Signalment:
Gross Description:
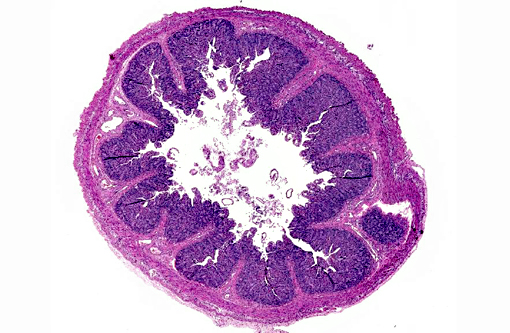
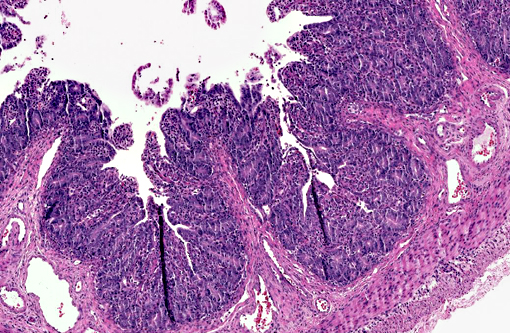
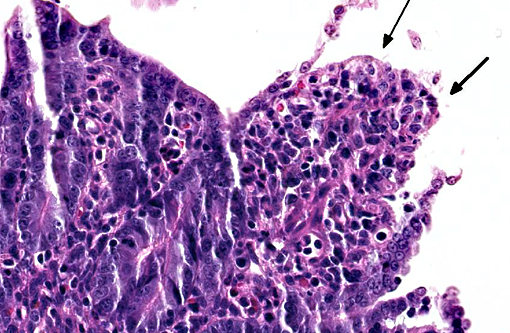
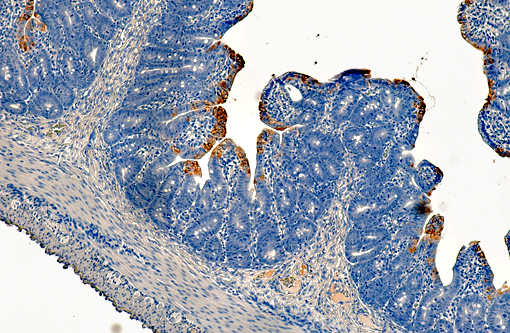
Histopathologic Description:
Morphologic Diagnosis:
Lab Results:
Condition:
Contributor Comment:
The pathology of PEDV has been described in gnotobiotic pigs.(3) The lesions were similar to those observed in conventional pigs infected with PEDV.
JPC Diagnosis:
Conference Comment:
PEDV is an Alphacoronavirus that contains an enveloped, single-stranded positive-sense RNA genome.(2) It was first discovered in the UK in 1971 and continued to cause sporadic outbreaks in Europe and Asia for decades.(2) Though it is similar in structure and pathogenesis as TGE, its genome is more closely related to bat Alphacoronaviruses.(8) The virus targets intestinal epithelial cells of nursing pigs; however, infection of alveolar macrophages has also been demonstrated.(7) Typical microscopic findings include severe atrophic enteritis throughout the small intestine and viral shedding can precede and continue beyond the observation of clinical signs.(5) In this case, slides from multiple blocks were submitted and among them, the lesions varied from mild blunting and fusion of villi with moderate crypt hyperplasia to severe necrosis. There are also sections with mild loss of lymphocytes in Peyer's patches. Syncytial cell formation was evident throughout the more severely affected lesions.Â
There has been much attention on the possible mechanisms of virus transmission. Transportation equipment and contaminated feedstuffs are the most often cited culprits, as this may explain the sporadic, simultaneous outbreaks in facilities separated by hundreds of miles.(3) Transmission between animals is largely fecal-oral, but airborne transmission is also suspected.(1)
PEDV contains a transmembrane envelope glycoprotein virulence factor called spike, which is responsible for binding and fusion to host epithelial cells and macrophages. Spike contains two domains, S1 and S2, and the S1 domain is the primary target of vaccines due to its specific high-affinity binding to cell receptors.(6) The first licensed vaccine for PEDV was released in the U.S. in 2014, and now there are several on the market. Incidence rate has been dropping recently and, while the disease has killed over seven million pigs in just the last year, it appears the peak of this devastating epidemic has already passed.
References:
1. Alonso C, Goede DP, Morrison RB, et al. Evidence of infectivity of airborne porcine epidemic diarrhea virus and detection of airborne viral RNA at long distances from infected herds. Vet Res. 2014;45:73.
2. Stevenson GW, et al. Emergence of porcine epidemic diarrhea virus in the United States: clinical signs, lesions, and viral genomic sequences. J of Vet Diagn Invest. 2013;25(5):649654.Â
3. Jung K, et al. Pathology of US porcine epidemic diarrhea virus strain PC21A in gnotobiotic pigs. Emerg Infect Dis. 2014;20(4):662-665.
4. Lowe J, Gauger P Harmon K, et al. Role of transportation in spread of porcine epidemic diarrhea virus infection, United States. Emerg Infect Dis. 2014;20(5):872-874.
5. Madson DM, Magstadt DR, Arruda PH, et al. Pathogenesis of porcine epidemic diarrhea virus isolate (US/Iowa/18984/2013) in 3-week-old weaned pigs. Vet Microbiol. 2014 Sep 22. pii: S0378-1135(14)00425-8. [Epub ahead of print]
6. Oh J, Lee KW, Choi HW, Lee C. Immunogenicity and protective efficacy of recombinant S1 domain of the porcine epidemic diarrhea virus spike protein. Arch Virol. 2014;159(11):2977-2987.
7. Park JE, Shin HJ. Porcine epidemic diarrhea virus infects and replicates in porcine alveolar macrophages. Virus Res. 2014;191:143-152.
8. Vlasova AN, Marthaler D, Wang Q, et al. Distinct characteristics and complex evolution of PEDV strains, North America, May 2013-February 2014. Emerg Infect Dis. 2014;20(10):1620-1628.