medicine and Surgery, 2nd ed. St Louis, MO: Saunders Elsevier, 2006: 217–38
- Paré JA, Sigler L, Hunter DB, Summerhall RC, Smith DA & Machin KL. Cutaneous mycoses in chameleons caused by the Chrysosporium anamorph of Nannizziopsis vriesii (Apinis) Currah. J Zoo Wildl Med 1997;28:443-453
- Stchigel AM, Sutton DA, Cano-Lira JF, Cabanes FJ, Abarca L, Tintelneot K, Wickes BL, Garcia D, Guarro J.
- Sigler L, Hambieton S, Pare. Molecular characerization of reptile pathogens currently known as members of the Chrysoporium anamorph of Nannizziopsis vriesii complex and relationship with some human-associated isolates. J Clin Microbiology 2013: 51(10)3338-3357.
- Toplon DE, Terrell SP, Sigler L, Jacobson ER. Dermatitis and cellulitis in leopard geckos cause by the Chyrsosporium anamorph of Nannizziopsis vriesii. Vet Pathol 2012; 50(4): 585-589.