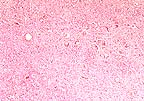
 Neuronal vacuolation
in the brain stem of a Suffolk ewe with scrapie. (HE, 100X, 61K)
Neuronal vacuolation
in the brain stem of a Suffolk ewe with scrapie. (HE, 100X, 61K)Signalment: Three-year-old female Suffolk ovine.
 Neuronal vacuolation
in the brain stem of a Suffolk ewe with scrapie. (HE, 100X, 61K)
Neuronal vacuolation
in the brain stem of a Suffolk ewe with scrapie. (HE, 100X, 61K)
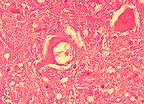 Intraneuronal vacuole characteristic
of scrapie in a 3-year-old a Suffolk ewe. (HE, 400X, 61K)
Intraneuronal vacuole characteristic
of scrapie in a 3-year-old a Suffolk ewe. (HE, 400X, 61K)
History: This animal displayed nervous ataxia with wobbly gait and seemed to be losing condition. The animal was euthanized by the owner.
Gross Pathology: The animal was relatively thin and in poor condition. There was a small abscess (2 cm dia) in the mediastinum.
Laboratory Results: None.
Contributor's Diagnosis and Comments: Brain stem: The sections of brain stem and spinal cord display cytoplasmic neuronal vacuolation.
Scrapie is an afebrile, non-inflammatory, central nervous system degenerative disease of sheep and goats. The incubation period is long, usually ranges from 1-4 years. The disease is recognized in sheep over 3 years of age. There is no humoral or cellular immune response detectable, and the outcome is inevitably fatal.
AFIP Diagnosis: Brain stem: Neuronal vacuolation, multifocal, with mild astrocytosis, and multifocal axonal degeneration, Suffolk, ovine.
Conference Note: Scrapie is a disease with international scope and implications. It is a chronic disease involving the central nervous system of sheep and goats and has an incubation period of 1.5 to 5 years, depending on the age at exposure. Infection usually occurs around the time of birth, and probably via ingestion, but the possibility of vertical modes of transmission remains. The agent initially proliferates within the lymphoid tissues and lower intestine and may take up to 2 years to reach the central nervous system. A further 2 years may elapse before clinical symptoms appear. Early in the disease, animals are alert but excitable, and may have seizures. Later, paresthesia may develop, manifested as agitated rubbing against posts and trees and nibbling at feet and legs. There is progressive dysmetria, emaciation, and finally paralysis and death. Apart from self-trauma, there are no gross lesions, and no inflammatory changes. Histologically, the most characteristic finding is the presence of intraneuronal vacuoles in the CNS.
The identity of the causative agent of scrapie is controversial. A recent theory is that the cause is a "prion" or proteinaceous infectious particle. Other scientists argue that the infectious agent is an as yet unidentified virus. Scrapie is a member of a family of diseases with similar similar clinical and pathologic features. Human diseases in this group include Creutzfeldt-Jakob disease, kuru, Gerstmann-Straussler-Scheinker disease, and fatal familial insomnia. Other related animal diseases include transmissible mink encephalopathy, bovine spongiform encephalopathy, and chronic wasting disease of deer and elk. In all prion diseases, a protease-resistant protein fragment is consistently found. This fragment is derived from a larger protein called PrPSc, by limited proteolysis of the amino terminus. PrPSc is produced by a post-translational conformational change from PrPC. PrPC is a membrane bound protein encoded by a chromosomal gene found in all mammals. PrP gene expression is found in many cells, but is highest in neurons. The function of PrPC is unknown. In studies with knockout mice lacking the PrP gene, the mice suffered no ill effects and did not develop lesions when infected with the scrapie agent. Normal mice develop scrapie-like disease when infected. This suggests that without the normal PrPC protein, disease does not occur. PrPSc seems to be responsible for the central nervous system dysfunction and the neuropathology of prion diseases as it accumulates in neurons. There is evidence that PrPSc accumulation precedes both neuronal vacuolation and astrocytic gliosis, which are the neuropathologic hallmarks of the prion diseases. One interesting discovery is that PrPSc molecules form amyloid plaques composed of PrP polymers.
Contributor: Utah Veterinary Diagnostic Laboratory, Utah State University, 950 East 1400 North, Logan, UT 84322-5700.
References:
1. Jubb KVF, Kennedy PC, Palmer N: Pathology of Domestic Animals,
3rd ed., Vol 1, pp. 380-383, 1993.
2. Foote WC, Pitcher JR: Approaches to controlling scrapie in the United States. Proceedings 92nd Meeting of the United States Animal Association, pp. 402-412, 1988.
3. Timoney JF, Gillespie HJ, Scott FW, Barlough JE: Hagan and Bruner's Microbiology and infectious Diseases of Domestic Animals, 8th Ed., Comstock Publishing Associates, pp. 922, 1988.
4. DeArmond, SJ, Prusiner, SB: Review, Etiology and Pathogenesis of Prion Diseases, Am J Path, 46(4), April, 1995.
International Veterinary Pathology Slide Bank: Laser disc frame #4970, 4971, 7521, 14195, 16984, 24302.
Signalment: Five-year-old male ferret (Mustela putorius furo).
History: The ferret presented with a raised nodule in the region of the tail.
Gross Pathology:. The skin specimen was comprised of a gray gelatinous, semitranslucent, lobulated, subcutaneous mass covered with skin.
Laboratory Results: None.
Contributor's Diagnosis and Comments: Skin, tumor, chordoma.
The tumor is composed of multiple lobules comprised of vacuolated cells with light pink or light blue cytoplasm. Scattered among the vacuolated cells are irregular islands of cartilage and bone. The differential diagnosis includes mucinous chondrosteoma and chordoma. Chordomas are rare in animals with the exception of mink and European ferrets. These tumors are presumed to arise from notochordal remnants.
AFIP Diagnosis: Haired skin, subcutis: Chordoma, ferret (Mustela putorius furo), mustelid.
Conference Note: Chordomas are neoplasms arising from remnants of the notochord and have been reported in the mink, ferret, rat, cat, dog, and human. Human chordomas generally occur in the sacrococcygeal and sphenooccipital areas. In ferrets, chordomas most commonly occur at the tail tip. Two cases of chordoma in the cervical spines of ferrets have been reported.
Microscopically, chordomas of ferrets are described as infiltrative, multilobulated masses with each lobule composed of three components: closely packed vacuolated (physaliferous) cells, cartilage, and bone. The three components are often arranged concentrically, with physaliferous cells surrounding cartilage with a central core of bone. Physaliferous cells contain abundant amphophilic, foamy cytoplasm, and ahve round to oval nuclei. Mitoses are rare. The physaliferous cells are supported by a mucinous matrix of variable thickness that blends gradually with mature cartilage and bone. The bone often contains marrow. Immunohistochemically, physaliferous cells stain strongly positive for both vimentin and keratin, and weakly positive for S-100 protein and neuron specific enolase. Only in ferrets do chordomas contain bone.
Contributor: St. Jude Children's Research Hospital, Comparative Medicine Division/ARC, 332 North Lauderdale, Memphis, TN 38105-2794.
References:
1. Jubb, KVF, Kennedy, PC, Palmer, N: Pathology of Domestic Animals,
4th ed. Volume 1, pg. 429, 1993.
2. Fox, JG: Biology and Diseases of the Ferret., Lea & Febiger, Philadelphia, PA, pg. 284, 286, 287.
3. Dunn, DG, et al.: A histomorphologic and Immunohistochemical Study of Chordoma in Twenty Ferrets (Mustela putorius furo): Vet Pathol 28:467-473, 1991. 4. Williams, BH, et al.: Cervical Chordoma in Two Ferrets (Mustela putorius furo): Vet Pathol 30: 204-206, 1993.
International Veterinary Pathology Slide Bank: Laser disc frame #7940, 19466, 20089, 21585.
Signalment: Three-year-old male Domestic Shorthair cat.
History: The cat initially presented with a mediastinal lymphoma. The cat underwent treatment for lymphoma. Due to lack of improvement while on therapy, the owners elected to have the cat euthanized.
Gross Pathology: The lung had depressed gray areas distributed throughout the upper lobes.
Laboratory Results: None.
Contributor's Diagnosis and Comments: Lung: verminous pneumonitis. Lung, pulmonary artery: medial hypertrophy. Lung: pulmonary nematodiasis.
Etiology: Aelurostrongylus abstrusus.
Alveoli and alveolar ducts in a large area of lung parenchyma are distended with nematode larvae and embryonated eggs. Some alveoli are disrupted due to overexpansion with the larvae. The parasite larvae are compatible with Aelurostrongylus abstrusus.
The cause of medial hypertrophy of the pulmonary arteries of cats has been a source of contention. It has been associated with infection with Aelurostrongylus abstrusus and Toxocara cati, but others report similar arterial changes in specific pathogen-free cats.
AFIP Diagnosis: Lung: Pneumonitis, subacute to chronic, multifocal, minimal, with numerous metastrongylid nematode eggs and larvae, bronchiolar smooth muscle hyperplasia, submucosal gland hyperplasia, and marked arterial medial hypertrophy and hyperplasia, Domestic Shorthair, feline.
Conference Note: Aelurostongylus abstrusus is a common metastongylid lungworm of cats. The genus is ovo-viviparous, the eggs hatching in the air passages. The L1 larvae are passed in the feces, enter a mollusc (snail or slug) intermediate host where they develop to L3 larvae in approximately 18 days. The mollusc may be eaten by birds, rodents, frogs, and lizards, which will carry the L3 larvae as paratenic hosts. The cat is infected by ingestion of the paratenic host or the mollusc. The L3 larvae migrate to the lung parenchyma and small bronchioles to complete the life cycle.
The pulmonary lesions are quite characteristic, usually being in the form of 1-10 mm diameter nodules, which represents nests of eggs and larvae. These nodules are yellowish and firm, scattered throughout the parenchyma but more common in the peripheral lung. A small amount of creamy exudate can be expressed from each nodule. Severe, confluent consolidation can produce clinical signs of chronic coughing and weight loss. Histologic lesions include dense collections of mononuclear cells, and occasional giant cells especially around degenerate larvae. There is often bronchiolar smooth muscle hyperplasia, catarrhal and eosinophilic bronchiolitis, hyperplasia of submucosal glands and smooth muscle hypertrophy and hyperplasia within pulmonary arterial walls. The granulomatous alveolitis and catarrhal bronchiolitis gradually regresses. However, the hypertrophy and hyperplasia of the smooth muscle in arteries, bronchioles and alveolar ducts persists.
Contributor: St. Jude Children's Research Hospital, Comparative Medicine Division/ARC, 332 North Lauderdale, Memphis, TN 38105-2794.
References:
1. TC Jones and RD Hunt: Veterinary Pathology, pg. 819-821; pg.
1285.
2. Jubb KVF, Kennedy PC, Palmer N: Pathology of Domestic Animals, 3rd ed., Vol 2, pp. 684-685, 1993.
International Veterinary Pathology Slide Bank: Laser disc frame #4873, 6493, 6494, 8264.
Signalment: 1«-year-old male Hovawart, canine.
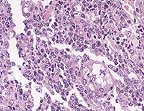 Cryptococcus neoformans
yeasts contained within splenic macrophages. (HE, 200X, 118K)
Cryptococcus neoformans
yeasts contained within splenic macrophages. (HE, 200X, 118K)
History: The dog was presented to a veterinarian with enlarged mandibular lymph nodes but good general condition. In the following weeks a progressing lameness of the right forelimb was observed. On X-ray, there was a periosteal reaction at the right carpus. Antiphlogistic and antibacterial treatment were without success. During the next days, the dog developed elevated body temperature (39-40 C) and became increasingly apathetic and cachectic. A mass (1 cm in diameter) at the upper right canine, extending into the nasal cavity and the dorsum nasi, was noticed. An aspirate from this mass was examined cytologically, and an infection with Cryptococcus neoformans was suspected. The dog was submitted to a small animal clinic for further treatment. The clinic confirmed the diagnosis of the local veterinarian. Additionally, a mild exophthalmus and strabismus divergens of the left eye was noticed. After ophthalmologic examination, a granulomatous chorioretinitis was suspected. By ultrasound investigation, a mass in the left orbita with lysis of the adjacent bone was observed. The dog was euthanized because of poor prognosis.
Gross Pathology: At necropsy, yellowish, soft masses were seen throughout the whole nasal cavity extending from the conchae to the ethmoid bone, infiltrating the dorsum nasi, the palatum durum, the left orbita and left side of the sinus frontalis where gelatinous, white masses were seen. Similar masses were noticed at the optic chiasma. The macroscopic examination of the eyes revealed several white foci in the fundic region. Multifocally, the retina had a greyish appearance. At the right distal radius a yellowish mass (several cm in diameter) was noticed, destroying the compacta and extending into the medullary cavity. Both mandibular lymph nodes were enlarged. The cut surface of the spleen had numerous white glassy foci of several millimeter in diameter.
Laboratory Results: Cryptococcus neoformans was cultured from a nasal aspirate and identified by its typical morphology.
Chonchae nasalis: severe pyogranulomatous rhinitis with numerous yeast-like intracellular and extracellular microorganisms, findings indicative for cryptococcosis.
Spleen: multifocal moderate granulomatous splenitis with yeast-like-intracellular and extracellular microorganisms, findings indicative for cryptococcosis.
Contributor's diagnosis & Comments: The yeast Cryptococcus neoformans belongs to the family of Cryptococcaceae. The organism is 3.5-7 m in diameter with a gelatinous capsule of 1-30 m. Infections with Cryptococcus neoformans are rare in Europe. Cats are most often infected but all mammals are susceptible to this infection. The yeast is found in soil, grass, dust and avian excreta. Inhalation is thought to be the most frequent route of infection.
Humans who acquire the infection are often immunocompromised. In a study performed in the U.S., the majority of dogs with cryptococcosis, however, appeared immunocompetent (Berthelin et al, 1994).
The yeast has a predilection for the CNS. Systemic infections, as in the present case, are rare. The dog presented here had granulomatous lesions with microscopically detectable organisms in nasal cavity, spleen, lung, mandibular lymph nodes, cortex and medulla of the radius, leptomeninx extending along the optic nerve to the orbita, and granulomatous lesions were also found multifocally in the retina of both eyes.
Systemic C. neoformans infection with severe granulomatous reaction is thought to be due to organisms with a small or even without gelatinous capsules (Lichtensteiger and Hilf 1994). These organisms can be phagocytized by macrophages. Thus, a reactive cellular response is induced. In the present case, a capsule was seen around many but not all organisms.
AFIP Diagnosis: 1. Nasal conchae: Rhinitis, granulomatous, multifocal, severe, with numerous yeast, etiology consistent with Cryptococcus neoformans, Hovawart, canine. 2. Spleen: Splenitis, granulomatous, multifocal, mild, with numerous yeast, etiology consistent with Cryptococcus neoformans.
Conference Note: Participants agreed that the morphology of the budding yeast is consistent with Cryptococcus neoformans. In H&E stained sections, C. neoformans appears thin-walled, slightly basophilic and spherical, with single narrow-based buds. The fungus is surrounded by a wide clear zone; the shrunken capsular material stains positively with mucicarmine and the fungal wall stains with PAS. In some sections, scattered foci of osteonecrosis of the turbinates were noted.
Cryptococcosis occurs worldwide in many species, but cats are affected most frequently. Cats that are immunologically compromised, such as those infected by feline immunodeficiency virus or feline leukemia virus, those treated with corticosteroids or those that are malnourished, are most susceptible. Infection is acquired in most instances through inhalation of contaminated dust, with localization in the nasal turbinates or lungs sometimes followed by systemic dissemination. Cryptococcal mastitis in cows is believed to be initiated by local inoculation. In cases of systemic infection, a broad spectrum of lesions and clinical signs have been reported; however, rhinitis, pneumonia, ulcerative skin lesions, encephalitis and ocular infections are the most prevalent. The pulmonary lesion is a multifocal granulomatous pneumonia. CNS infection may involve the meninges and neuropil. The characteristic gross appearance of cryptococcal lesions is that of small, gelatinous, white foci. The microscopic features of the infection vary from little cellular reaction and many organisms (gelatinous form) to a more granulomatous reaction characterized by macrophages, giant cells, lymphocytes and a fibrous stroma.
Contributor: Institut fur Pathologie, Tierztliche Hochschule, Hannover, Bnteweg 17, 30559 Hannover, Germany.
International Veterinary Pathology Slide Bank: Laser disc frame #2580, 5081, 6968, 14436, 19344.
* The American Veterinary Medical Association and the American College of Veterinary Pathologists are co-sponsors of the Registry of Veterinary Pathology. The C.L. Davis Foundation also provides substantial support for the Registry.