Signalment:
Gross Description:
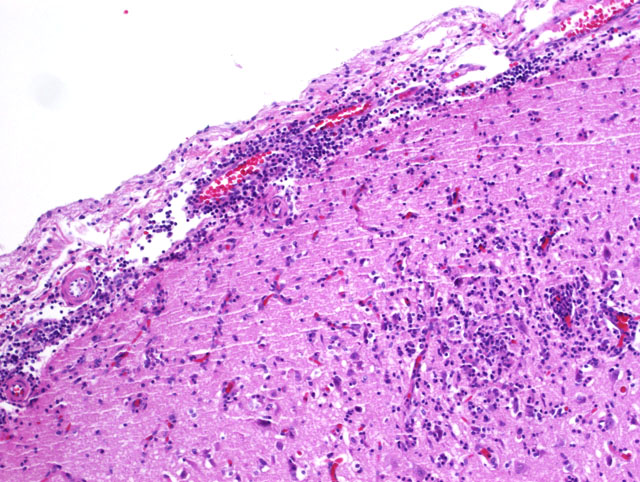
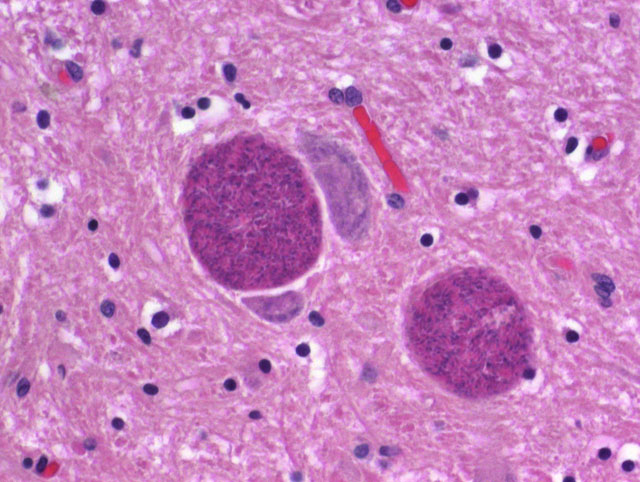
Histopathologic Description:
Morphologic Diagnosis:
Lab Results:
Condition:
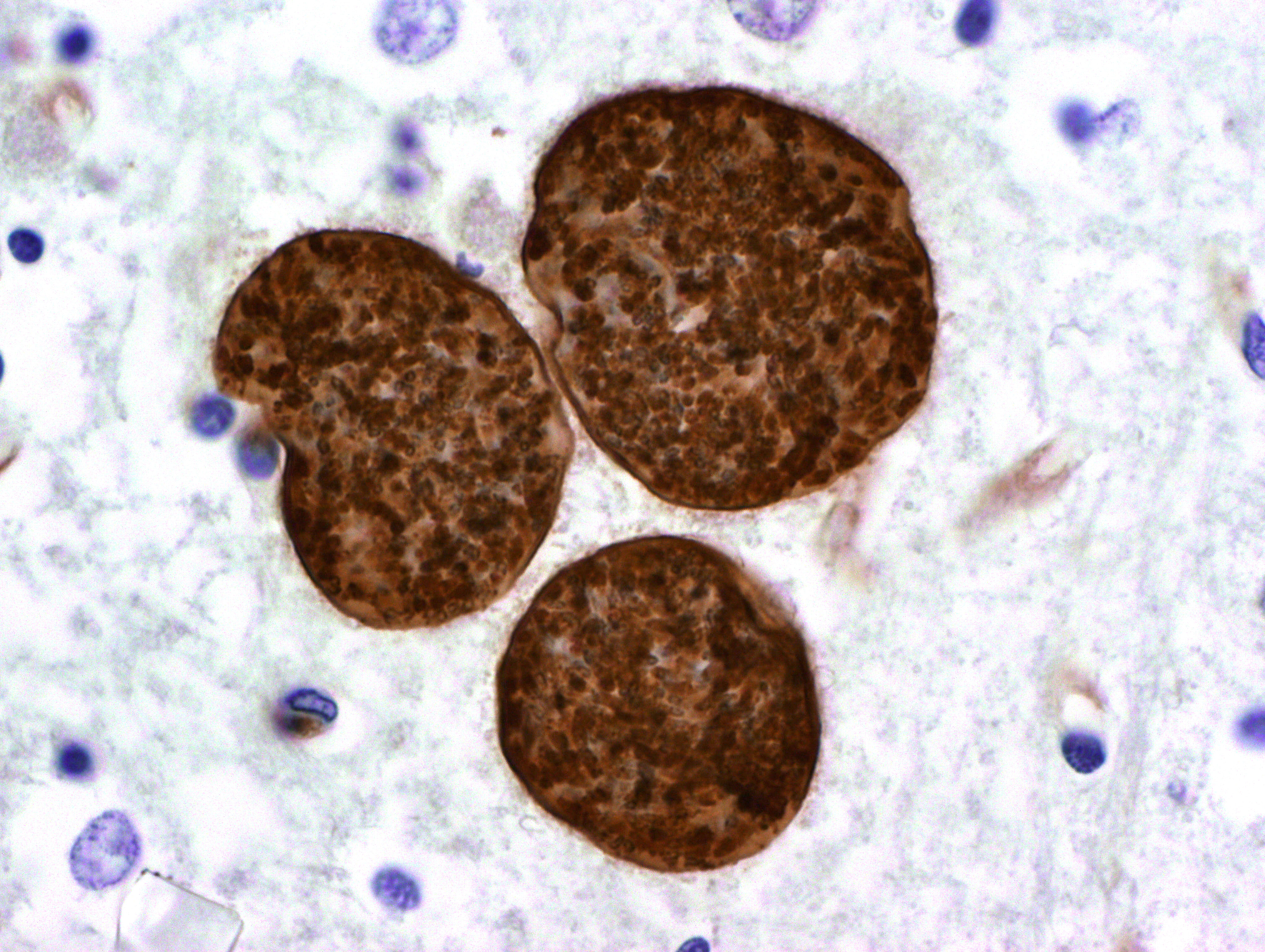
Contributor Comment:
Infection occurs by ingestion of sporulated oocysts excreted in the feces of felids, by ingestion of tissues of intermediate hosts that contain encysted bradyzoites or tachyzoites, and less frequently by vertical transmission. Once ingested, sporozoites excyst and multiply in the intestinal epithelial cells as tachyzoites. Tachyzoites can either disseminate and infect cells throughout the body resulting in the necrosis and non-suppurative inflammation characteristic of toxoplasmosis, or encyst in tissues as bradyzoites. Following ingestion of tissue cysts by an intermediate host, bradyzoites will excyst, become tachyzoites, and the cycle continues.(2,6,7)
There is one report of toxoplasmosis in an elephant seal pup. (4) Microscopic lesions included multifocal non-suppurative meningoencephalitis and multiple tissue cysts with and without associated inflammation in the cerebrum. Cyst morphology was consistent with T. gondii, and protozoa stained positively with T. gondii, but not with N. caninum, polyclonal antibody. Focal lymphoplasmacytic inflammation was present in the brain, retina, optic nerve and renal tubules, and non-suppurative glossitis with necrosis and ulceration was also observed.(4)
Toxoplasmosis in marine mammals has recently become of particular concern since being identified as a leading cause of encephalitis and death in the threatened Southern sea otter (Enhydra lutris nereis).(8) Since 1951, toxoplasmosis has been reported in various species of seals, dolphins, a sea lion, a West Indian manatee and a beluga whale.(5) Serological assays of numerous species of marine mammals suggests common and widespread exposure. (5)
It is unclear how marine mammals become infected with T. gondii as they rarely consume recognized intermediate hosts, and T. gondii is not known to parasitize fish or invertebrates. It has been proposed that infection occurs through consumption of oocysts that enter the marine environment via surface run-off or municipal sewage contaminated by cat feces.(9,11) In support of this theory, T. gondii oocysts have been shown to sporulate and survive in seawater for several months. (9) Laboratory experiments have shown that bivalves can concentrate T. gondii oocysts (9) and recently, a wild California mussel was confirmed positive for T. gondii(10) suggesting that invertebrate filter feeders can serve as a source of infection for marine mammals. Additionally, a type X strain of T. gondii that has recently been isolated in over 72% of all sea otter infections(2) was identified in the California mussel as well as in several coastal dwelling felids and canids.(10)
JPC Diagnosis:
Conference Comment:
Numerous organ systems are affected by toxoplasmosis, with pulmonary lesions and central nervous system lesions having the highest prevalence.(1) Within the lung, lesions are characterized by necrosis of alveolar walls, bronchiolar epithelium, and the vasculature with an accompanying interstitial pneumonia with mononuclear cell invasion into the alveolar walls.(1) Multifocal necrosis within the central nervous system and accompanying non-suppurative inflammation can occur with toxoplasmosis. Microglial nodules are occasionally seen with chronicity within the parenchyma of the central nervous system.(1)
References:
2. Conrad PA, Miller MA, Kreuder C, James ER, Mazer J, Dabrtiz H, Jessup DA, Gulland F, Grigg ME: Transmission of Toxoplasma: Clues from the study of sea otters as sentinels of Toxoplasma gondii flow into the marine environment. Int J Parasitol 35:1155-1168, 2005
3. Dubey JP: Toxoplasmosis-a waterborne zoonosis. Vet Parasitol 126:57-72, 2004
4. Dubey JP, Lipscomb TP, Mense M: Toxoplasmosis in an elephant seal (Mirounga angustirostris). J Parasitol 90:410-411, 2004
5. Dubey JP, Zarnke R, Thomas NJ, Wong SK, Van Bonn W, Briggs M, Davis JW, Ewing R, Mense M, Kwok OCH, Romand S, Thulliez P: Toxoplasma gondii, Neospora caninum, Sarcocystis neurona and Sarcocystis canis-like infections in marine animals. Vet Parasitol 116:275-296, 2003
6. Gardiner CH, Fayer R, Dubey JP: Apicomplexa Toxoplasma and Hammondia. In: An atlas of protozoan parasites in animal tissues, 2nd ed., pp. 53-56. Armed Forces Institute of Pathology, Washington, DC 1998
7. Hill DE, Chirukandoth S, Dubey JP: Biology and epidemiology of Toxoplasma gondii in man and animals. Animal Health Research Reviews 6:41-61, 2005
8. Kreuder C, Miller MA, Jessup DA, Lowenstine LJ, Harris MD, Ames JA, Carpenter TE, Conrad PA, Mazet JAK: Patterns of mortality in Southern sea otters (Enhydra lutris nereis) from 1998-2001. J Wildl Dis 39:495-509, 2003
9. Lindsay DS, Collins MV, Mitchell SM, Cole RA, Flick GJ, Wetch CN, Lindquist A, Dubey JP: Sporulation and survival of Toxoplasma gondii oocysts in seawater. J Eukaryot Microbiol 50:687-8, 2003
10. Lindsay DS, Collins MV, Mitchell SM, Wetch CN, Rosypal AC, Flick GJ, Zajac AM, Lindquist A, Dubey JP: Survival of Toxoplasma gondii oocysts in Eastern oysters (Crassostrea virginica). J Parasitol 90:1054-1057, 2004
11. Miller MA, Gardner IA, Kreuder C, Paradics DM, Worcester KR, Jessup DA, Dodd E, Harris MD, Ames JA, Packham AE, Conrad PA: Coastal freshwater runoff is a risk factor for Toxoplasma gondii infection of southern sea otters (Enhydra lutris nereis). Int J Parasitol 33:997-1006, 2002
12. Miller MA, Miller WA, Conrad PA, James ER, Melli AC, Leutenegger CM, Dabritz HA, Packham AE, Paradics DM, Harris M, Ames J, Jessup DA, Worcester K, Griggs ME: Type X Toxoplasma gondii in a wild mussel and terrestrial carnivores from coastal California: New linkages between terrestrial mammals, runoff and toxoplasmosis of sea otters. Int J Parasitol article in press, 2008