Results
AFIP Wednesday Slide Conference - No. 7
- 14 October 1998
-
- Conference Moderator:
Dr. Catherine Wilhelmsen, Diplomate, ACVP
Geo-Centers, Inc.
U.S. Army Medical Research Institute of Infectious Disease
Department of Aerobiology and Product Evaluation
Ft. Detrick, MD 21702-5011
NOTE: Click on images for larger views. Use
browser's "Back" button to return to this page.
Return to WSC Case Menu
- Case I - 4312-98 (AFIP 2643733)
-

- Case 7-1. Gross photo. There is diffuse cyanosis
of both ears.
-
- Signalment: Ten-day-old, mixed breed, commercial piglets.
-
- History: The piglets were from a first litter sow.
Other sows were reported to have had skin lesions that were thought
to be erysipelas and were successfully treated with penicillin.
Gross Pathology: The piglets had red-black discoloration
of the skin of the ears, hind legs, and feet. The skin sections
had dark blood oozing from a layer under the epidermis. The upper
hind leg muscle of one piglet had a well-demarcated, 3 to 4 centimeter,
dark red, infarcted area immediately under the skin. The spleens
of both animals were moderately enlarged. The lungs of one piglet
were diffusely, moderately reddened but not consolidated. The
other internal organs were grossly normal.
Laboratory Results:
1. Serum was negative for Porcine Reproductive and Respiratory
Syndrome virus.
2. Erysipelothrix rhusiopathiae was isolated from: both lungs
(1+ and 4+); spleen (4+); infarcted muscle (4+); and foot (2+).
Contributor's Diagnoses and Comments:
- 1. Moderate necrosuppurative myositis with fibrinoid necrosis
and fibrin thrombosis of blood vessels.
2. Multifocal, moderate hemorrhagic dermatitis with fibrin thrombi
and intravascular bacteria.
-
- There was also moderate, diffuse subacute interstitial pneumonia
consistent with septicemia. Many capillaries contained hyaline
thrombi and rare, gram-positive bacilli. Neither microscopic
lesions, nor gram-positive bacteria were present within sections
of kidney, liver, brain, spleen, and intestine. Gram-positive
bacteria and fibrin thrombi are numerous in some sections of
the skin. The submitted section of muscle from the leg is not
the most severely affected area, but myofiber atrophy, individual
necrotic fibers surrounded by neutrophils, and fibrin thrombi
in the capillaries are present. The more affected muscle section
(not submitted) had more extensive necrosuppurative and hemorrhagic
myositis.
-
- All pigs are susceptible to infection with Erysipelas rhusiopathiae,
but most cases occur between two months and one year of age.
The disease has three forms: acute, subacute, and chronic. Acute
infection has septicemia with disseminated intravascular coagulation
and hyaline thrombi throughout the body. By four days post infection,
the bacteria invade the endothelium, and there is diapedesis
of erythrocytes. The purple skin is usually due to congestion,
and sometimes thrombosis, of dermal vessels. Fibrinoid necrosis
of vessels may be due to a hypersensitivity (Arthus) reaction.
Arteriolar fibrinoid necrosis is thought to be the cause of the
"diamond skin" lesions and may not be present in our
piglets due to the rapid nature of the infection which may not
have allowed enough time for full hypersensitivity vasculitis
to develop. The discoloration of the skin can be used as a prognosticator.
Pigs with pink to red skin lesions usually recover, while those
with dark red-purple lesions usually die, as was our experience.
In another report of acute erysipelas in piglets, the dermal
and hypodermal hemorrhage also occurred mostly on the ears and
limbs.
Muscle degeneration is seen with acute erysipelas, but the locally
severe pattern of hemorrhagic infarction seen in one of our pigs
is unusual. Less specific lesions can be seen in any organ, with
leukothrombi or bacterial emboli. The choroid plexus in the brain
and the ciliary body of the eye are reportedly the most likely
sites to have sequestered bacteria. Synovitis may occur in acute
or chronic disease. The subacute form is similar but less severe
than the acute disease. Chronic infection is characterized by
arthritis or bacterial valvular endocarditis, with the bacteria
localized at these sites.
-
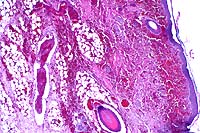 4x
obj
4x
obj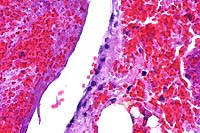 40x
obj
40x
obj
- Case 7-1. Dermis. The 4x view demonstrates a
thrombus within a valved vein (left of center). The dermal connective
tissue is expanded by hemorrhage beneath the epidermis and around
blood vessels. The 40x view shows the thrombus and vessel wall
in which there are vague outlines of bacilli.
 20x
obj
20x
obj
- Case 7-1. Subcutis, skeletal muscle. Several
vessels are occluded by fibrinocellular thrombi and the vessel
walls are disrupted by neutrophils and macrophages. Some skeletal
muscle fibers have hyaline degeneration and fragmentation.
AFIP Diagnoses:
- 1. Haired skin, dermis and subcutis: Thrombosis, fibrinoid
necrosis, and acute vasculitis, multifocal, moderate, with diffuse
hemorrhage, mixed breed, porcine.
2. Skeletal muscle: Thrombosis, fibrinoid necrosis, and acute
vasculitis, multifocal, moderate, with interstitial edema and
myodegeneration.
-
- Conference Note: Erysipelas rhusiopathiae is a small,
gram-positive, non-spore forming, unencapsulated, pleomorphic
bacillus that is the cause of swine erysipelas. Infection has
been reported in several avian and mammalian species, including
humans. The organism is a facultative anaerobe that has a worldwide
distribution and may be found in alkaline soil, decaying organic
matter, and water. The bacterium is resistant to many chemical
and food preservative processes, including salting, pickling,
and smoking, and may remain viable in the environment for up
to several weeks under optimal conditions.
-
- Swine are the most important reservoir hosts, and many pigs
carry the organism in the oropharynx; the organism can be cultured
from the tonsils of clinically healthy pigs. An infected or subclinically
diseased pig is often the source of infection to other herd animals.
The bacterium is shed into the environment, and susceptible pigs
may acquire the infection by ingestion of contaminated soil or
water (most common), percutaneously through skin wounds, or possibly
via ticks and flies. Septicemia develops within 24 hours of exposure
and produces disseminated intravascular coagulation characteristic
of acute disease which may be fatal. Animals surviving the acute
phase develop lesions of subacute to chronic infection, including
cutaneous necrosis, polyarthritis, and endocarditis. Pregnant
sows may abort due to infection, and bacteria have been isolated
from aborted and stillborn fetuses.
-
- Infection with E. rhusiopathiae has been reported in a wide
variety of domestic and wild birds, cattle, sheep, horses, fish,
moose, and dolphins. Erysipelas rhusiopathiae causes polyarthritis
in sheep, and is most often seen in lambs in which the organism
gains entry through docking or castration wounds. The disease
in turkeys is similar to that of swine. In dogs, erysipelosis
is caused by E. tonsillarum, a commensal found in the tonsils
of pigs. Canine erysipelosis is similar to the disease in pigs,
and is clinically characterized by fever, shifting-leg lameness,
and cardiac murmur. In humans, E. rhusiopathiae causes a localized
skin lesion termed erysipeloid which may progress to septicemia
in rare cases. Erysipeloid is characterized by a self-limiting,
painful, red swelling of the fingers, with or without lymphadenopathy.
Human infections are usually acquired through occupational exposure
in meat or chicken slaughterhouses, or fish plants. The term
"erysipelas" in human disease is reserved for infections
caused by Group A beta-hemolytic streptococci.
-
- Contributor: Arkansas Livestock and Poultry Commission,
1 Natural Resources Drive, Little Rock, AR 72205.
References:
- 1. Bastianello S, Spencer BT: A report of swine erysipelas
in a litter of piglets. J South African Veterinary Assoc 55:195-198,
1984.
- 2. Palmer, N: Diseases of bones. In: Pathology of Domestic
Animals, Jubb, Kennedy, Palmer eds., 4th ed., vol. 1, pp. 164-166,
Academic Press, San Diego, 1993.
- 3. Legendre AM: Streptococcal and other gram-positive bacterial
infections. In: Infectious Diseases of the Dog and Cat, 2nd ed.,
pp. 213-214, WB Saunders Co., 1998.
- 4. Jones TC, Hunt RD, King NW: Diseases caused by bacteria.
In: Veterinary Pathology, 6th ed., pp. 435-438, Williams and
Wilkins, 1997.
- 5. Charlton BR: Erysipelas. In: Whiteman and Bickford's Avian
Disease Manual, 4th ed., pp. 101-104, American Association of
Avian Pathologists, University of Pennsylvania, PA, 1996.
- International Veterinary Pathology Slide Bank:
Laser disc frame #08966; 03453; 21206; 20375; 19720; 20476-77.
-
- Case II - VN 87-98 (AFIP 2642427)
-

- Case 7-2. Gross photo. There is a focal area
of pale discoloration affecting 20% of the skeletal muscle (necrosis).
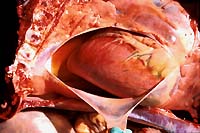
- Case 7-2. Gross photo. Within the pericardial
sac there is 30-50ml of serosanguinous fluid. The epicadium is
pale toward the heart base and contains dull reddish-pink paintbrush
like hemorrhages apically. There are scattered bright red 2-3mm
petechial hemorrhages within the cranial mediastinal and parietal
pleura.
-
- Signalment: Two-year-old, female, quarter horse.
-
- History: On May 13, 1998, three Quarter horses were started
on a new batch of commercial ration. This particular batch was
from a ration targeted for feeder calves. Affected horses were
a six-year-old stallion of 500 kg (horse A), a three-year-old
filly of 350 kg (horse B), and a two-year-old filly of 250 kg
(horse C). Horses A and B died on May 18, 1998, and horse C died
on May 19, 1998. Clinical courses were of 24-48 hours duration
and included sudoresis, hindlimb incoordination which progressed
to forelimb incoordination, muscle weakness, stumbling and death.
The urine of horse A had a dark red discoloration. Horse C was
necropsied. The ration manufacturer was contacted by phone and
confirmed that monensin was added to this particular ration as
a growth promoter for cattle. He stated that the label did not
indicate its use in horses and declined to disclose the exact
amount of monensin present in the ration, but mentioned that
it was the recommended dose for growth promotion in cattle.
Gross Pathology: The main necropsy findings were confined
to the skeletal muscles. Large, patchy areas of white-yellow
discoloration (color transparency A) were present in the heavy
muscle groups (quadriceps femoris, adductor, semitendinosus,
semimembranosus, longissimus dorsi, subscapularis, pectineus,
and gastrocnemius). Abundant yellowish gelatinous edema was present
in the intermuscular fasciae. There was an accentuation of the
hepatic lobular pattern and marked hydropericardium. The myocardium
was diffusely pale (color transparency B).
-
- Laboratory Results: Analysis of two samples of the
ration performed at an independent laboratory revealed 174 ppm
and 180 ppm of monensin.
-
- Contributor's Diagnoses and Comments:
- 1. Skeletal muscle, degenerative myopathy, subacute, moderate
to severe, Quarter Horse, equine. Etiology: Ionophore antibiotic
(monensin) toxicosis.
2. Skeletal muscle, Sarcocystis sp., Quarter Horse, equine (incidental
finding).
3. Liver, centrilobular fatty degeneration, moderate, diffuse,
Quarter Horse, equine (slides not included).
4. Cardiomyopathy, degenerative, toxic, minimal to mild, acute,
Quarter Horse, equine (slides not included).
Ionophore antibiotics are compounds that form lipid-soluble,
dipolar reversible complexes with cations (Ca++, K+, Mg++), enhancing
ionic transport through biological membranes with resultant disturbances
in intracellular ionic homeostasis. The main therapeutic uses
of ionophores include coccidiostasis for several animal species,
growth promotion, and anti-bloat therapy for cattle. A large
spectrum of other therapeutic uses has been described.
-
- The best known ionophores in veterinary practice are monensin,
lasalocid, salinomycin, and narasin. Although these drugs are
reportedly safe when used in target species within the recommended
dosage range, many cases of accidental or intentional poisoning
by ionophores have been reported in horses, cattle, sheep, dogs,
pigs, rabbits, poultry, and ostriches. In general, the hallmark
of toxicosis is degenerative/necrotic myopathy and/or cardiomyopathy.
-
- Factors determining the occurrence of ionophore toxicosis
include overdosage and misuse (e.g., inadequate mixture of the
premix, administration to non-target animal species). The latter
is no doubt the cause of the poisoning of the horses in this
report. Susceptibility to ionophores varies among species: LD50
(mg/kg or ppm) of monensin is 50-80 for cattle and only 2-3 for
horses. The amount of monensin found in the ration in this case
is too high even for cattle (maximum recommended amount as growth
promoter in the ration for cattle is usually 33 ppm).
-
- Clinical signs described for horses vary according to the
location of lesions (skeletal muscle versus myocardium). Usually,
there is profuse sweating, reluctance to move, repeated rising
up and lying down, restlessness, and muscular weakness. Horses
with cardiomyopathy may exhibit ill-thrift, intolerance to exercise,
and sudden death. The dark discoloration of the urine found in
one horse (not necropsied) of this report was most likely due
to myoglobinuria, frequently present in cases of myodegeneration
and also reported in association with ionophore toxicosis.
-
- The gross and histologic lesions found in this case, although
not pathognomonic for the condition, are the ones typically described.
The lesions seen in the skeletal muscles of this case are degeneration
and necrosis. Affected myofiber segments are markedly swollen,
lack normal striation, and are transformed into coagulated proteinaceous
tubes (hyaline necrosis). Frequently, fragmented necrotic segments
of myofibers are seen as irregularly-shaped clumps of eosinophilic
material (floccular necrosis) with invasion of fibers by neutrophils
and macrophages. Edema between fibers is marked. A regenerative
component is also observed characterized by proliferation of
the satellite cells.
-
- Ionophore antibiotic poisoning in horses induces myopathy
and, commonly, cardiomyopathy. Myocardial lesions of the necropsied
horse of this report were only mild. This might be related to
the amount of the toxicant present in the ration. Differential
diagnosis for ionophore antibiotic toxicity should include exertional
rhabdomyolysis (azoturia), coffee senna poisoning, and colic.
-
 20x
obj
20x
obj
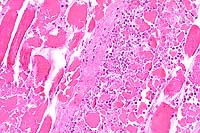
- Case 7-2. Skeletal muscle. Muscle fibers are
hyalinized, fragmented, and have a loss of cross striations,
and are infiltrated and separated by neutrophils, macrophages,
serocellular debris.
-
- AFIP Diagnoses:
- 1. Skeletal muscle: Degeneration and necrosis, diffuse, with
multifocal histiocytic and neutrophilic inflammation, quarter
horse, equine.
2. Skeletal muscle: Sarcocysts, few.
-
- Conference Note: Cases of ionophore antibiotic toxicity
have been reported in numerous species, but monogastric animals,
in particular the horse, are more sensitive than ruminants or
domestic poultry. Monensin alters the membrane transport system
for sodium and potassium, leading to disruption of the electrolyte
modulated calcium gating mechanism. Mitochondrial failure and
energy depletion ensue, with subsequent failure of calcium ion
sequestration from the cytosol. Constantly elevated levels of
cytosolic calcium lead to myofiber hypercontraction and degeneration.
Marked mitochondrial swelling and disintegration are the earliest
ultrastructural changes of monensin toxicity. The heart is particularly
susceptible to disruption of cellular physiology due to its high
energy requirements.
-
- Clinical signs and lesions in the horse are due to cardiac,
skeletal muscle, renal, and hepatic disturbances. Gross and microscopic
lesions are varied depending upon dose and duration of exposure.
In peracute cases, death may occur before gross and histologic
changes become evident, particularly if there is extensive involvement
of the cardiac mitochondria. Complicating the cardiac mitochondrial
disturbance are the derangements in serum electrolyte levels
that can occur due to extensive skeletal muscle necrosis. Skeletal
muscle composes up to 55% of the total body mass of a horse and
is a major reservoir for potassium and phosphorous, while containing
very little sodium, chloride, and calcium. Necrosis of muscle
causes disruption of the boundary between the intracellular fluid
(ICF) and extracellular fluid (ECF), resulting in an efflux of
potassium into the ECF and an influx of water, sodium, chloride,
and calcium. Hyperkalemia, hyponatremia, and hypocalcemia may
follow, causing myocardial conduction disturbances that exacerbate
the disturbance of mitochondria. Influx of significant quantities
of water into necrotic muscle from the ECF may cause hypovolemia,
potentiating the toxic effects of myoglobin on the kidney.
-
- The severity and distribution of lesions also varies among
species. Myocardial lesions reportedly predominate over skeletal
muscle lesions in cattle, the reverse is true in swine, and there
is equivalent severity of lesions in both types of muscle in
chickens and sheep. The long term prognosis for horses surviving
the initial toxic insult probably depends upon the extent of
myocardial lesions. Some horses may suffer from delayed monensin
toxicity. Myocardial fibrosis leads to conduction disturbances
and cardiac arrhythmias, pleural effusion, poor performance,
unthriftiness, muscular weakness, and subcutaneous edema. Signs
may not become evident until the animal is returned to work.
Cardiac abnormalities have been seen in horses three to six months
after toxic exposure.
-
- Differential diagnosis considered by conference attendees
for the skeletal muscle lesions in this horse included ionophore
antibiotic toxicity (monensin), vitamin E/selenium deficiency,
exertional rhabdomyolysis, and ingestion of Cassia sp. plants.
Because the histological lesions in monensin toxicity differ
very little from nutritional or exertional myopathy, clinical
history and feed analysis are critical in the diagnosis. Simultaneous
onset of pronounced clinical signs in multiple, especially adult,
animals suggests exposure to a toxic agent. Owner reports of
recent purchase of a new or "bitter smelling" feed
is an additional helpful historical finding. Vitamin E/selenium
deficiency most often occurs in young foals, commonly involves
the masticatory muscles and tongue, and concurrent steatitis
is often present.
-
- Nutritional myopathy occurs sporadically in older horses,
and steatitis is usually absent. Myocardial lesions may be present
in both foals and adult horses with nutritional myopathy. The
myocardium is infrequently involved in exertional rhabdomyolysis,
and there is usually significant damage to the renal proximal
convoluted tubules secondary to myoglobinuric nephrosis and ischemia.
In addition to the muscle lesions induced by ingestion of Cassia
sp., hepatic lesions are also found, characterized by extensive
hepatocellular degeneration and necrosis.
Contributor: Universidade Federal de Santa Maria, Departamento
de Patologia, 97105-900, Santa Maria RS, Brazil.
-
- References:
- 1. Amend JF, et al.: Equine monensin toxicosis: Some experimental
clinicopathological observations. Comp Cont Ed Pract Vet 11:S173-S182,
1980.
- 2. Boemo CM, et al.: Monensin toxicity in horses. An outbreak
resulting in the deaths of ten horses. Aust Eq Vet 9:103-106,
1991.
- 3. Doonan GR, Brown CM, Mullaney TP, Brooks DB, Ulmanis EG,
Slanker MR: Monensin poisoning in horses - an international incident.
Can Vet J 30:165-169, 1989.
- 4. Hanson LJ, Eisenbeis AB, Givens SV: Toxic effects of lasalocid
in horses. Am J Vet Res 42:456-461, 1981.
- 5. Irigoyen LF, Graça DL, Barros CSL: Intoxicação
experimental por Cassia occidentalis (Leg. Caes.) em eqüinos
[Experimental poisoning by Cassia occidentalis (Leg. Caes.) in
horses]. Pesq Vet Bras 11:35-44, 1991.
- 6. Ordidge RM, Schubert FK, Stoker JW: Death of horses after
accidental feeding of monensin. Vet Rec 104:375, 1979.
7. Rollinson J, Taylor FGR, Chesney J: Salinomycin poisoning
in horses. Vet Rec 121:126-128, 1987.
- 8. Salles MS, Barros CSL, Barros SS: Ionophore antibiotic
(narasin) poisoning in rabbits. Vet Human Toxicol 36:437-444,
1944.
- 9. Hulland TJ: Muscle and tendon. In: Pathology of Domestic
Animals, Jubb KVF, Kennedy PC, Palmer N, eds., 4th ed., vol.
1, pp. 217-244. Academic Press, San Diego, CA, 1993.
- 10. Perkins G, et al.: Electrolyte disturbances in foals
with severe rhabdomyolysis. J Vet Intern Med 12:173-177, 1998.
-
- International Veterinary Pathology Slide Bank:
Laser disc frame #05232 through 05235; 04273.
-
- Case III - 98-746-11 (AFIP 2640640)
-
- Signalment: A third trimester abortus and placenta
from a 5-year-old Swiss Braunvieh cow.
-
- History: Two years ago, several abortions occurred
in the herd of origin. At that time the submitting practitioner
suspected an infection with the bovine virus diarrhea virus (BVDV).
Gross Pathology: Necropsy findings were nonspecific, and
consisted of generalized edema in the subcutaneous tissue and
moderate autolytic changes of fetal organs. Intercotyledonary
areas of submitted placental portions were edematous and thickened.
-
- Laboratory Results: Arcanobacterium pyogenes was isolated
from lung, liver, kidney and placenta. Routinely applied immunohistochemistry
for BVD virus infection was negative. A mixed infection with
Chlamydia sp. was excluded by immunohistochemistry of placental
tissue sections.
Contributor's Diagnoses and Comments:
- 1. Placenta: Placentitis, necrotizing, subacute, multifocal
to diffuse, moderate, with numerous intra- and extracytoplasmic
bacteria.
2. Placenta: Vasculitis, subacute, focal, mild and calcification
of villous mesenchyme, focal to diffuse, mild.
3. Lung: Alveolitis and bronchiolitis, necrotizing, multifocal,
moderate, with numerous intra- and extracytoplasmic bacteria.
4. Lung: Intrauterine asphyxia with aspiration of chorionepithelium
and meconium (bovine abortion due to fetal infection with Arcanobacterium
pyogenes).
5. Intrahepatic cholestasis and extramedullary hematopoiesis
in liver and spleen were additional microscopic findings.
-
- Arcanobacterium (A.) pyogenes (formerly Actinomyces pyogenes
and Corynebacterium pyogenes respectively) is widespread throughout
the world as a common cause of pyogenic infection in a variety
of domestic animals. In cattle, it is one of the most common
causes of sporadic bacterial abortion. Abortions can occur at
any stage of gestation, but are most often observed during the
last trimester and usually occur as a single event in the herd.
-
- A. pyogenes is a small, pleomorphic, gram-positive bacterium
and is a common inhabitant of the nasal, conjunctival, vaginal,
and preputial mucous membranes of clinically healthy cattle.
A. pyogenes is believed to reach the pregnant uterus by a hematogenous
route and produces suppurative endometrial lesions; the fetus
may become septicemic by transplacental transmission. Fetal death
may be caused by hypoxia following placental destruction.
-
- A yellow to brown exudate covering swollen, edematous cotyledons
and marked autolysis are the main gross lesions. The lungs of
infected fetuses less than five months of gestation are dark
red and swollen, and yellow foci are visible on the pleural surface.
-
- Microscopic examination of the placenta reveals a necrotizing,
suppurative placentitis, which is a consistent, but not pathognomonic
lesion. In some cases, large numbers of bacteria can be found
in sections of placenta with minimal changes. Not as consistently
as placental lesions, an acute, fibrinous fetal bronchopneumonia
occurs, and bacterial colonies within these lesions are probably
due to aspiration of contaminated amniotic fluid. Inconsistently,
a fibrinous pericarditis, pleuritis, or peritonitis may be present;
bacteria may colonize in vessels and the surface of the skin
and conjunctiva, with destruction of the epithelium. A. pyogenes
is not often present as a contaminant or commensal in tissues
of aborted fetuses or their placentas, and thus, its presence
is usually of significance. Diagnosis of abortion induced by
A. pyogenes is based on typical lesions and the isolation of
the organism, preferably from the placenta, lung, and abomasal
contents. In the absence of lesions, A. pyogenes induced abortion
can only be suspected.
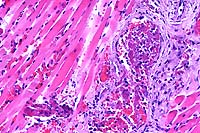
 20x
obj
20x
obj
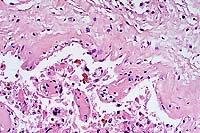
- Case 7-3. Placenta. Trophoblastic cells are detached
from the surface of the fetal placenta and placenal villi. The
intervillus space contains abundant necrotic cellular debris,
scattered yellow-orange pigment (meconium), and occassional macrophages
and neutrophils. Villous connective tissue is thickened and hyalinized.
 40x
obj H&E
40x
obj H&E
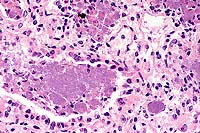
- Case 7-3. Lung. There are abundant coccobacilli
and fewer neutrophils filling scattered bronchioles. Occassional
plump macrophages contain bacteria which compress the nucleus
against one pole of the cell. The interstitium is expanded by
macrophages, fewer lymphocytes and fibroblasts admixed with finely
granular proteinaceous debris.
 40x
obj B&H stain
40x
obj B&H stain
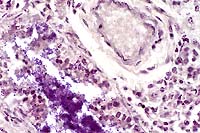
- Case 7-3. Lung. Brown and Hopps staining demonstrate
aggregates of Gram positive bacilli within many of the bronchioles.
Periarteriolar connective tissue is expanded by clear space (edema).
AFIP Diagnoses:
- 1. Chorioallantois: Placentitis, acute to subacute, diffuse,
mild to moderate, with multifocal necrosis, vasculitis, and numerous
intracellular and extracellular bacilli, Swiss Braunvieh, bovine.
2. Lung (fetus): Intrabronchiolar and intra-alveolar bacteria,
histiocytes, amorphous debris, yellow pigment, and epithelial
cells, multifocal, consistent with aspiration of contaminated
amniotic fluid and meconium.
-
- Conference Note: This case was studied in consultation
with the Department of Neonatal and Pediatric Pathology. Conference
participants agreed with the description of the histologic lesions
in the placenta; however, several did not recognize conclusive
evidence of an inflammatory response or pneumonia within the
examined sections of fetal lung. Clumped within alveoli and bronchioles
are abundant intracellular and extracellular bacteria, mononuclear
cells (histiocytes), amorphous debris, small amounts of yellow
pigment (meconium), and a few desquamated epithelial cells. The
clumping of this material, along with the lack of interstitial
inflammatory infiltrates and vascular changes, is consistent
with aspiration of contaminated amnionic fluid and meconium.
In human fetal pathology, the differentiation between aspiration
of infected amniotic fluid and fetal pneumonia is, likewise,
often problematic.
-
- Several conference participants identified prominent, wide
interlobular septa in the fetal lung and speculated whether this
was the result of edema. In humans, the fetal lung has very wide
interstitial septa during the 1st and 2nd trimesters. These septa
remain wider than those of adults throughout the 3rd trimester
and the first months of postpartum life. The expanded pulmonary
interstitium of this bovine fetus likely represents normal development.
-
- Recently, phylogenetic analysis of 13 bacterial species within
the genus Actinomyces was performed; the 16S ribosomal RNA gene
sequences were determined in this study. Based on the results,
the study proposed that Actinomyces pyogenes be assigned to the
genus Arcanobacterium as Arcanobacterium pyogenes comb. nov.
While the results of this genetic analysis are certainly enlightening,
we will refer to the organism throughout the remainder of this
text by its more commonly known name, Actinomyces pyogenes, to
avoid confusion.
-
- Reports of natural infection and experimental evidence suggest
that A. pyogenes may be a cause of sporadic abortion in cattle
and sheep. In most cases, the organism is presumed to reach the
placenta hematogenously. Infected animals may or may not have
signs of systemic illness. Placentitis develops and leads to
placentomal dysfunction, fetal hypoxia, fetal death, and abortion.
An ascending route of placental infection is possible, but is
less likely as this would require disruption of the cervical
plug.
- The fetus may become infected during the progression of placental
lesions, but this is not a consistent feature. In aborted bovine
fetuses in which A. pyogenes has been isolated, colonies of gram-positive
bacteria are often present in the lungs. The bacteria are frequently
confined to the bronchioles and are generally unaccompanied by
inflammation. The gross and microscopic changes in experimentally
infected ovine fetuses are primarily autolytic in nature. Widespread
autolysis is noted grossly in the viscera and musculature, while
microscopic findings include congestion, the presence of bacteria,
and varying degrees of autolysis.
-
- Contributor: Institute of Veterinary Pathology, University
of Zurich, Winterthurerstrasse 286, CH-8057 Zurich, Switzerland.
-
- References:
- 1. Addo PB, Dennis SM: Experimental production of Corynebacterium
pyogenes abortion in sheep. Cornell Vet 69:20-32, 1979.
- 2. Hinton M: Bovine abortion associated with Corynebacterium
pyogenes. Vet Bull 42:753-756, 1972.
- 3. Sorensen GH: Studies on the occurrence of Peptococcus
indolicus and Corynebacterium pyogenes in apparently healthy
cattle. Acta Vet Scand 17:15-24, 1976.
- 4. Ramos CP, Foster G, Collins MD: Phylogenetic analysis
of the genus Actinomyces based on 16S rRNA gene sequences: Description
of Arcanobacterium phocae sp. nov., Arcanobacterium bernardiae
comb. nov., and Arcanobacterium pyogenes comb. nov. Int J Syst
Bacteriol 47:46-53, 1997.
-
- Case IV - X-1555 (AFIP 2642319)
-
- Signalment: 13-year-old, Tennessee walking horse,
male (intact), equine.
-
- History: The stallion had cutaneous lesions of three
months duration on the caudal surfaces of the pasterns of both
rear legs and one front leg. Otherwise, the horse was in good
condition. This stallion was housed individually in a stall and
was infrequently taken out as needed to service mares.
-
- Gross Pathology: The lesions consisted of raised,
coalescing, nodular and papillomatous masses, with multifocal
areas of erosion, crusts, and hemorrhage that covered most of
the distal caudal aspect of the affected pasterns.
Laboratory Results: None.
-
- Contributor's Diagnoses and Comments:
- 1. Chronic, moderate to severe, exudative, papillomatous
dermatitis with intraepidermal bacterial rods and spirochetes,
haired skin, pastern.
2. Mild verminous folliculitis due to Pelodera strongyloides,
haired skin, pastern.
-
- Differential diagnosis for the gross lesions in this case
included: Staphylococcal folliculitis/furunculosis, dermatophilosis,
dermatophytosis, deep dermal mycosis, pythiosis, habronemiasis,
acariasis, sarcoid, papilloma, exuberant granulation tissue,
squamous cell carcinoma, allergic contact dermatitis, contact
irritant dermatitis, and autoimmune disease. In addition to the
epidermal hyperplasia and inflammatory changes visible in H&E
stained sections, there are small to moderate numbers of nematode
larvae in crevices of the hyperplastic epidermis and in hair
follicles (nematodes are not present in all microslides; see
enclosed 2x2 color transparency). Fresh tissue from the lesions
was digested and yielded 3rd stage larvae of Pelodera strongyloides.
-
- Due to similarity grossly and histologically between the
lesions in this horse and papillomatous digital dermatitis (PDD)
of cattle, a modified Steiner stain was applied to histologic
sections of the pastern lesions and revealed numerous spirochetes
within the superficial epidermis (see enclosed 2x2 color transparency).
Numerous gram-negative rods that often form linear associations
are also present; the rods are often visible in the H&E sections.
Transmission electron microscopy of the affected tissue confirmed
the location of both the rods and the spirochetes within epidermal
cells. No viral particles were seen in the electron micrographs.
-
- PDD is a contagious, painful, wart-like digital dermatitis
of unknown etiology in dairy cattle. One of the defining characteristics
of PDD histologically is the presence of an eroded acanthotic
epidermis attended by parakeratotic papillomatous proliferation
colonized by spirochete-dominant bacterial flora. Using molecular
techniques, researchers have recently determined that the spirochetes
associated with PDD are closely related to Treponema denticola,
an oral treponeme of humans that is commonly associated with
human periodontal disease. PDD has been associated with housing
of cattle in persistently wet, unsanitary conditions.
-
- Pelodera dermatitis in the horse has previously been reported,
and it presents as a dry, exfoliative dermatitis with pustules
on the underline and forelegs. Pelodera strongyloides is a free-living
nematode found in decaying organic matter or moist soil. The
nematodes may invade skin and cause dermatitis in animals maintained
on dirty, persistently moist bedding such as soiled straw. The
lesions on the pasterns of the stallion in the present case appeared
more compatible with PDD, and the Pelodera folliculitis was interpreted
to be a concurrent, but slightly less significant, factor in
the pastern dermatitis.
- Additional history revealed that this stallion's stall was
rarely cleaned and was very dirty and moist with urine and feces.
The stallion was treated with IV tetracycline, and an unspecified
ointment was applied topically to the debrided pastern lesions.
The horse's stall was cleaned, and the lesions did not recur.
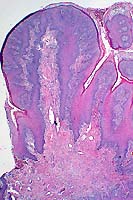 2x
obj
2x
obj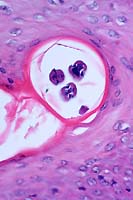 40x
obj
40x
obj
- Case 7-4. The 2x view demonstrates papillomatous to
polypoid character of the epidermis in affected areas. The 40x
view has cross-sections of 3 nematodes within a hair follicle.
An unusual feature of Pelodera (Rhabditis) strongyloides is the
minute double lateral alae extending from each side of the larva.
The musculature is platymyarian, and the gut has a simple low
epithelium (lumen not visible here).
 40x
obj
40x
obj
- Case 7-4. There are multiple rabditiform larva
surrounded by keratin scale.
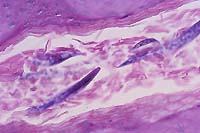
 Steiner
Stain 100x obj
Steiner
Stain 100x obj
- Case 7-4. There are abundant argyrophilic (silver
positive) spirochetes within and between multiple squamous epithelial
cells.
AFIP Diagnoses:
- 1. Haired skin: Dermatitis, proliferative and verrucous,
chronic-active, diffuse, moderate, with multifocal epidermal
hydropic degeneration, and moderate numbers of intraepidermal
filamentous bacilli and argyrophilic spirochetes, Tennessee walking
horse, equine.
2. Haired skin: Intrafollicular and superficial rhabditid nematodes,
few.
-
- Conference Note: The clinical and pathological syndrome
of PDD in dairy cows is incompletely understood, and intense
study is in progress to characterize the disease, its pathogenesis,
and the relationship of the intraepithelial spirochetes to the
cutaneous proliferative lesions. The lesions are often grossly
and histologically indistinguishable from squamous papillomas,
hence the name. Interdigital dermatitis (IDD) is histologically
similar to PDD, but lesions are found in the interdigital space.
Lesions of both PDD and IDD occur on the digits below the level
of the dewclaws, and the hindlimbs are most frequently affected.
-
- While Treponema-like organisms have been isolated from PDD
lesions, their pathological significance is unknown. In a study
of affected California dairy cows4, a few animals had either
deep ulcers or cutaneous lesions consistent with PDD in the flexural
skin folds of the pastern. The lesions differed histologically
from PDD, but the predilection for the same anatomical site under
similar environmental conditions suggests a common pathogenesis.
Deep ulcers or fissures also occur on the lower limbs of cattle
housed in wet, unsanitary conditions. Studies in southern California
dairies strongly linked muddiness of corrals to a high prevalence
of PDD. Poor foot hygiene and prolonged contact of the lower
limbs with manure has been associated with digital dermatitis
in Europe and England. These findings suggest that PDD is multifactorial,
and further study is needed to define the mechanisms and etiologies
of the disease.
-
- The significance of intraepidermal spirochetes in this horse
is uncertain. The unsanitary conditions in which the horse was
housed, coupled with the potential of a multifactorial etiology,
make the pathogenesis of this lesion difficult to determine.
However, the response to treatment, husbandry conditions, anatomical
location, and histological features of the cutaneous pastern
lesion in the horse seem to parallel the syndrome described in
dairy cattle. Continuing research is needed to identify similar
equine cutaneous lesions and to determine whether an entity comparable
to PDD exists in horses.
-
- Contributor: Diagnostic Laboratory Services, College
of Veterinary Medicine, Box 9825, Mississippi State University,
Mississippi State, MS 39762.
-
- References:
- 1. Armed Forces Institute of Pathology Wednesday Slide Conference
1996-1997 (AFIP 2550164). Papillomatous digital dermatitis in
a Holstein cow.
- 2. Choi B-K, et al.: Spirochetes from digital dermatitis
lesions in cattle are closely related to treponemes associated
with human periodontitis. International Journal of Systemic Bacteriology
47:175-181, 1997.
- 3. Farrington DL, Lundvall RL, Greve JH: Pelodera strongyloides
dermatitis in a horse in Iowa. Veterinary Medicine/Small Animal
Clinician 71:1199-1201, 1976.
- 4. Read DH, Walker RL: Papillomatous digital dermatitis (footwarts)
in California dairy cattle: Clinical and gross pathologic findings.
J Vet Diag Invest 10:67-76, 1998.
- 5. Rijpkema SG, David GP, Hughes SL, Woodward MJ: Partial
identification of spirochetes from two dairy cows with digital
dermatitis by polymerase chain reaction analysis of the 16S ribosomal
RNA gene. Vet Rec 140:257-259,1997.
-
- Ed Stevens, DVM
Captain, United States Army
Registry of Veterinary Pathology*
Department of Veterinary Pathology
Armed Forces Institute of Pathology
(202)782-2615; DSN: 662-2615
Internet: STEVENSE@afip.osd.mil
-
- * The American Veterinary Medical Association and the American
College of Veterinary Pathologists are co-sponsors of the Registry
of Veterinary Pathology. The C.L. Davis Foundation also provides
substantial support for the Registry.
- Return to WSC Case Menu

 4x
obj
4x
obj 40x
obj
40x
obj
 20x
obj
20x
obj


 20x
obj
20x
obj
 20x
obj
20x
obj
 40x
obj H&E
40x
obj H&E
 40x
obj B&H stain
40x
obj B&H stain
 2x
obj
2x
obj 40x
obj
40x
obj
 40x
obj
40x
obj
 Steiner
Stain 100x obj
Steiner
Stain 100x obj