Signalment:
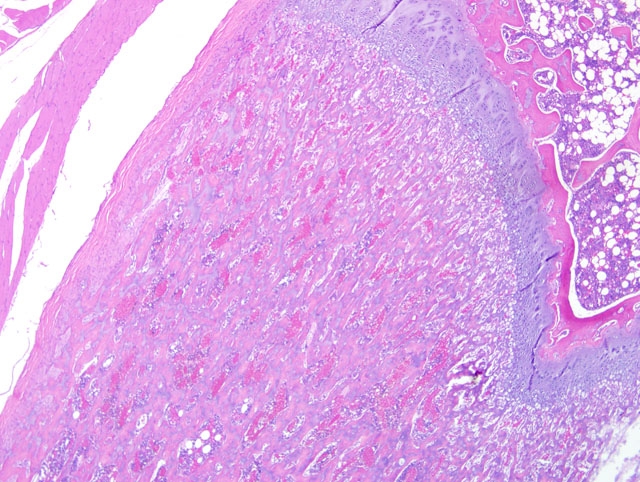
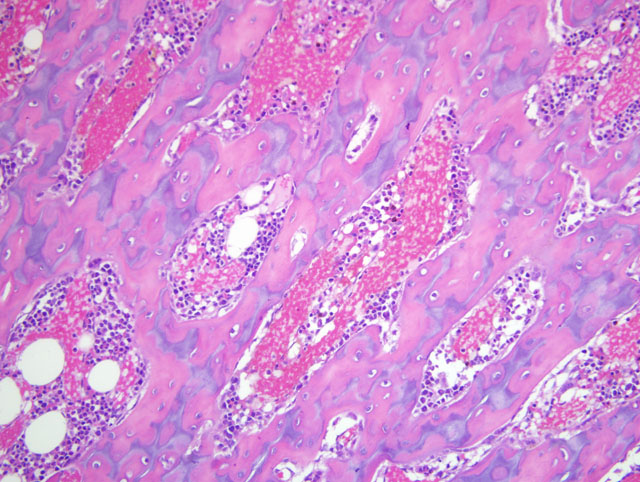
Histopathologic Description:
Morphologic Diagnosis:
Condition:
Contributor Comment:
Normal longitudinal bone growth occurs as a result of endochondral ossification, a highly coordinated process whereby the production of a cartilaginous scaffold at the growth plate is replaced by bone. Many factors have been identified as playing a role in this process; hormones such as estrogens, thyroid and growth hormone, leptin, sex hormones and glucocorticoids, as well as growth factors such as insulin-like growth factor, Indian hedgehog, PTHrp, fibroblast growth factor, bone morphogenetic proteins, vascular endothelial growth factor (VEGF) and vitamin D have all been implicated.(2,6)
Glucocorticoids exert their effect via the ubiquitous glucocorticoid receptor or the less widely distributed mineralocorticoid receptor. The glucocorticoid receptor is a cytoplasmic receptor which, upon ligand binding, forms a multi-component complex, translocates to the nucleus and functions as a transcription factor, binding to promoter regions of glucocorticoid responsive genes. This activity may promote or repress transcription depending on the gene involved. As well, the glucocorticoid-bound receptor complex may exert an indirect effect, by interfering with the normal function of other transcription factors (nuclear factor-κB or smad-3 for example). The result is a myriad of effects, both desired and unwanted, across multiple systems.(4)
Glucocorticoids are generally considered inhibitory to endochondral ossification and have been associated with a number of adverse skeletal affects, including delayed or reduced growth and osteopenia. Glucocorticoids exert a direct effect on growth plate chondrocytes, which express the glucocorticoid receptor, by decreasing the rate of chondrocyte proliferation as well as increasing the rate of chondrocyte apoptosis at the zone of hypertrophy.(2) Glucocorticoids also interact with other mediators of bone growth. Insulin-like growth factor-1 (IGF-1) is considered a key mediator of bone growth, and its expression at the growth plate is determined by the relative influence of locally acting glucocorticoids, thyroid and growth hormones. In this regard, glucocorticoids have been shown to modulate both IGF-1 production and growth hormone receptor expression at the growth plate.(2) Other cell populations responsible for growth and remodeling are also impacted by glucorticoid exposure; osteoblast activity is typically inhibited, with decreased matrix/osteoid production whilst osteclastic activity is often increased, resulting in an uncoupling of the tightly controlled interplay between these two populations.(4)
Angiogenesis is an important part of the endochondral ossification process. In piglets treated with short term prednisolone, there was a decrease in VEGF mRNA expression by hypertrophic chondrocytes, with diminished capillary ingrowth and loss of parallel organization in the metaphysis.(5) Interestingly, trabecular bone length in the primary spongiosa was also diminished, in contrast to the case presented here. Elongation of the primary spongiosa, as was seen in our case, appears not to be specifically referenced within the literature; it seems reasonable to hypothesize that the net outcome of glucorticoid exposure seen here results from the balance of relative dose or exposure both systemically and at the growth plate, acting in concert at multiple levels in young, actively growing rats under the conditions of this study.
This case also demonstrates marrow hematopoietic hypocellularity with a corresponding increase in marrow lipocytes. This is also a recognized effect of glucocorticoid therapy and has been implicated in the pathogenesis of steroid-induced osteonecrosis. Rather than unmasking existent adipocytes due to loss of hematopoietic elements, a shift in differentiation of pluripotential marrow stem cells has been proposed to occur under the influence of glucocorticoids.(1)
JPC Diagnosis:
Conference Comment:
As in the section of proximal humerus discussed in case II of this conference, metaphyseal osteosclerosis in this case is the result of retention of unmodeled primary trabeculae. The contributors other assessments, including thinning of the physeal cartilage, increased growth plate-to-spongiosa ratio, and bone marrow hypocellularity are substantiated by the microscopic images supplied from unaffected control animals; without the benefit of the control images for reference, conference participants were unable to confidently make the same interpretations. Regardless, the conference moderator emphasized that the presence of retained unmodeled primary trabeculae is far more reliable evidence of deficient osteoclasis than is the subjective determination that osteoclast numbers are reduced, particularly when a section from an age-matched control is not available for evaluation. As in case II, this finding prompts the consideration of other anti-osteoclast agents, including lead and bisphosphonates. Participants noted the apparent lack of similar lesions in the epiphysis, and speculated that this unexpected finding may have resulted from a different growth rate or plane of section.
The finding of osteosclerosis in this case is counterintuitive; due to their well-known catabolic properties, glucocorticoids would be expected to result in increased osteoclast lifespan and osteopenia. The contributor offers several plausible explanations for this irony. Conference participants discussed the important roles that dosage route and frequency may have on the net effect of a given drug, using as a corollary the paradox of parathyroid hormone. As alluded to in the discussion of fibrous osteodystrophy in case I of this conference, persistent hyperparathyroidism is known to increase bone resorption. Less obviously, parathyroid hormone (PTH) can also stimulate bone formation and increased bone mass, depending on the route and frequency of administration. Specifically, while continuous infusions of PTH cause bone resorption, intermittent injections of the same hormone cause increased bone formation and bone mineral density. This is the therapeutic basis for teriparatide (Forteo-�-�), a recombinant drug composed of the first 34 amino acids of PTH, which exerts its main biologic effects and is clinically shown to decrease the risk of fractures and increase bone mineral density in postmenopausal women with osteoporosis when administered as a daily subcutaneous injection.(2)
References:
2. Neer RM, Arnaud CD, Zanchetta JR, Prince R, Gaich GA, Reginster JY, Hodsman AB, Eriksen EF, Ish-Shalom S, Genant HK, Wang O, Mitlak BH: Effect of parathyroid hormone (1-34) on fractures and bone mineral density in postmenopausal women with osteoporosis. N Engl J Med 344:1434-1441, 2001
3. Nilsson O, Marino R, De Luca F, Phillip M, Baron J: Endocrine regulation of the growth plate. Horm Res 64:157-165, 2005
4. Sch+�-�cke H, D+�-�cke WD, Asadullah K: Mechanisms involved in the side effects of glucocorticoids. Pharmacol Ther 96:23-43, 2002
5. Smink JJ, Bucholz IM, Hamers N, van Tilburg CM, Christis C, Sakkers RJ, de Meer K, van Bull-Offers SC, Koedam JA: Short-term glucocorticoid treatment of piglets causes changes in growth plate morphology and angiogenesis. Osteoarthritis Cartilage 12:864-871, 2003
6. van der Eerden BC, Karperien M, Wit JM: Systemic and local regulation of the growth plate. Endocr Rev 24:782-801, 2003