Signalment:
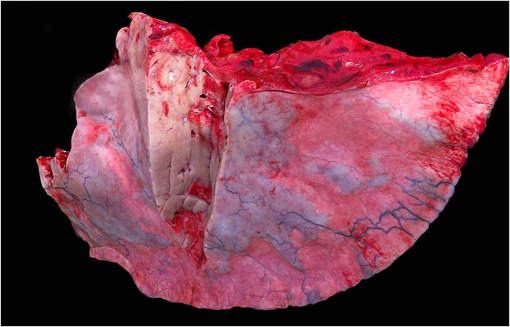
Gross Description:
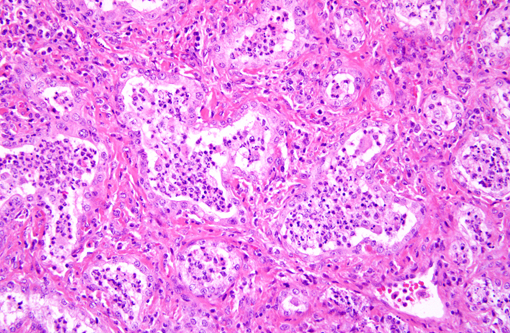
Histopathologic Description:
Morphologic Diagnosis:
Lab Results:
Condition:
Contributor Comment:
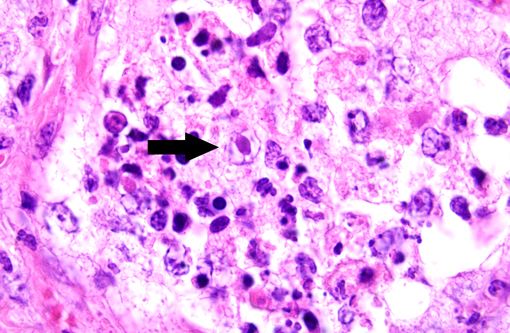
Histologically, well-organized, mature fibrous tissue expand the interalveolar septa with preservation of alveolar architecture.(1) Lymphocytes infiltrate the fibrotic interstitium. Alveolar spaces are lined by cuboidal cells, and contain neutrophils and macrophages. A few of the alveolar macrophages have eosinophilic intranuclear inclusions.
Multinodular pulmonary fibrosis is histologically distinct from the pulmonary interstitial fibrosis of silicate pneumoconiosis, which is associated with granulomatous inflammation, and from idiopathic pulmonary fibrosis, which more commonly affects foals than adults and is attributed to diffuse alveolar damage. In the second report, equine multinodular pulmonary fibrosis was tentatively diagnosed antemortem, as in this case, on the basis of clinical presentation and radiologic findings (nodular pulmonary interstitial pattern) with supportive PCR detection of EHV-5 from bronchoalveolar lavage specimens. Inappetance, weight loss, fever, cough, and respiratory distress were common to all 5 cases in that study.(2)
To date, Kochs postulates have not been fulfilled to establish EHV-5 as the definitive cause of equine multinodular pulmonary fibrosis. However, the virus is consistently associated with this unique pulmonary lesion, so pathologic findings of nodular pulmonary fibrosis with herpetiform inclusions and supportive PCR analysis should prompt consideration of EMPF in adult horses with respiratory distress and nodular interstitial pneumonia.
JPC Diagnosis:
Conference Comment:
An interstitial pneumonia of donkeys has been reported which is associated with asinine herpesvirus.(1) This disease differs from EMPF in that it is a diffuse inflammatory disease with syncytial cell formation without viral inclusions; interstitial fibrosis is considered a secondary component.
Conference participants noted that pleural arteries were often hypertrophied and surrounded by abundant collagen. This is likely due to increased intrapulmonary blood pressure due to the diffuse fibrosis, which inhibits adequate blood flow through large portions of the affected lung.
Conference participants also discussed a differential diagnosis that included that paraquat and diquat toxicosis, which causes fulminant pulmonary fibrosis, although due to the dwindling availability of these compounds, this differential is becoming exceedingly rare. Another possibility is exercise-induced pulmonary hemorrhage, which also has large areas of pulmonary fibrosis, but is characterized by numerous hemosiderophages, and lacks intranuclear inclusion bodies.
Alveolitis, a term commonly used in human respiratory pathology, was used in our morphologic diagnosis because of the striking inflammation centered on alveolar lumens as separate from the fibrosing interstitial pneumonia, which is characteristic of EMPF. This histologic finding is expected with interstitial pneumonias in which there is abundant protein exudation, as well as viral-induced leukocyte chemotaxis. A common feature of EMPF is the preservation of an alveolar-like architecture(2), which are often filled with neutrophils and macrophages; hence the designation of alveolitis in addition to fibrosing interstitial pneumonia.
References:
2. Williams KJ, Maes R, Del Piero F, Lim A, Wise A, Bolin DC, Caswell J, Jackson C, Robinson NE, Derksen F, Scott MA, Uhal BD, Li X, Youssef SA, Bolin SR: Equine multinodular pulmonary fibrosis: a newly recognized herpesvirus-associated fibrotic lung disease. Vet Pathol 44:849-862, 2007
3. Wong DM, Belgrave RL, Williams KJ, Del Piero F, Alcott CJ, Bolin SR, Marr CM, Nolen-Walston R, Myers RK, Wilkins PA: Multinodular pulmonary fibrosis in five horses. J Am Vet Med Assoc 232:898-905, 2008