Signalment:
Adult male Djungarian hamster (
Phodopus sungorus).Four adult dwarf hamsters with no significant prior medical history were on display at a museum petting exhibit. The animals were in the process of being retired from the exhibit with the intention of adoption when skin lesions were noticed on two hamsters. A male hamster described to have one necrotic pinna was culled and both pinnae were submitted for biopsy. Within one week of receiving biopsy results, a second male adult hamster with an abdominal skin lesion was culled and submitted for autopsy. The submitted tissue section is from the second hamster.
Gross Description:
A 50g adult male hamster is presented for postmortem examination. The carcass is in good postmortem condition with a euthanasia-to-necropsy interval of 13 hours. Throughout the body, adequate adipose stores are present. A focally extensive area of caudal abdominal skin, extending from the umbilicus to the perineal region and encompassing both the left and right inguinal areas, has an asymmetric, poorly haired, moist, red patch with a 0.1 cm in diameter paramedian ulcer and several small irregular brown crusts. No internal gross abnormalities are noted. Caudal abdominal skin is submitted.
Histopathologic Description:
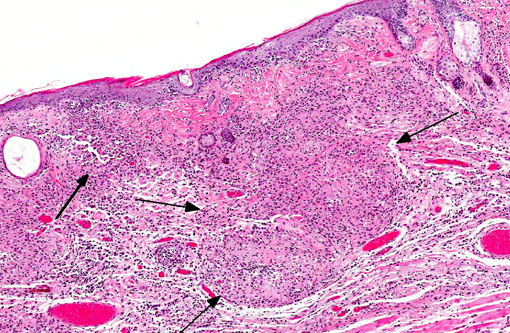
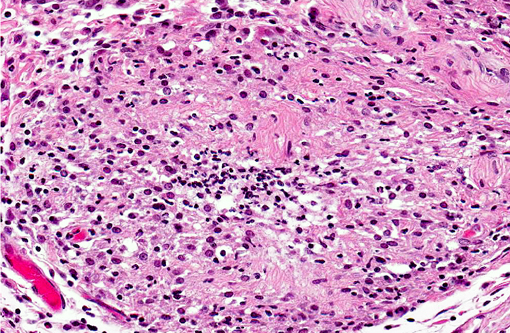
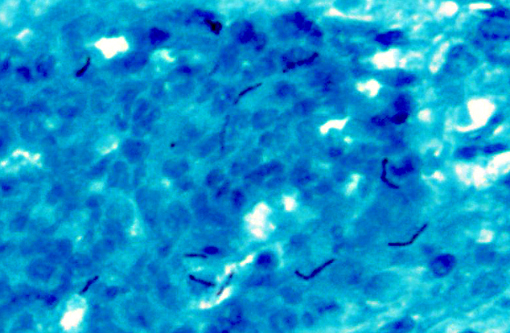
Abdominal skin: The dermis and panniculus are expanded by multifocal to coalescing nodular infiltrates of abundant histiocytes and neutrophils that form multifocal pyogranulomas with abundant central degenerative neutrophils and necrotic cellular debris surrounded by a thick rim of epithelioid macrophages. Low numbers of gram-positive, acid-fast bacilli are present in scattered histiocytes. The overlying epithelium is multifocally, irregularly hyperplastic with multifocal intracellular edema, frequent neutrophil and lymphocyte transmigration which frequently disrupts the basal layer, few intraepidermal neutrophil aggregates, and mild orthokeratotic hyperkeratosis. (The original pinna biopsy submission from the male hamster has similar histological findings.)
Morphologic Diagnosis:
Haired skin: Marked, multifocal to coalescing pyogranulomatous dermatitis and panniculitis with intralesional acid -fast bacilli (mycobacteriosis).
Lab Results:
Formalin-fixed, paraffin-embedded tissue was submitted for mycobacterial PCR, and the resulting sequence most closely matched that of
Mycobacterium marinum and
M. ulcerans (greater than 99% sequence identity with GenBank acc#AB026701 and CP000325).
Condition:
Mycobacteriosis
Contributor Comment:
Mycobacteriosis in mammals typically occurs via contamination of skin wounds or traumatic inoculation and rarely via inhalation.(6) Direct contact with environmental inhabitants generally results in local disease, but can pose a greater risk to immunocompromised individuals.Â
M. marinum is ubiquitous in fresh, brackish, and salt water, and causes disease in fish and humans (fish tank granulomas or swimming pool granulomas). Human infection typically results from direct inoculation into skin abrasions while handling fish or tank water, leading to slow growing nodules at the site of inoculation and local lymphadenopathy.(3)
M. ulcerans is the causative agent of Buruli ulcer, the third most common mycobacterial disease in humans. Epidemiologic factors are not completely understood, as the disease is most common in dry periods in wetlands of tropical or subtropical regions.(6) The toxin mycolactone is believed to cause the characteristic ulcerative and necrotic cutaneous and subcutaneous lesions.(2)
M. ulcerans is a rare cause of atypical mycobacteriosis in cats, causing ulcerative and nodular skin lesions.(7) Koalas and possums in Australia are naturally infected with
M. ulcerans.(5) Recently it has been proposed that small mammals may serve as a reservoir for
M. ulcerans.(1)
The source of mycobacterial infection in the museum hamsters was not identified, although there are numerous aquatic displays at the museum. Despite the zoonotic potential, no human cases of infection were reported.
JPC Diagnosis:
1. Haired skin and subcutis: Dermatitis, pyogranulomatous, multifocal to coalescing, moderate, with edema and rare intrahistiocytic acid-fast bacilli.
2. Haired skin, epidermis: Hyperplasia, multifocal, mild to moderate with mild orthokeratotic hyperkeratosis, intracorneal pustules and occasional acarid parasites.
Conference Comment:
Mycobacterium ulcerans and
M. marinum are closely related, slow growing, opportunistic pathogens most notable for causing skin disease in humans(8) (see
WSC 2013-14 conference 2, case 2 for a summary of the mycobacterial classification system).Â
Aquatic Acanthamoeba sp. has been implicated as a natural host of
M. marinum and
M. ulcerans, and may play an important role in disease transmission;(9) however, in this case there is no indication that the affected hamsters were housed near the museums aquatic displays.Â
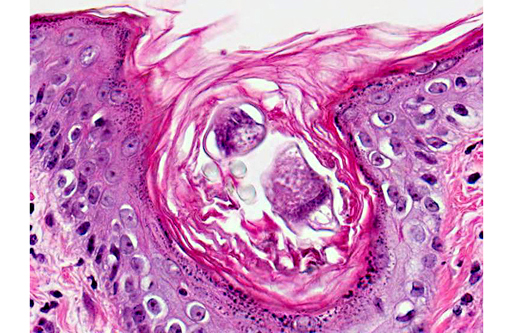
In addition to the striking dermal lesions in this hamster, attributed to infection with
M. marinum/ulcerans, there are rare intrafollicular and intracorneal segments of arthropods, up to 40 μm in diameter and 200 μm in length, with a thin, eosinophilic, chitinous exoskeleton, short jointed appendages, a hemocele, striated muscle, and digestive and reproductive tracts. These arthropods are associated with epidermal hyperplasia, orthokeratosis, and occasional intracorneal pustules. The differential diagnosis includes
Demodex sp. and
Notoedres sp.Â
Demodex criceti and
D. aurati infestation is common in many hamster colonies; however, these mites exhibit low pathogenicity and rarely cause clinical signs.Â
Notoedres notoedres, a mite that burrows into the stratum corneum, is less common, although it can be enzootic in some hamster colonies. Males typically have a higher parasite load than females, and factors such as advanced age or stress from handling may predispose skin lesions in animals with no previous clinical signs.(4)
References:
1. Durnez L, Suykerbuyk P, Nicolas V, Barriere P, Verheyen E, Johnson CR, et al. Terrestrial small mammals as reservoirs of Mycobacterium ulcerans in Benin. Appl Environ Microbiol. 2010;76(13):4574-4577.
2. George KM, Pascopella, L, Welty DM, Small PL. A Mycobacterium ulcerans toxin, mycolactone, causes apoptosis in guinea pig ulcers and tissue culture cells. Infec. Immun. 2000;68(2):877-883.
3. Iowa State Animal Disease Fact Sheet: Mycobacteriosis. 2007. www.cfsph.iastate.edu/DiseaseInfo/factsheets.php
4. Percy DH, Barthold SW. Hamster. In: Pathology of Laboratory Rodents and Rabbits. 3rd ed. Ames, IA: Blackwell Publishing; 2007:193-194.
5. Portaels F, Chemlal K, Elsen P, Johnson PDR, Hayman JAA, Hibble J, et al. Mycobacterium ulcerans in wild animals. Rev Sci Tech. 2001;20(1):252-264.Â
6. Portaels F. Mycobacterial diseases of the skin. 1995:207.
7. Songer JG, et al. Veterinary Microbiology: Bacterial and Fungal Agents of Animal Disease. St. Louis, MO: Elsevier Saunders; 2004:95.
8. Wayne LG, Sramek HA. Agents of newly recognized or infrequently encountered mycobacterial diseases. Clin Microbiol Rev. 1992;5(21):1-25.
9. Wilson MD, Boakye DA, Mosi L, Asiedu K. In the case of transmission of Mycobacterium ulcerans in buruli ulcer disease Acanthamoeba species stand accused. Ghana Med J. 2011;45(1):31-34.