Signalment:
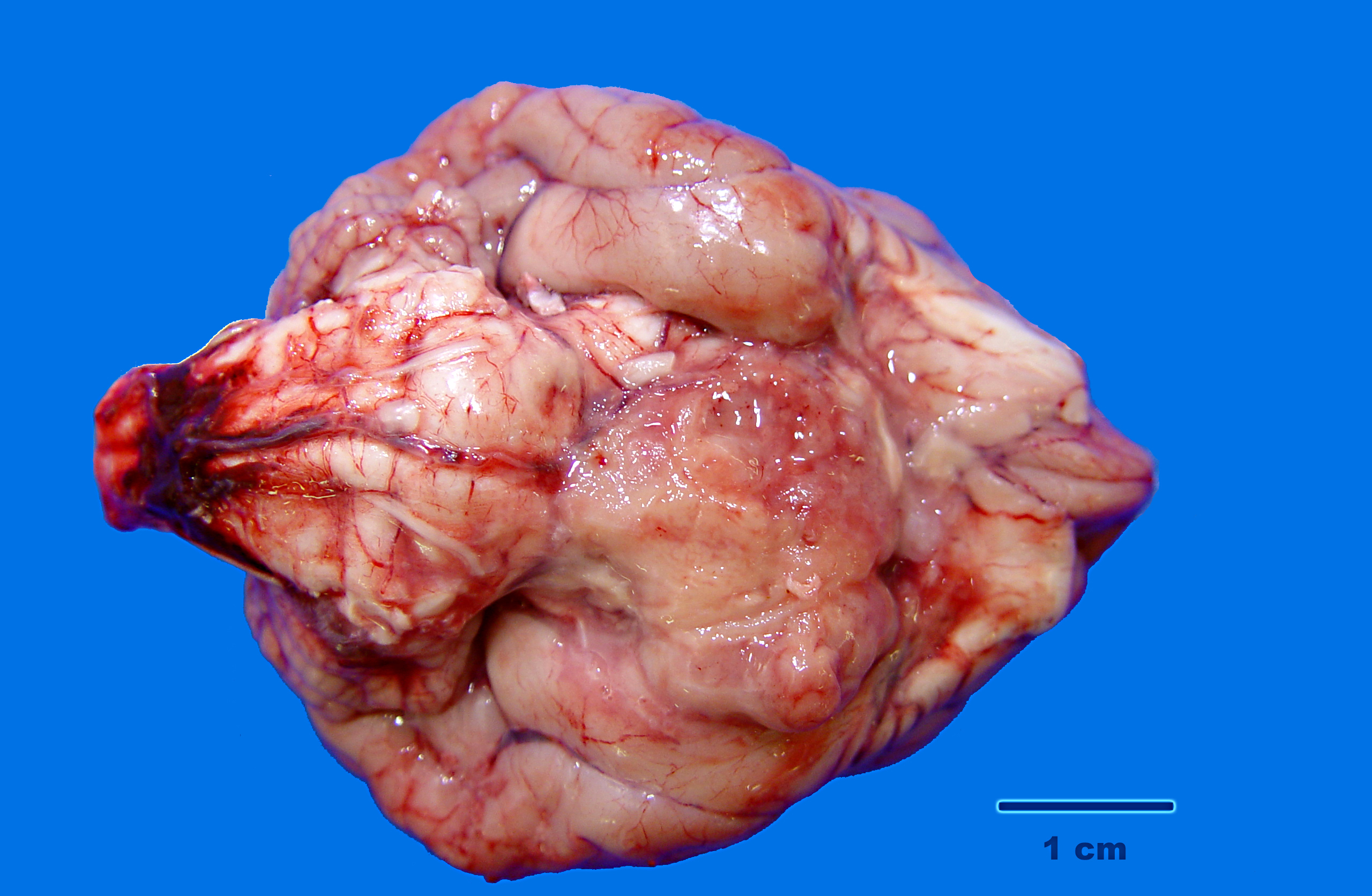
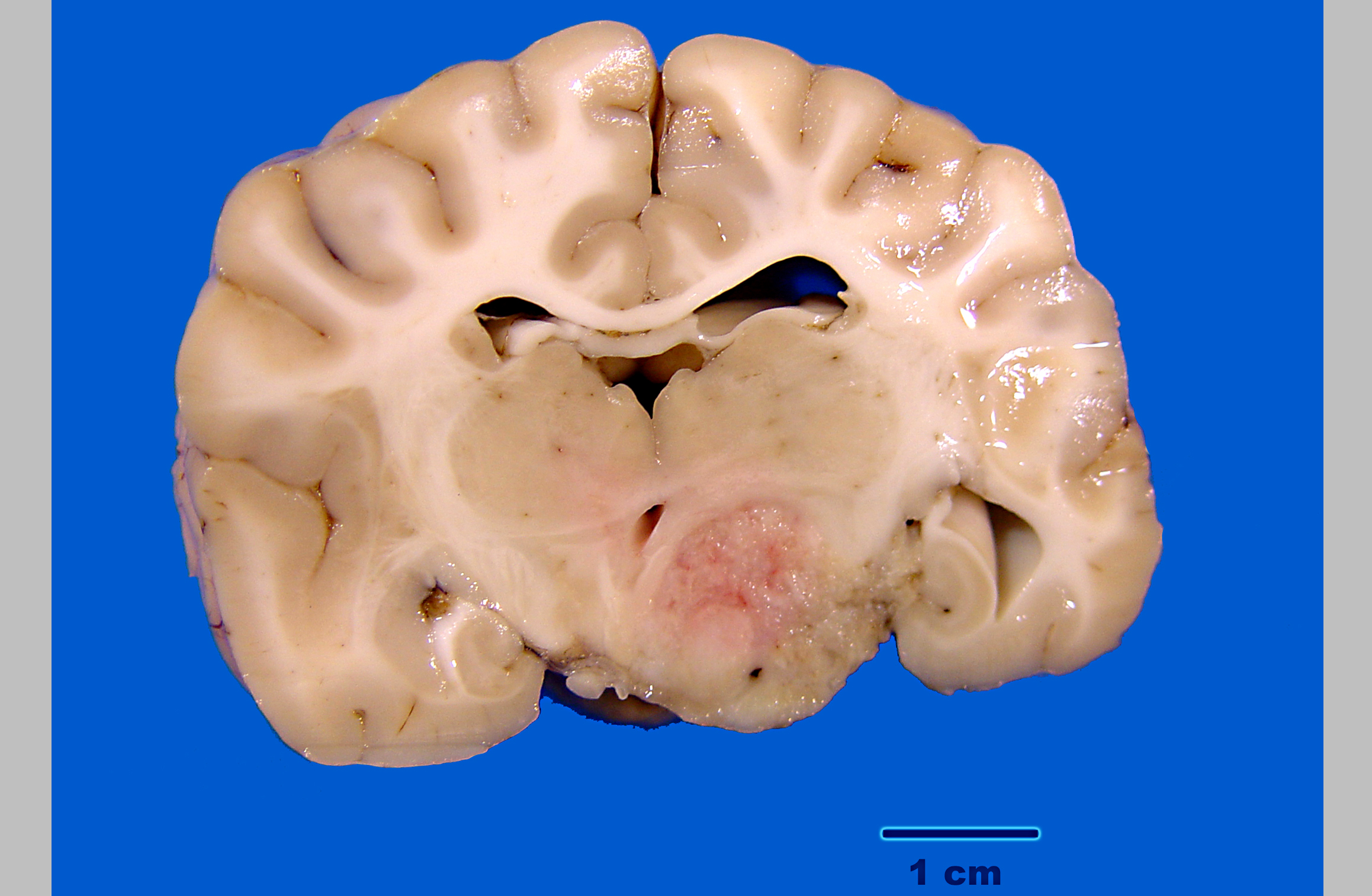
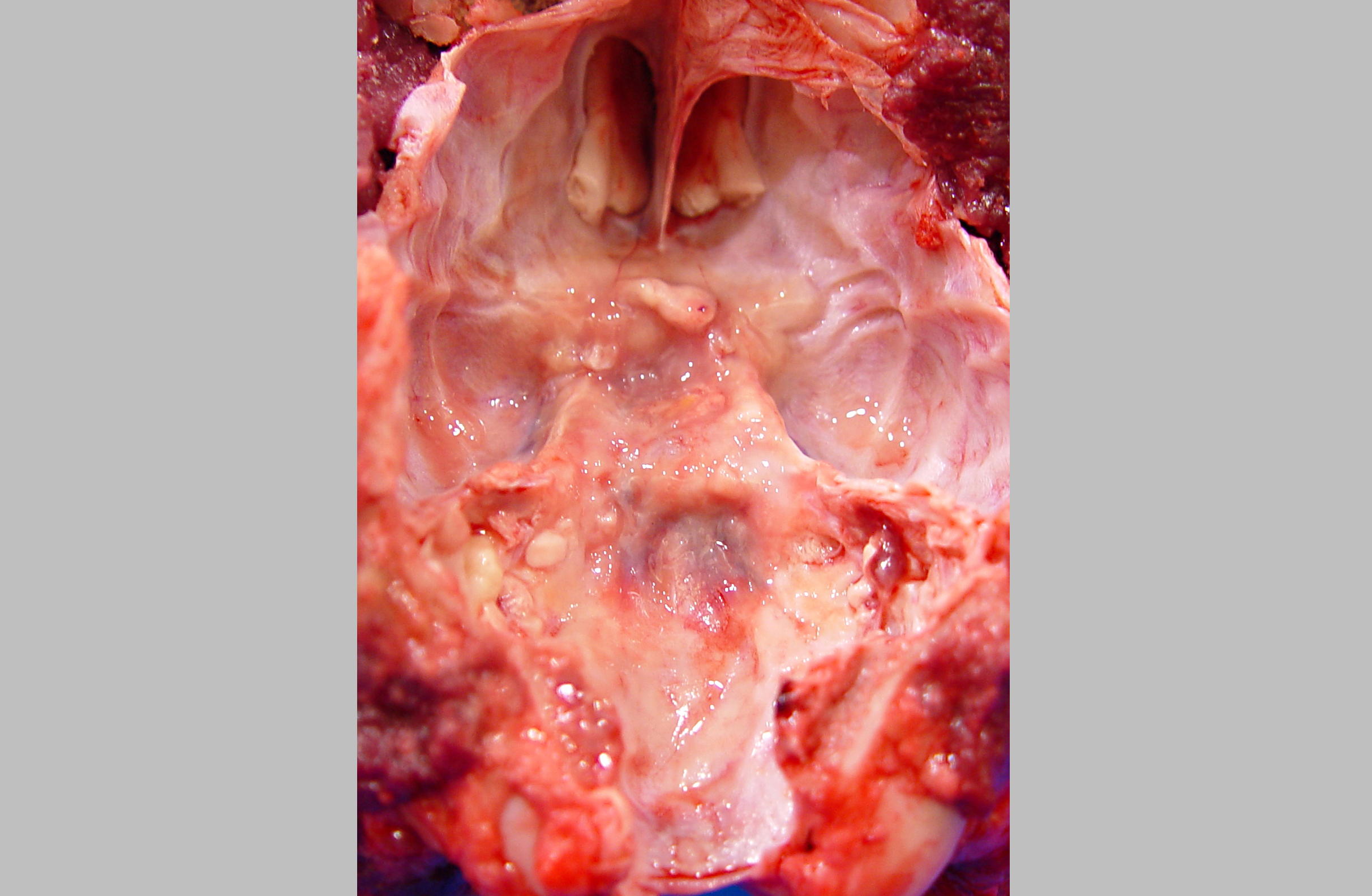
Gross Description:
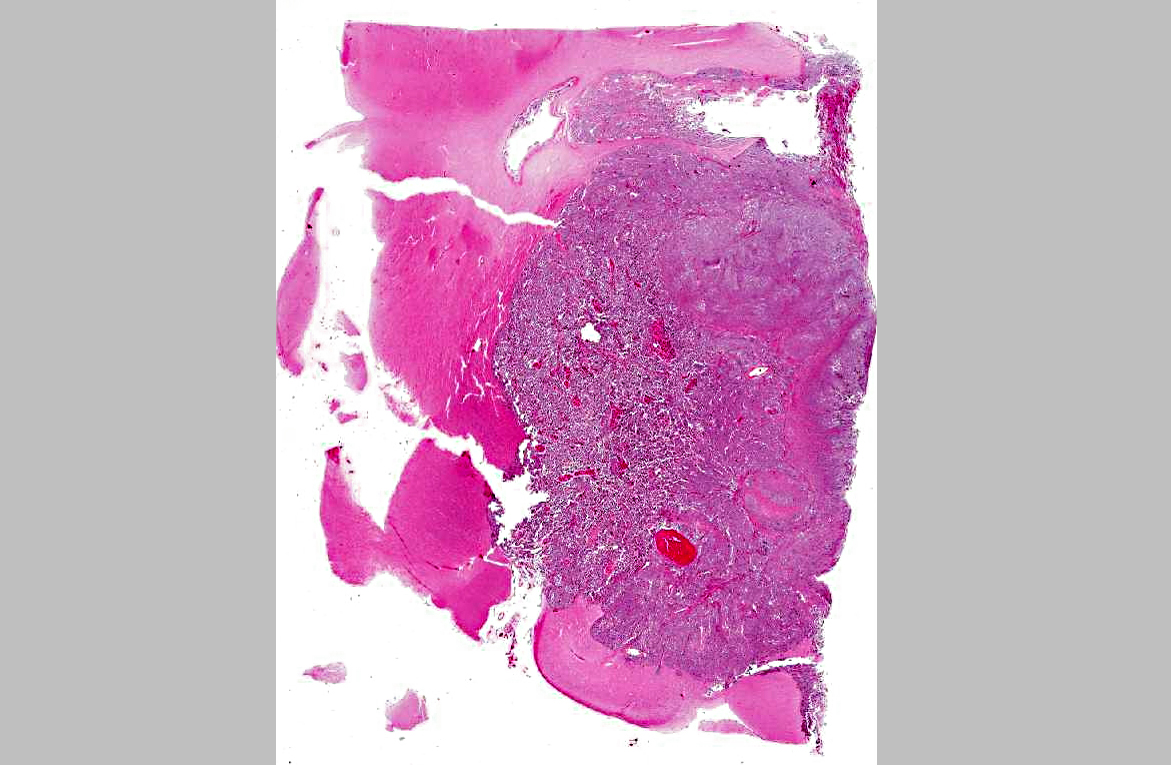
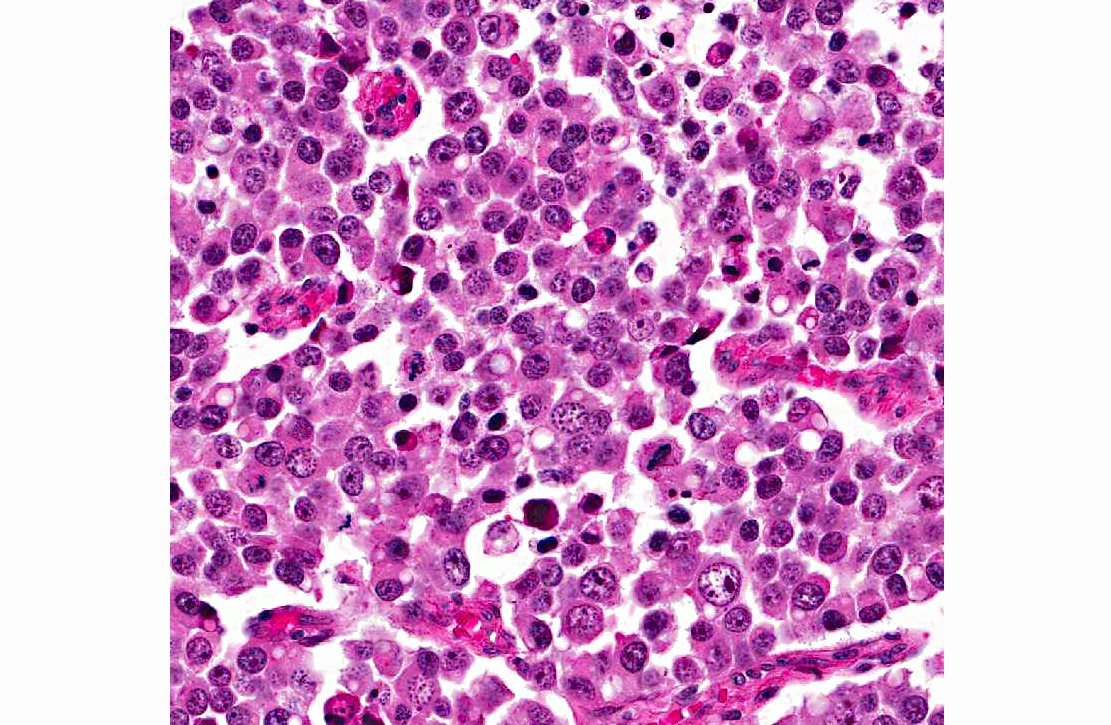
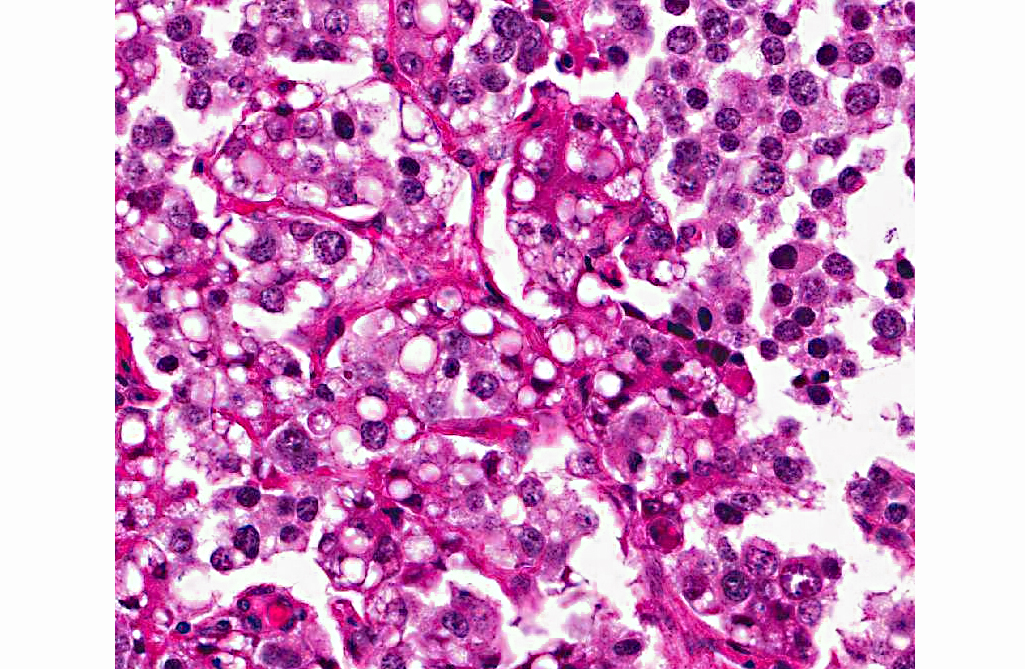
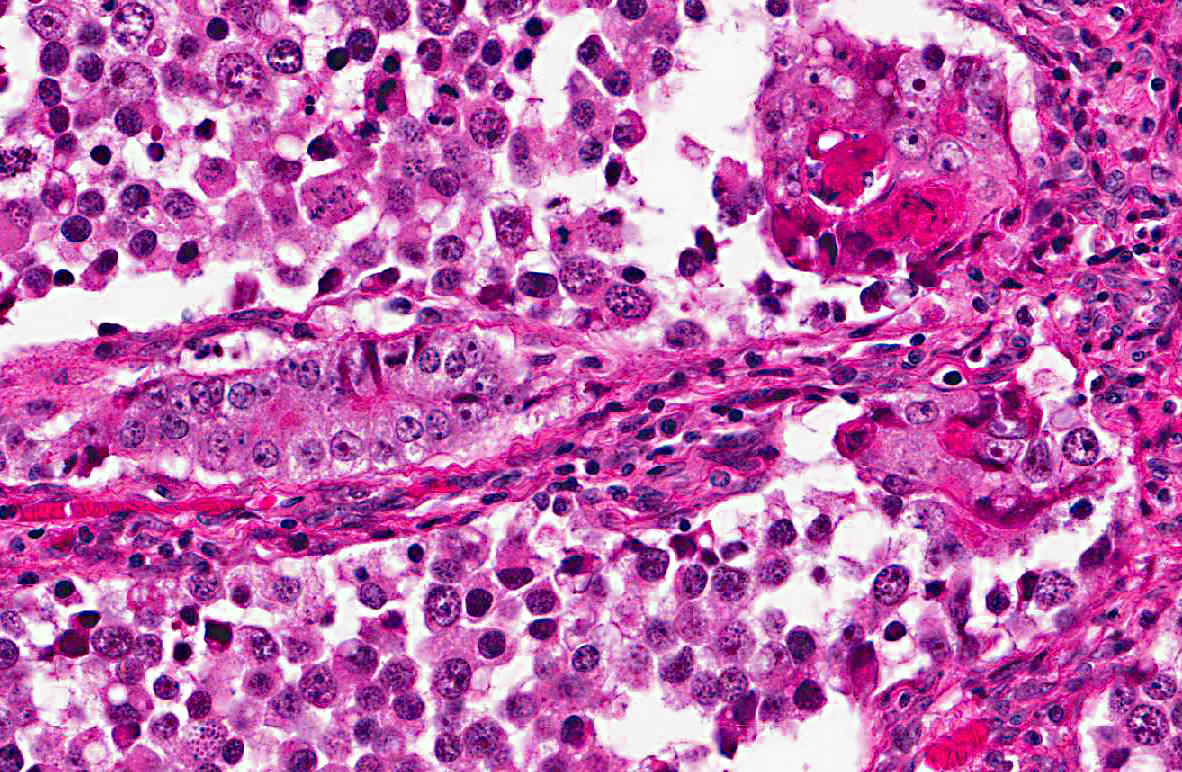
Histopathologic Description:
Morphologic Diagnosis:
Lab Results:
Condition:
Contributor Comment:
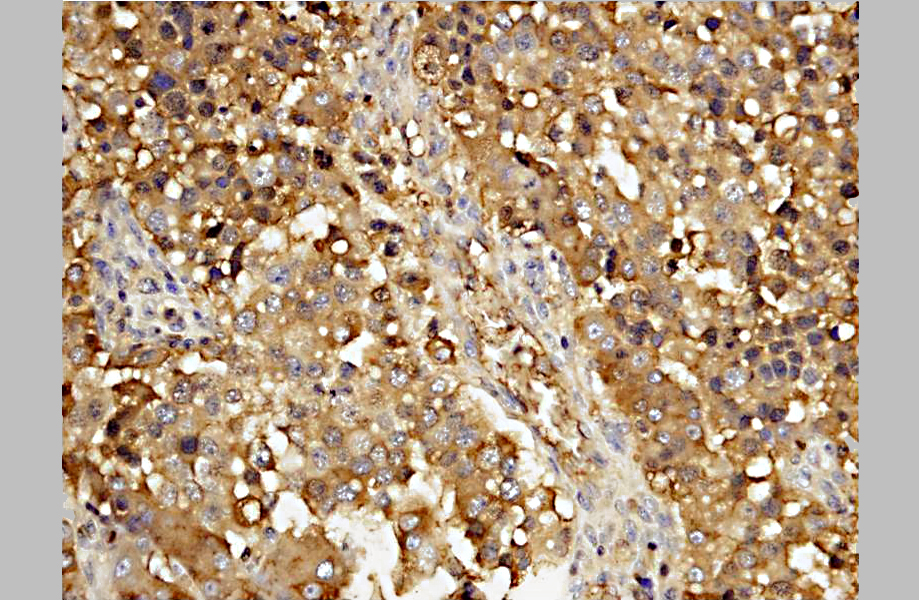
A diagnosis of suprasellar germ cell tumor was made based on three criteria: 1) midline suprasellar location, 2) presence within the tumor of several distinct cell types; one population resembling seminoma or dysgerminoma and others suggesting teratomatous differentiation into secretory glandular and squamous elements, and 3) positive staining for alphafetoprotein.(14) Alpha-fetoprotein is a commonly used marker for germ cells in human and has been shown to be a useful marker for canine intracranial germ cell tumors.(14)
Germ cell tumors most commonly occur in the ovaries and testes (dysgerminomas and seminomas respectively). Extragonadal germ cell tumors, with similar histology, are reported less commonly and typically occur on midline, affecting the mediastinum, brain, and coccyx in humans.(13) In dogs, these rare tumors have been reported in the brain, the eye, and the spinal cord.(5,10,11,12,14) Intracranial germ cell tumors tend to localize in the pineal and suprasellar regions in humans, while the suprasellar region is preferentially affected in dogs.(14) Suprasellar germ cell tumors have also been described in three lake whitefish (Coregonus clupeaformis).(9)
Presenting clinical signs in affected dogs include visual disturbance, pupil abnormality, lethargy, anorexia, and less often, signs of pituitary dysfunction.(10,12,14) Typically young adult dogs are affected and there is a possible breed predilection for the Doberman Pinscher.(14)
The pathogenesis of these tumors remains poorly understood, with the main question being the origin of the germ cells in the brain.(6) During embryonic development, progenitor germ cells originate in the epiblast. These germ cells then migrate to the caudal yolk sac endoderm and reach the gonadal ridge via the dorsal mesentery of the hindgut. Extragonadal germ cell tumors are presumed to arise from ectopic embryonic germ cells which may become displaced during development, survive physiologic dissolution, and undergo neoplastic transformation.(3,6)
JPC Diagnosis:
Conference Comment:
Recently, a supracellar germ cell tumor was described in a 16-month-old Wagyu heifer calf adding to the list of species affected.(1) In the human literature, intracranial germ cell tumors have been divided into germinoma, teratoma, yolk sac tumor, embryonal carcinoma, and choriocarcinoma. Mixed germ cell tumors contain elements from more than one of these types.(2)
Conference participants noted there was significant slide variation, with some slides having polygonal neoplastic cells forming tubular structures, while others lacked these features. In addition, due to slide variation, the participants chose the generic location cerebrum for the morphologic diagnosis, as specimens varied in their anatomic location.
References:
2. Burger PC, Scheithauer BW. Tumors of the Central Nervous System.Third series, Fascicle 10. Washington, DC: Armed Forces Institute of Pathology/ American Registry of Pathology. 1993:251-257.
3. Echevarria ME, Fangusaro J, Goldman S. Pediatric central nervous system germ cell tumors: a review. The Oncologist. 13: 690 -699, 2008.
4. Evans HE. The skull. In: Evans HE, ed. Millers Anatomy of the Dog. Philadelphia, PA: WB Saunders Company; 1993:139.
5. Ferreira AJA, Peleteiro MC, Carvalho T, et al. Mixed germ cell tumor of the spinal cord in a young dog. J Small Anim Pract. 2003;44:81-84.
6. Hoei-Hansen CE, Sehested A, Juhler M, et al. New evidence for the origin of intracranial germ cell tumors from primordial germ cells: expression of pluripotency and cell differentiation markers. J Pathol. 2006;209:2533.
7. Hullinger RL. The endocrine system. In: Evans HE, ed. Millers Anatomy of the Dog. Philadelphia, PA: WB Saunders Company; 1993:139, 561.
8. Maxie MG, Youseff S. Neoplastic diseases of the nervous system. In: Maxie MG, ed. Jubb, Kennedy and Palmers Pathology of Domestic Animals. Vol. 1. 5th ed. Philadelphia, PA: Saunders Elsevier Limited; 2007:446-457.
9. Mikaelian I, Lapointe J-M , de Lafontaine Y, -+et al. Suprasellar germinoma in three lake whitefish (Coregonus clupeaformis). Acta Neuropathol. 2000;100: 228-232.
10. Nyska A, Harmelin A, Baneth G, et al. Suprasellar differentiated germ cell tumor in a male dog. J Vet Diagn Invest. 1993;5:462-467.
11. Patterson-Kane JC, Schulman FY, Santiago N, et al. Mixed germ cell tumor in the eye of a dog. Vet Pathol. 2001;38:712-714.
12. Rech RR, de Souza SF, da Silva MC, et al. Suprasellar germ cell tumor in a dog. Ciencia Rural. 2008;38:830-832.
13. Schneider DT, Schuster AE, Fritsch MK, et al. Multipoint imprinting analysis indicates a common precursor cell for gonadal and nongonadal pediatric germ cell tumors. Cancer Research. 2001;61:7268 7276.
14. Valentine BA, Summers BA, de Lahunta A, et al. Suprasellar germ cell tumors in the dog: a report of five cases and review of the literature. Acta Neuropathol. 1988;76:94-100.