Joint Pathology Center
Veterinary Pathology Services
Wednesday Slide Conference
2019-2020
Conference 7
9 October 2019
Dr. John Cullen, VMD, PhD, DACVP, FIAT
Professor, Anatomic Pathology
College of Veterinary Medicine
North Carolina State University
Raleigh, North Carolina
CASE III: 15-0318 (JPC 4101080).
Signalment: 6-month-old intact male Mi-Ki dog (Canis familiars).
History: The puppy presented with a history of lethargy, hyporexia, occasional vomiting, and abnormal behavior including opisthotonus and head pressing. A single seizure was observed, which resolved with diazepam. Following diazepam administration, the puppy became stuporous with a lack of menace response and bilaterally decreased pupillary light reflexes. The puppyâs mentation gradually improved with treatment (lactulose, metronidazole, omeprazole, ondansetron and intravenous fluids with dextrose). The patient was sedated with butorphanol and alfaxalone for an abdominal ultrasound, and became obtunded with abnormal vocalization that persisted despite supportive care. Due to a poor prognosis, the puppy was euthanized.
Gross Pathology: The puppy weighed 1.9 kg with adequate visceral adipose. The liver was diffusely small with normal lobation, color, and consistency. A single dilated and tortuous anomalous vessel, approximately 3 mm wide, connected the gastroduodenal vein with the caudal vena cava. There was mild, bilaterally symmetric dilation of the lateral ventricles. The brain was otherwise grossly unremarkable. The lungs were mildly, diffusely wet and oozed a small amount of serosanguineous fluid and foam on cut section. Sections of lung from all lobes floated in formalin. No other gross lesions were observed.
Laboratory results: Chemistry abnormalities: Albumin 2.8 g/dL, Globulins 2.2 g/dL, Creatinine 0.3 mg/dL, ALP 177 U/L, Phosphorous 6.6 mg/dL, Glucose 66 mg/dL, Cholesterol 67 mg/dL
Ammonia: 213 μmol/L (reference range: 11-35 μmol/L)
Bile acids: 112 μmol/L (pre-prandial) and 104 μmol/L (post-prandial)
Urine specific gravity: 1.026
Urine sediment: 11-20 ammonium biurate crystals /hpf
Imaging:
Abdominal ultrasonography identified a small liver with a suspected single extrahepatic portosystemic shunt from the right gastric or gastroduodenal vein to the caudal vena cava.
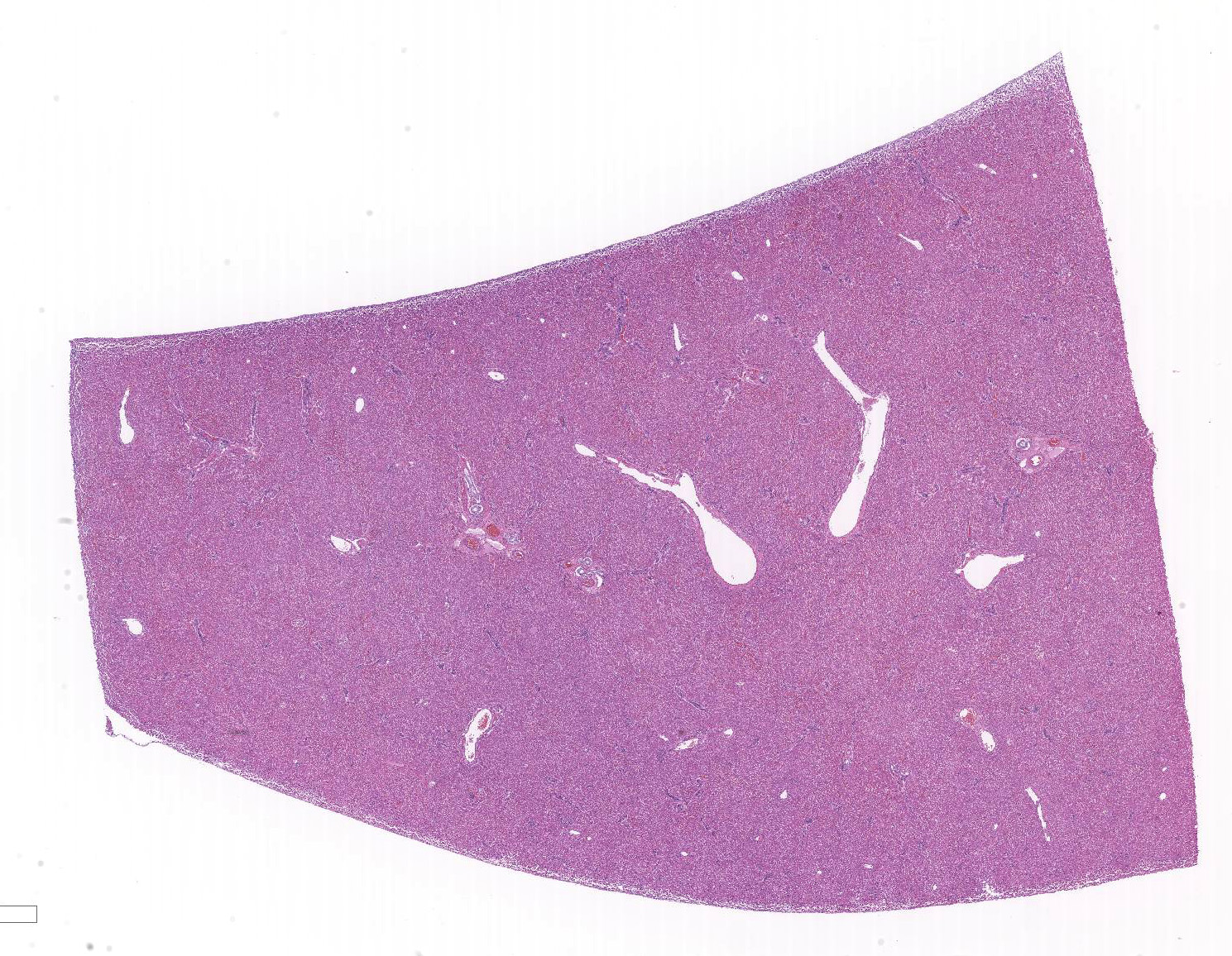
Microscopic Description:
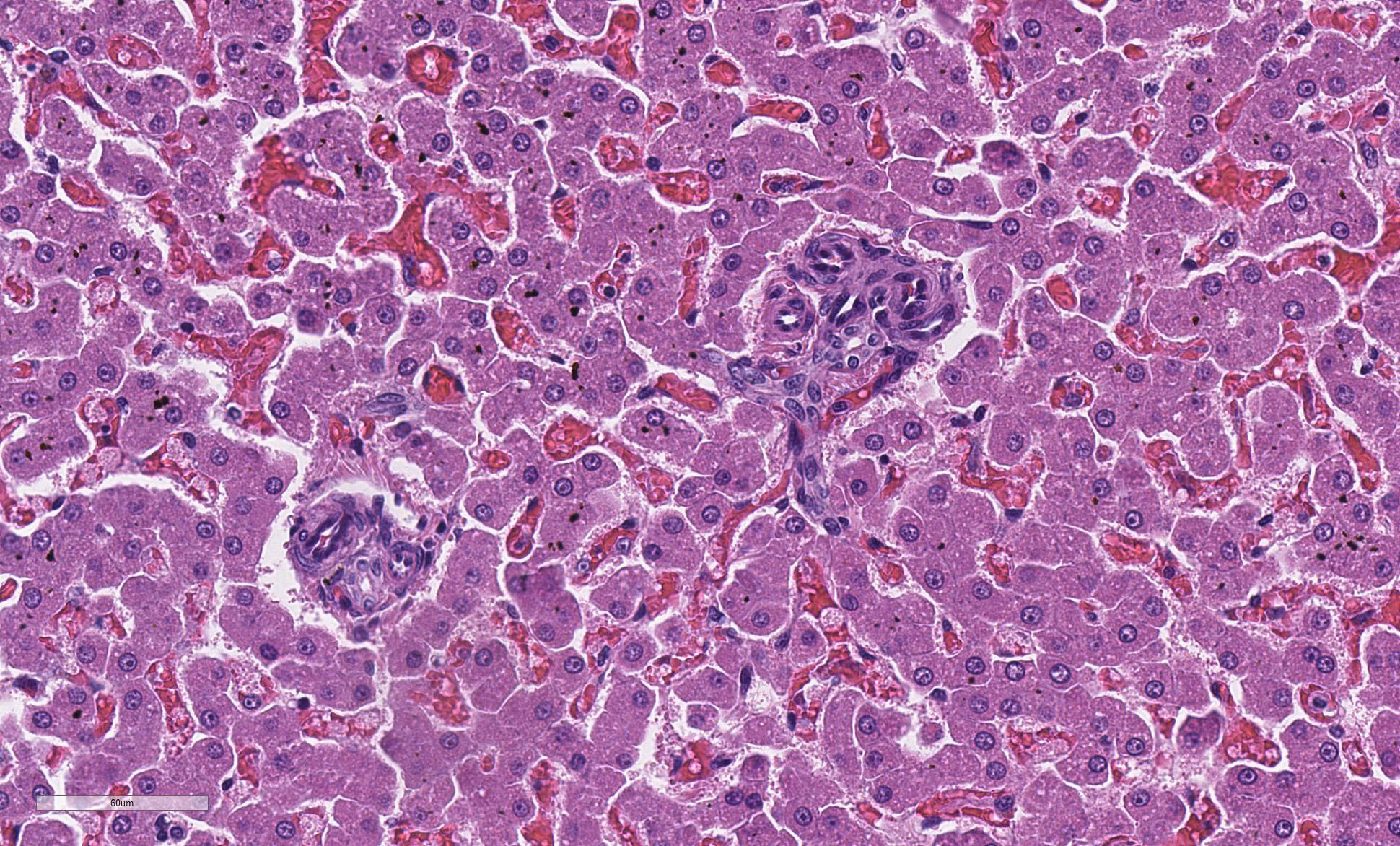
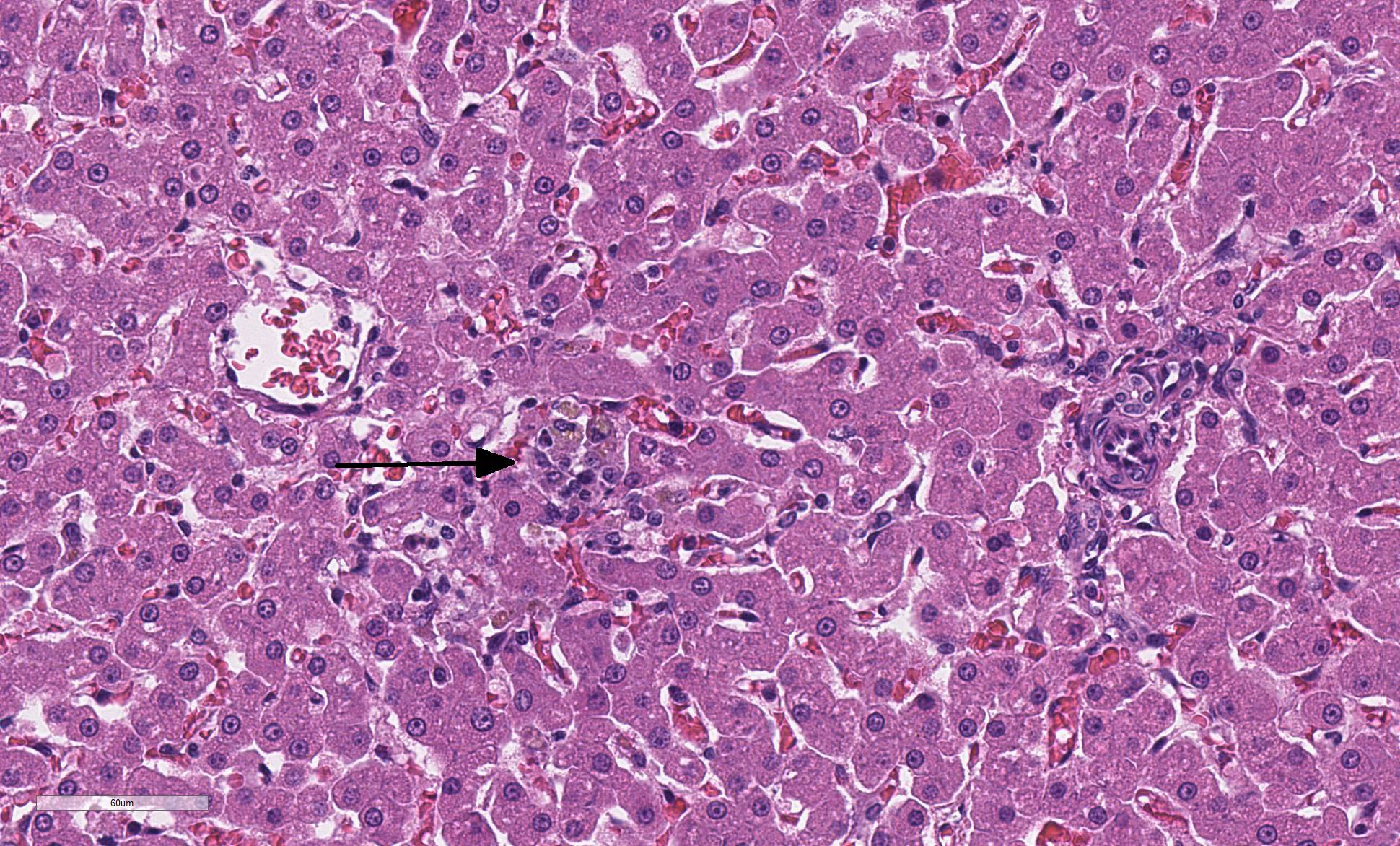
The liver is composed of small hepatic lobules with irregularly and closely spaced portal triads. Portal triads contain increased numbers of arteriolar profiles and portal venules are often small or absent. Larger portal tracts contain dilated lymphatic vessels. Cords of hepatocytes surrounding portal tracts are thin, and individual hepatocytes are small, with a small amount of eosinophilic, occasionally finely vacuolated cytoplasm. The periportal sinusoids are infrequently dilated. Some sections contain randomly distributed small foci of inflammation composed of low numbers of lymphocytes, macrophages, and neutrophils, with individually necrotic hepatocytes. Scattered rarely throughout the parenchyma of some sections are small clusters of lipid and golden-brown pigment laden macrophages with fewer lymphocytes (pigment lipogranulomas).
In sections of brain (not submitted), the white matter, and to a lesser extent, the grey matter throughout the cerebrum and midbrain contain numerous variably sized, discrete to coalescing clear vacuoles. The lateral ventricles are mildly bilaterally and symmetrically enlarged.
Contributor Morphologic Diagnosis:
Dog, liver: hepatopathy characterized by lobular hypoplasia with hepatocellular atrophy, increased arteriolar profiles, and portal vein hypoplasia
Dog, liver: mild multifocal random lymphohistiocytic and neutrophilic hepatitis with single cell necrosis
Contributor Comment: Portosystemic shunts are any anomalous connections of the portal circulation to the systemic circulation that bypass the liver. Acquired hepatic shunts are distinguished from congenital shunts on ultrasonography, exploratory laparotomy, or gross examination, as acquired shunts are typically composed of multiple anastomosing vessels rather than a single large vessel. Acquired shunts most often arise secondary to portal hypertension, such as in cirrhosis or portal vein obstruction, but can also develop in the presence of microvascular malformations1,2.
Congenital hepatic shunts are categorized into intra- and extrahepatic shunts, and are thought to be an inherited condition in some breeds (Cairn terriers, Yorkshire terriers, Irish wolfhounds, Maltese, Australian cattle dogs)3,4,5,6. Large breed dogs tend to develop intrahepatic shunts, most commonly due to a patent ductus venosus6. Extrahepatic shunts are more common in small breed dogs and cats, most frequently connecting the portal vein, gastric vein, or splenic vein with the caudal vena cava or azygous vein1. Patients often present at less than 1 year of age, but can progress into adulthood before clinical signs become apparent. Clinical signs may include an overall small size compared to littermates, depressed mentation, circling, seizures, and other manifestations of hepatic encephalopathy. Clinical pathology findings can include microcytosis, hypoalbuminemia, hypocholesterolemia, hypoglycemia, low BUN, increased bile acid concentration, hyperammonemia, and ammonium biurate crystalluria1,7.
Blood in the portal circulation contains hepatotrophic growth factors such as insulin, glucagon, and hepatocyte growth factor, which are essential for normal hepatocellular development8. Lack of blood flow and delivery of growth factors to the developing liver results in small hepatic lobules and hepatocytes, increased arteriolar profiles, small to absent portal veins, and pigment lipogranulomas2,9. These histologic features are characteristic of portal vein hypoperfusion; however, are not specific for a portosystemic shunt. Similar histologic lesions are also seen in cases of primary portal vein hypoplasia1,2.
Primary portal vein hypoplasia is the current preferred term for conditions previously described as microvascular dysplasia, hepatoportal fibrosis, and idiopathic noncirrhotic portal hypertension2. In contrast to a portosystemic shunt, portal vein hypoplasia can result in portal hypertension, ascites, and acquired shunts1,2. Microscopically, portal fibrosis and bile duct hyperplasia are sometimes noted in addition to the characteristic histologic features of portal vein hypoperfusion2,7. Without an adequate history, clinical findings or imaging results, it is often impossible to differentiate between primary portal vein hypoplasia and congenital portosystemic shunts with histology alone. Concurrent primary portal vein hypoplasia and congenital portosystemic shunts have been reported in dogs7.
In this case, the microscopic hepatic and brain lesions correlated well with the gross identification of a distinct anomalous vessel, clinical signs, and clinical pathologic abnormalities. This constellation of findings is consistent with an extrahepatic congenital portosystemic shunt and hepatic encephalopathy. The cause of the mild, multifocal random hepatitis is unclear, though the distribution suggests possible early sepsis rather than an ischemic or toxic etiology.
Contributing Institution:
University of Pennsylvania, School of Veterinary Medicine, Department of Pathobiology
http://www.vet.upenn.edu/research/academic-departments/
JPC Diagnosis: Liver, portal veins: Hypoplasia, diffuse, severe, with lobular and hepatocellular atrophy
JPC Comment: The contributor has provided a concise but illustrating explanation of congenital hepatic shunts and portal vein hypoplasia in the dog.
In a recent study of 125 dogs with developmental hepatic vascular disease8, the initial diagnosis was confirmed in 89.3% of cases in which suspicion of portosystemic shunt was raised following liver biopsy, demonstrating the sensitivity of liver biopsy in such cases. In this study, animals were divided into those with intrahepatic shunts, those with extrahepatic shunts, and those with hepatic microvascular dysplasia - portal vein hypoplasia. Cases of microvascular dysplasia without apparent shunting were most commonly seen in Yorkshire terriers. In addition to more classic histologic findings of compressed lobules, multiple sections of tortuous arterioles in portal triads, and absence of portal veins within triads, this study also noted consistent but less common histologic lesions. Approximately 20% of cases of either intra- or extrahepatic shunting had hypertrophy of smooth muscle around sublobular veins, which increased to 80% in cases of microvascular dysplasia. Fibrosis of the central veins was noted in 22% of extrahepatic shunts and microvascular dysplasia but 60% of intrahepatic shunts. Calcification was visible in approximately 2% of animals with developmental hepatic vascular disease, but did not appear to be the result of dystrophic calcification of the vessel wall itself. Finally, lipogranuloma formation appeared to be most common in extrahepatic shunts (50%) but twenty percent or less in the other two categories, and tended to be more prominent in older dogs in the study.8
Regarding evalution of this particular specimen, the moderator made several observations. The moderator mentioned that in liver biopsies (not autopsies) you should not see sinusoids, else you are likely viewing hepatocellular atrophy. You may see it in autopsy specimens following postmortem consumption of glycogen, resulting shrinkage up to 33% of overall size. Addtionally, in humans, 20% of subcapsular portal triads lack portal veins, which could be problematic in small biopsies with few portal triads to evaluate. The moderator reiterated a generalization from his previous visits that the visualization of any lymphatics in the liver is evidence of disease but not specific to any particular one â evaluation of serum proteins, vascular or biliary disease, inflammation, etc. should all be considered, but it should never be considered a normal finding. In cases of microvascular dysplasia, dilated lymphatics is likely the result of decreased venous but normal arterial perfusion, as well as concomitant hypoproteinemia.
References:
1. Cullen JM, Stalker MJ. Liver and biliary system. In: Maxie MG, ed. Jubb, Kennedy, and Palmer's Pathology of Domestic Animals. Vol 2. 6th ed. St. Louis, MO: Elsevier; 2016:266-268.
2. Cullen JM, van den Ingh TSGAM, Bunch SE, Rothuizen J, Washabau RJ, Desmet VJ. Morphological classification of the circulatory disorders of the canine and feline liver. In: WSAVA Standards for Clinical and Histological Diagnosis of Canine and Feline Liver Diseases. St. Louis, MO: Elsevier Limited;2006:51-52; 47-52.
3. van Straten G, Leegwater PAJ, de Vries M, van den Brom WE, Rothuizen J. Inherited congenital extrahepatic portosystemic shunts in Cairn terriers. J Vet Intern Med. 2005;19:321-324.
4. Tobias KM. Determination of inheritance of single congenital portosystemic shunts in Yorkshire terriers. J Am Anim Hosp Assoc. 2003;39:385-389.
5. van Steenbeek FG, Leegwater PAJ, van Sluijis FJ, Heuven HCM, Rothuizen J. Evidence of inheritance of intrahepatic portosystemic shunts in Irish wolfhounds. J Vet Intern Med. 2009;23:950-952.
6. Tisdall PLC, Hunt GB, Bellenger CR, Malik R. Congenital portosystemic shunts in Maltese and Australian cattle dogs. 1994;71(6):174-178.
7. Schermerhorn T, Center SA, Dykes NL, Rowland PH, Yeager AE, Erb HN, Oberhansley K, Bonda M. Characterizaton of hepatoportal microvascular dysplasia in a kindred of Cairn terriers. J Vet Intern Med. 1996;10(4):219-230.
8. Sobczak-Filipiak M, Szarek J, Badurek I, Pdmanabhan J. Trebacz P, Januchta-Kurmin M, Galanty M. Retrostpective liver histomorphological analysis in dogs in instances of clinical suspicion of congenital portosystemic shunt. J Vet Res 2019; 63:243-249.
9. van Steenbeek FG, van den Bossche, Leegwater PAJ, Rothuizen J. Inherited liver shunts in dogs elucidate pathways regulating embryonic development and clinical disorders of the portal vein. Mamm Genome. 2012;23:76-84.