Signalment:
5-year-old, male castrated American quarter horse,
Equus caballusThe horse was used for roping. This horse had moderate, recent weight loss and intermittent reluctance to work on the right hand. The horse was presented for colic that was non-responsive to sedation and anti-inflammatory medications. An abdominal mass and small intestinal distension was diagnosed by palpation and ultrasound. The horse was euthanized due to a poor prognosis.
Gross Description:
The body is in poor body condition (2/9). In the right anteriodorsal quadrant of the abdomen, a multinodular mass, approximately 60 cm in diameter and weighing ~50 pounds invades and compresses the adjacent organs. The mass is in intimate contact with and invades the parenchyma of the right kidney, liver, and pancreas and extends into the mesentery, causing compression of the duodenum. On sectioning, the mass is firm, solid, and mottled white and pale yellow. The neoplasm effaces approximately half of the parenchyma of the right kidney and extends into the dilated renal pelvis of that kidney. Throughout the abdomen, multiple, firm, white, round nodules, ranging from 0.5 to 5 cm in diameter, are attached to or embedded within the mesentery and omentum. The stomach is distended by gas and approximately 2.5 liters of cloudy, green fluid. The lungs and tracheobronchial lymph nodes are diffusely red-pink, wet, and heavy.
Histopathologic Description:
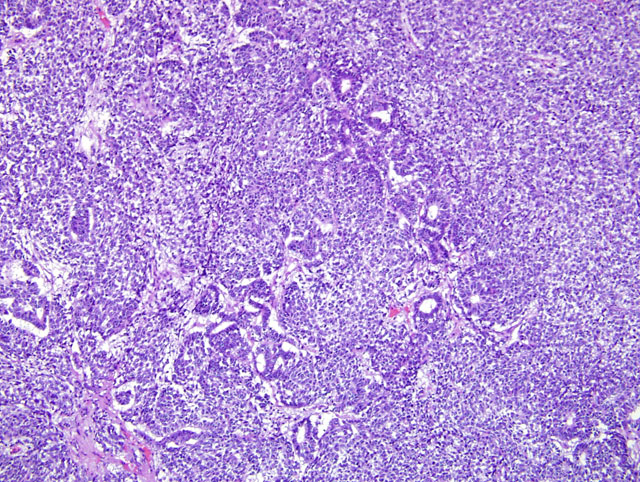
Kidney and liver. The parenchyma of both the kidney and liver is invaded by a well-demarcated, partially encapsulated, expansile and infiltrative neoplasm consisting of haphazardly arranged and densely packed sheets of polygonal to spindle-shaped (blastemal and mesenchymal) cells, and, less commonly, groups of cuboidal to columnar (epithelial) cells that form incomplete tubular structures (Fig. 3-1). Sheets of cells are encapsulated by a fibrous capsule or compressed residual stroma of the kidney and are subdivided by variably thick bands of connective tissue. Cells of the blastemal component are polygonal, have indistinct cell borders, scant, pale, eosinophilic cytoplasm, and a round, hyperchromatic nucleus. The blastemal component blends with spindle-shaped (mesenchymal) cells separated by scant to moderately abundant, fibrillar, eosinophilic (collagenous) extracellular matrix. A loose, myxoid, extracellular matrix is present between spindle-shaped cells in some areas. Less commonly and usually located adjacent to collagenous stroma, cuboidal to columnar cells form tubular structures with indistinct lumens. These cells have scant eosinophilic cytoplasm and often a basally located nucleus. Blastemal and mesenchymal cells are strongly immunopositive for vimentin and are cytokeratin-negative. Approximately 40% of the spindle-shaped cells are immunopositive for desmin and all of the spindle-shaped cells are immunonegative for smooth muscle actin. The cells in the trabeculae of connective tissue between sheets of cells are faintly immunopositive for smooth muscle actin. Trichrome staining demonstrates scant collagen within the sheets of spindle-shaped cells and abundant collagen in the trabeculae between sheets of cells. The cuboidal cells forming tubules and some groups of less organized, polygonal cells are strongly immunopositive for cytokeratin and -negative or faintly -positive for vimentin. Staining with Periodic acid-Schiff demonstrates a scant, discontinuous basement membrane subjacent to some tubular structures. Mitotic figures are 8-9 per 400X field among the blastemal/ mesenchymal component. Anisokaryosis is prominent. The adjacent renal parenchyma is atrophic, with widespread loss of tubules and glomeruli and collapse of the interstitium. The hepatic parenchyma is atrophic with loss of hepatocytes and collapse of portal regions adjacent to the neoplasm.
Lab Results:
24 mg/dl, creatinine 2.1 mg/dl. Abdominocentesis fluid: protein <2.5 g/dl.
Condition:
Nephroblastoma
Contributor Comment:
Nephroblastomas (also called embryonal nephromas in older literature and Wilms tumor in human beings) are theorized to arise from rests of metanephric blastema and usually develop in young animals and children.(1,7,8) Nephroblastomas are rare in horses and most other animal species, except for chickens and swine.(6,12,2,10,5,11,4) The gross and histologic features of nephroblastoma in the horse are rarely described.(6) Nephroblastomas are occasionally diagnosed in adult animals, as in the presented case in a 5-year-old horse. (5,11,4)
Nephroblastomas represent defective nephrogenesis and their component subtypes reflect the conversion of metanephric mesenchymal cells to epithelial structures that occurs during nephrogenesis.(7,8,3) The neoplasm presented here contains all three elements required for the diagnosis of a nephroblastoma: blastemal, mesenchymal, and epithelial, although not evenly represented in the presented section of kidney and in other organs. Immunohistochemical staining of the tissues from this case confirms the coexistence of mesenchymal and epithelial components within the sheets of embryonic cells. Myofibroblastic differentiation was demonstrated by vimentin and desmin immunopositivity. Cells forming tubular structures or located adjacent to trabeculae often were immunopositive for cytokeratin. Other samples of this neoplasm from the kidney, pancreas and liver contain more of the epithelial component, consisting primarily of tubular structures; rudimentary glomeruli were not identified in examined sections from this case. The neoplasm presented here extended to anatomic structures adjacent to the right kidney, but not to the lung or more distant regions of the liver, suggesting coelomic metastasis.
Historically, nephroblastomas have been categorized according to the relative amount of each of the three cellular components, with a triphasic nephroblastoma containing approximately equal amounts of each of the three cell lineages.(7,8) In the neoplasm presented here, cells of all three differentiation types are identified by cytomorphology and using immunohistochemistry, in varying amounts among different regions of the neoplasm. Cells that do not demonstrate cytomorphologic features of mesenchymal or epithelial differentiation, i.e., the blastemal cells, predominate in this neoplasm. In human beings, nephroblastomas that have cytologic features of anaplasia, including enlarged nuclei, hyperchromasia of nuclei, and enlarged, multipolar mitotic figures, are designated as having unfavorable histology in the currently used staging protocol.(7,9)
The genetic pathology that results in Wilms tumor in children appears to be complex, and, in some cases, the development of Wilms tumor in children is associated with other congenital malformations. (1,7) The protein product of the Wilms tumor suppressor gene-1 (WT-1) is a zinc-finger DNA binding protein and an essential regulator of renal development. Inactivation of the WT1 gene is documented in a small number of Wilms tumors in children and is believed to prevent the differentiation of primitive metanephric cells. The remaining Wilms tumors in human beings are assumed to be due to defects in other genes, including WT3 and others. Genetic analysis was not performed on tissue from this case.
JPC Diagnosis:
Kidney; liver: Nephroblastoma, horse, equine.
Conference Comment:
Nephroblastoma is the most common tumor of the kidney in both the chicken and pig.(8) These tumors are less common in calves and dogs and apparently very rare in horses, cats, and sheep. This neoplasm has been found in rats exposed to different tumor producing agents.(8) Metastasis in canine tumors occurred in over 50% of the reported cases, whereas in pigs and calves metastasis is uncommon. In dogs, particularly in German Shepherds, these tumors can form extramedullary, intradural spinal masses usually found between spinal cord segments T10 and L2.(8)
The typical hallmark histologic features of the nephroblastoma are loosely arranged spindle cells amongst primitive glomeruli, haphazardly arranged tubules, and densely cellular blastema. (8) Proportions of these elements vary from tumor to tumor and even within regions of the same tumor. Canine nephroblastomas have been shown to stain for human Wilms tumor gene product C-19.
In the sections examined during conference, the blastemal component compromised the majority of neoplastic cells. The epithelial component, including rudimentary tubules was present multifocally, but glomeruloid structures were not seen. Loose mesenchymal areas were uncommon.
References:
1. DAngio GJ: The National Wilms Tumor Study: a 40 year perspective. Lifetime Data Anal 13:463-470, 2007.
2. Goens SD, Moore CM, Brasky KM, et al.: Nephroblastomatosis and nephroblastoma in nonhuman primates. J Med Primatol 34:165-170, 2005.
3. Grieco V, Riccardi E, Belotti S, Scanziani E: Immunohistochemical study of porcine nephroblastoma. J Comp Path 134:143-151, 2006.
4. Headley SA, Saut JPE, Maiorka PC: Nephroblastoma in an adult sheep. Vet Record 159:850-852, 2006.
5. Henry CJ, Turnquist SE, Smith A, et al.: Primary renal tumors in cats: 19 cases (1992-1998). J Feline Med Surg 1:165-170, 1999.
6. Jardine JE, Nesbit JW: Triphasic nephroblastoma in a horse. J Comp Pathol 114:193-198, 1996.
7. Khoury JD: Nephroblastic neoplasms. Clin Lab Med 25(2) 341-361, 2005.
8. Maxie MG, Newman SJ: Nephroblastoma, Urinary system. In: Jubb, Kennedy, and Palmers Pathology of Domestic Animals, 5
th ed., pp. 501-503. Saunders Elsevier, Philadelphia, PA, 2007.
9. Perlman EJ: Pediatric renal tumors: Practical updates for the pathologist. Pediatric Dev Pathol 8:320-338, 2005.
10. Terrell SP, Platt SR, Chrisman CL, Homer BL, de Lahunta A, Summers BA: Possible intraspinal metastasis of a canine spinal cord nephroblastoma. Vet Pathol 37:94-97, 2000.
11. Yamamoto Y, Yamada M, Nakamura K, et al.: Nephroblastoma with transcoelomic metastasis in a Japanese black bull. J Vet Med Sci 68(8):891-893, 2006.
12. Zoller M, Matz-Rensing K, Fahrion A, Kaup FJ: Malignant nephroblastoma in a common marmoset (
Callithrix jacchus). Vet Pathol 45:80-84, 2008.