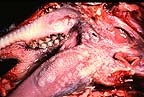
 Multifocal erosions and ulcers
in the oral cavity of a goat infected with peste des petits ruminants
virus. (38K).
Multifocal erosions and ulcers
in the oral cavity of a goat infected with peste des petits ruminants
virus. (38K).Signalment: 7-month-old mixed breed goat.
History: The goat was experimentally inoculated with 1.0 ml of goat blood stabilate containing peste des petits ruminants virus (Meilig-Sudan, also called Sudan 72-2 strain). The goat became febrile and depressed 5 days following inoculation. Subsequently, there was some diarrhea and crusting of nasal and oral mucocutaneous junctions. At 10 days postinoculation, the goat became quite dyspneic and died.
Gross Pathology: Postmortem findings included extensive oral erosions and a generalized pneumonia consisting of multifocal consolidation most prominent in the ventral areas.
Laboratory Results: None submitted.
Contributor's Diagnosis and Comments: Pneumonia, bronchointerstitial, necrotizing, proliferative, severe, extensive, subacute, with syncytial cells and intracytoplasmic and intranuclear viral inclusion bodies.
Etiology: Peste des petits ruminant virus (Genus Morbillivirus, Family Paramyxoviridae).
Peste des petits ruminants (PPR) is a viral disease of goats and sheep characterized principally by stomatitis, diarrhea, oculonasal discharge, and pneumonia. Originally described in West Africa, the disease is now known to be responsible for considerable morbidity and mortality throughout sub-Saharan Africa north of the equator, the Arabian peninsula, and India. The causative agent is closely related to rinderpest virus and these two are classified along with the viruses of measles, canine distemper, and phocine distemper in the genus Morbillivirus in the Family Paramyxoviridae.
There are many similarities between PPR and rinderpest. Both diseases are characterized by a severe enterocolitis. Although an erosive stomatitis is often present in rinderpest, it is a much more consistent and dramatic feature in PPR. Also, animals infected with PPR often develop pneumonia, a lesion that is not reported in rinderpest.
Histologically, PPR is characterized by necrosis of oral and intestinal epithelium and a generalized lympholysis. The pneumonia which occurs with PPR, like that associated with canine distemper or measles, is "bronchointerstitial", with disorganization of bronchiolar epithelium, and hyperplasia of type II alveolar cells. Although multinucleate syncytial cells may be see in many organs, they are most numerous in lung. Eosinophilic viral inclusion bodies may be either nuclear or cytoplasmic and are present in a variety of cell types.
AFIP Diagnosis: Lung: Pneumonia, bronchointerstitial, subacute, diffuse, severe, with bronchiolar epithelial and type II pneumocyte hyperplasia, syncytial cells, and eosinophilic intranuclear and intracytoplasmic inclusion bodies, mixed breed goat, caprine, etiology consistent with peste des petits ruminants virus.
Conference Note: Peste des petits ruminants virus is thought to be transmitted by direct contact, the virus being very sensitive to environmental degradation. The virus enters via the nasopharyngeal mucosa and replicates in the palatine tonsils and regional lymph nodes. After a 10-day incubation period, the virus is disseminated hematogenously within lymphocytes. The virus then replicates in lymphoid tissues, respiratory and gastrointestinal epithelium, and bone marrow. Recovered animals do not appear to be carriers.
Moribilliviruses possess two proteins which facilitate binding to host membranes, hemagglutinin and F protein. The F protein mediates fusion of the viral envelope with the cellular membrane and assists in viral attachment. It also causes host cell fusion and is responsible for the formation of syncytial cells. The ability to fuse host cells allows the virus to spread without being exposed to antibody. To be biologically active the F protein must be cleaved by a host protease into two disulfide-linked polypeptides, F1 and F2. If a host cell lacks the necessary proteases, the virus formed is not infectious, since the F protein is required for viral attachment.
Contributor: Foreign Animal Disease Diagnostic Laboratory, USDA P.O. Box 848, Greenport, NY 11944.
References:
1. Bundza A, Afshar A, Dukes TW, Myers DJ, Dulac GC, Becker SAWE. Experimental peste des petits ruminants (goat plague) in goats and sheep. Canadian Journal of Veterinary Research 52:46-52, 1988.
2. Brown CC, Mariner JC, Olander HJ. An immunohistochemical study of the pneumonia caused by peste des petits ruminants virus. Veterinary Pathology, 28:166-170, 1991.
3. Barker IK, Van Dreumel AA, and Palmer N: The Alimentary System in Pathology of Domestic Animals. Jubb KVF, Kennedy PC, and Palmer N eds., Academic Press, Inc., San Diego, 4th ed, Vol 2, pp. 159-163, 1993.
Signalment: 6-month-old cross-bred lambs.
History: 15 lambs were found dead and a further 30 appeared ill seven days after they were introduced to a paddock containing abundant Amaranthus hybridus plants but little other feed.
Gross Pathology: Two sheep were necropsied. There was abundant clear fluid in thoracic and abdominal cavities and marked perirenal edema. Kidneys were swollen, pale, and had pinpoint hemorrhages over the capsular surface.
Laboratory Results:
BIOCHEMISTRY
(Results Normal)
T. bili m/l 1.5 0-6.8
T. Protein g.l 64 55-80
Alb g/l 35 26-36
Glob g/l 29 30-57
ALB/Glob 1.19 0.5-1.1
AST U/L 268 0-90
GGT U/L 57 <40
GLDH U/L 23.6 <20
Creat mM/L 15.89 0.6-2.65
Urea mM/L 105.5 2.9-7.1
Na mM/L 127 139-152
k mM/L 7.7 3.9-5.4
CO2 mM/L 10.1 26-34
Contributor's Diagnosis and Comments:
Kidney: Nephrosis, tubular, subacute, extensive, severe.
Etiology: Amaranthus hybridus poisoning.
There is a diffuse degeneration and attenuation of the epithelium of proximal and distal tubules. There are many intratubular proteinaceous casts and some necrotic cell debris in collecting ducts. There are a few foci of early tubular regeneration.
Mortalities were probably associated with cardiac arrest due to electrolyte imbalances, including hyperkalemia, resulting from acute renal failure. There are no previous documented reports of Amaranthus hybridus causing renal failure in sheep.
Reports of toxicity following the grazing of the closely related species Amaranthus retroflexus (redroot) have been associated with severe perirenal edema and nephrosis in cattle and pigs. The nephrotoxic principle in Amaranthus has not yet been described. Phenolic compounds which have been identified in the leaves of Amaranthus retroflexus may be involved in the syndrome. The abundance of Amaranthus hybridus on otherwise bare ground had resulted from its rapid germination after 7 months drought, then a good fall of rain. Oak shrubs and trees (Quercus spp.) cause a similar condition in ruminants and horses; in this case the toxic principle has been shown to be tannins.
AFIP Diagnosis: Kidney: Nephrosis (tubular epithelial necrosis), subacute, diffuse, moderate, with hyaline and granular casts, hyaline droplet change, and mild regeneration, cross-breed, ovine.
Conference Note: Redroot pigweed (Amaranthus retroflexus) has been reported as a cause of perirenal edema and acute renal failure in swine, cattle, and, less frequently, horses. Grossly, Amaranthus intoxication produces perirenal edema and occasionally edema of the intestine, mesentery, and ventral abdominal wall. Ascites and hydrothorax may also develop. The kidneys are pale, but usually not enlarged. Histologically, there is degeneration and necrosis of tubular epithelium. Glomerular hypercellularity and necrosis may also be present; in this case, some of those attending the conference noted multifocal necrosis of cells within glomerular tufts. Another finding in this case is the presence of hyaline droplets, eosinophilic homogeneous globules of protein, within the cytoplasm of tubular epithelial cells. Hyaline droplets are lysosomes distended by protein that is being degraded for return to the circulation as amino acids. The presence of hyaline droplets indicates that the process of protein resorption by proximal tubules is saturated.
The primary differential diagnosis for Amaranthus intoxication is oak (Quercus spp.) poisoning. Ingestion of acorns, leaves, and buds of these shrubs and trees causes renal tubular degeneration and necrosis, perirenal edema, and ulceration of the intestinal tract. The toxic substances are gallotoxins that are metabolized to tannic acid, gallic acid, and pyrogallol, which are the active toxic metabolites. In the case of oak poisoning, the edema is caused by endothelial damage induced by binding of tannins to these cells. The intestinal lesions are due to the ability of tannins to bind peptide linkages and precipitate protein.
Contributor: Regional Veterinary Laboratory, Wollongbar, NWS 2477, Australia.
References:
1. Maxie MG (1993) The urinary system. In Pathology of Domestic Animals (Vol 2), ed Jubb KVF, Kennedy PC and Palmer N, 4th Edn. Academic Press, New York, PP 493-494.
2. Everest SL (1981) Poisonous Plants of Australia. Angus and Robertson, Sydney, pp 66-71.
International Veterinary Pathology Slide Bank: Laser disc frame #9756.
Signalment: 30-week-old female TG.NK (neu) transgenic mouse.
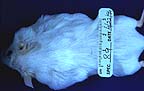 Multiple cervical and thoracic
subcutaneous nodules in a female TG.NK(neu) transgenic mouse.
(18K)
Multiple cervical and thoracic
subcutaneous nodules in a female TG.NK(neu) transgenic mouse.
(18K)
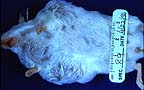 Multiple cervical and thoracic
subcutaneous nodules in a female TG.NK(neu) transgenic mouse.
(21K)
Multiple cervical and thoracic
subcutaneous nodules in a female TG.NK(neu) transgenic mouse.
(21K)
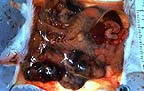 Multiple firm white foci of
Type B mammary carcinoma in a female TG.NK(neu) transgenic mouse.
(27K)
Multiple firm white foci of
Type B mammary carcinoma in a female TG.NK(neu) transgenic mouse.
(27K)
History: The mouse was sacrificed during a short-term study to examine the anti-carcinogenic effects of vitamin A and vitamin A analogues.
Gross Pathology: Bilateral, subcutaneous nodular masses developed in the ventrolateral cervical and thoracic regions over a period of 4 weeks.
Laboratory Results: None submitted.
Contributor's Diagnosis and Comments: Mammary gland, cervical and thoracic - Adenocarcinoma type B.
Essential microscopic features are those of a circumscribed, non-encapsulated, multi-lobulated, nodular mass effacing the gland. Lobules are composed of a uniform population of cells forming densely cellular sheets and thick cords which surround multiple, variably-sized spaces that are either empty or contain variable amounts of blood and/or fibrin, proteinaceous secretory material and degenerate cells. In some areas, the cells form poorly-defined tubular structures. Cells are polygonal with indistinct margins, abundant eosinophilic cytoplasm and oval to round nuclei with clumped chromatin and occasionally 1-2 prominent nucleoli. Mitotic figures number 6 to 12 per HPF. Focal areas of necrosis occur within lobules.
The currently accepted classification of spontaneous mouse mammary gland neoplasms is that of Dunn. Primary histologic types identified in laboratory mice include adenocarcinoma types A, B, C and adenoacanthomas. Type A adenocarcinomas are composed of well-differentiated, uniform, small diameter acini or elongate tubules. Type B adenocarcinomas are highly pleomorphic. Morphologic patterns such as acinar, cystic (hemorrhagic), papillary, solid sheets, nests or islands of neoplastic cells can be present in variable combinations with very little or no glandular differentiation. Type C tumors are composed of multiple uniform small cysts lined by cuboidal epithelial cells. Adenoacanthomas are characterized by variable combinations of glandular and squamous differentiation with marked keratinization and are the most common chemical-induced mammary gland neoplasm in mice. Although not particularly invasive, these neoplasms may metastasize to the lung.
The development of mouse mammary neoplasms can be influenced by several factors which include the presence of the mouse mammary tumor virus (MMTV), strain of mouse, hormones, and exposure to carcinogens. Mouse strains infected with the milk transmitted MMTV type B retrovirus or Bittner agent, have a high incidence of mammary gland neoplasms with an early age of onset. The majority of the neoplasms are type-A or type-B adenocarcinomas. Mouse strains such as the C3H, RII and DBA and some derivatives carrying the MMTV are high incidence strains, whereas MMTV-free or resistant strains such as the BALB/c, C57BI and B6C3F mice have a low spontaneous incidence of late onset mammary neoplasms. Hormonal stimulation (estrogen, somatotropin or mammotropin) appears to be essential for MMTV carcinogenic activity in the mammary gland. Mammary neoplasms may also be induced by physical or chemical agents and development also appears to be dependent upon hormonal stimulation.
The neu, ras and myc oncogenes have been implicated in the development of human breast neoplasms. Transgenic mice bearing the C-neu (TG-NK), v-HA-ras (TG-SH) or c-myc (TG-M) oncogenes under the regulatory control of the mouse mammary tumor virus (MMTV) long terminal repeat (LTR) develop a high incidence of mammary gland adenocarcinomas. Most of the neoplasms can be classified using the standard Dunn classification of mammary gland neoplasms and appear to be type A or type B adenocarcinomas. The relevance of the transgenic mouse model to human breast neoplasia has been suggested by the detection of: 1) B particles similar to those of murine MMTV in the milk of women; 2) neutralizing antibodies to MMTV in the sera of human breast cancer patients; 3) a 70S reverse transcriptase in the milk of women; and 4) an RNA homologous to murine MMTV in human breast cancer cells. Furthermore, c-myc,c-ras, and c-erb oncogenes have been found to be over-expressed and/or amplified in human and murine breast cancer. A cellular oncogene, int-2, that frequently contains the MMTV integrated into adjacent cellular sequences in MMTV-induced carcinomas, is also over-expressed and amplified in human breast cancer.
AFIP Diagnosis: Mammary gland: Mammary adenocarcinoma type B, TG.NK (neu) transgenic mouse, rodent.
Conference Note: Although Dunn's classification system for mouse mammary tumors has long been accepted, there are changes being proposed by the Society of Toxicologic Pathologists and the World Health Organization. The final proposal by the international expert panel for the Standardized System of Nomenclature and Diagnostic Criteria (SSNDC), classifies mouse mammary tumors as either adenocarcinoma, fibroadenoma, or adenoacanthoma, malignant.
Utilizing the SSNDC criteria, mammary adenocarcinoma is defined as arising from mammary gland epithelium. There is loss of lobular architecture, invasion of surrounding tissue, cellular pleomorphism, and frequent mitoses. These tumors may have a variety of histologic patterns, many of which may occur within the same tumor, including tubular, papillary, cystic, solid, comedo, and undifferentiated. The glandular components of these tumors may be associated with a myoepithelial component.
Fibroadenomas are composed of mammary gland epithelium and connective tissue. They are well circumscribed with discrete ducts and/or alveoli within a dense fibrous stroma.
Malignant adenoacanthomas arise from mammary gland epithelium. These tumors may be well circumscribed and have glandular and epidermoid differentiation. Squamous differentiation is usually well-defined and exceeds 25% of the tumor. Squamous differentiation may also occur in mammary adenocarcinoma; however, it composes less than 25% of the tumor.
Contributor: National Institutes of Health, National Institute of Environmental Health Sciences, P.O. Box 12233, Research Triangle Park, NC 27709.
References:
1. Cardiff RD, E Sinn, W Muller and P Leder. Transgenic Oncogene Mice: Tumor Phenotype Predicts Genotype. Am J. Pathol., 139;3, 495-501 (1991).
2. Squartini F, and R Pingitore. Tumors of the Mammary Gland. In International Agency for Research and Cancer (WHO). Pathology of Tumors of Laboratory Animals: Tumors of the Mouse, eds. V Tursusov and U Mohr. Vol. 12, second edition, Lyon 1994.
3. Tennant RW, GN Rao, A Russfieid, S Seikop and AG Braun. Chemical Effects of Transgenic Mice Bearing Oncogenes Express in Mammary Tissue. Carcinogenesis 14:1 29-25, (1993).
International Veterinary Pathology Slide Bank: Laser disc frame #13885, and 23892-3.
Signalment: Free-ranging juvenile (young of the year) male bobcat (Lynx rufus).
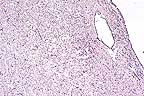 Demyelination in the brainstem
of a bobcat with canine morbillivirus infection. (HE, 40X, 42K)
Demyelination in the brainstem
of a bobcat with canine morbillivirus infection. (HE, 40X, 42K)
History: A bobcat displaying incoordination was found by a hunter in the woods close to the town of Fredericton, New Brunswick, on November 13, 1993. It was presented to a local veterinary clinical where it was kept under observation with supportive treatment for approximately one week. During that time, the animal showed progressively worsening nervous signs, including delayed pupillary light reflexes, head tilt, ataxia, and, terminally, inability to stand. It was euthanized by intravenous injection of barbiturates.
Gross Pathology: The animal had good internal fat reserves. The only gross finding consisted of multiple small white nodules within the liver, many of them surrounded by a thin zone of hemorrhage.
Laboratory Results: Hematology and a general blood chemistry panel were done. As compared to normal values in the domestic cat, the only abnormal finding was a marked lymphopenia (0.225 x109/L).
Bacteriology: No significant bacterium was isolated from the lungs, liver and kidneys.
Contributor's Diagnosis and Comments: Encephalomyelitis, demyelinating,
severe.
Etiology: Morbillivirus (canine distemper virus).
Microscopic lesions in the slide provided (cerebellum and medulla oblongata) are characterized by severe demyelination and edema, some neuronal necrosis, and diffuse gliosis but a relatively mild inflammatory reaction. Intranuclear inclusion bodies were seen in a few cells (mainly astrocytes) in the more severely affected areas. The cerebral hemispheres (mainly thalamic area) and spinal cord (cervical and lumbar regions) contained similar but less extensive lesions. With the use of a monoclonal antibody to the distemper viral nucleocapsid, Dr. Brian A. Summers (Department of Pathology, College of Veterinary Medicine, Cornell University) identified strong positive immunocytochemical staining in several foci of the brain. The hepatic lesions seen grossly were granulomas, some of which contained cestode larvae. Microscopic lesions were not seen in the lungs.
Disease and death caused by canine morbillivirus occurred among captive large felids in California in 1991 and 1992 and in free-ranging lions in the Serengeti region of Tanzania in 1994. This case may be the first one recognized in a free-ranging wild felid in North America. It contrasts with the cases in California, in which the brain lesions were often mild and patchy.
AFIP Diagnosis: Medulla oblongata and cerebellum: Demyelination, multifocal, severe, with edema of myelin sheaths, axonal degeneration, gliosis, minimal lymphohistiocytic meningoencephalitis, and eosinophilic intranuclear and intracytoplasmic inclusion bodies, bobcat (Lynx rufus), felid, etiology consistent with canine distemper virus.
Conference Note: Canine distemper virus (CDV) is a member of the genus Morbillivirus that also includes measles virus, rinderpest virus, peste des petits ruminants virus and phocine distemper virus. Cetacean morbilliviruses (either two closely related viruses or two stains of the same virus) have also been recognized recently. CDV is transmitted by the respiratory route and reaches the brain in circulating lymphocytes, platelets, and mononuclear cells. Viral infection initially develops in meningeal macrophages and then spreads to neurons, ependymal, and glial cells. In dogs, histopathologic changes can occur in grey and white matter; however, one usually predominates over the other. In grey matter disease, there is neuronal degeneration, gliosis, and lymphoplasmacytic inflammation in the cerebral and cerebellar cortices, basal nuclei, brain stem, and spinal cord. Satellitosis and neuronophagia affect necrotic neurons. Loss of the neuron and it's axonal processes induces secondary demyelination. Nonsuppurative inflammation often develops and includes the overlying meninges.
In white matter disease, the lesions are multifocal and numerous. The lesions are a mix of primary demyelination, sparing the axons, and secondary demyelination following degeneration of the axon. Early lesions consist of gliosis which later progresses to nonsuppurative inflammation. Intranuclear and intracytoplasmic inclusion bodies can be found in astrocytes, ependymal cells, microglia, and neurons. Astrocytes and ependymal cells occasionally form syncytia. The lesions in the present case closely resemble the white matter lesions of CDV in dogs.
The pathogenesis of primary demyelination due to canine distemper infection is unknown. Proposed mechanisms include viral infection and loss of oligodendrocytes; loss of infected astrocytes which may deprive oligodendrocytes of a nutritive or trophic factor necessary for survival; or an immune mechanism that is responsible for the selective destruction of myelin. The degree of demyelination in this bobcat is similar to that often found in dogs. Neural lesions described in CDV-infected large felids (lions, tigers, leopards, and jaguars) were less severe than those of dogs; however, like canids, the large cats had gastroenteritis and/or proliferative interstitial pneumonia with syncytial cells, findings absent in this bobcat.
Contributor: Atlantic Veterinary College, University of Prince Edward Island, Charlottetown, PEI, Canada C1A-4P3.
References:
1. Summers BA, Cummings JF, de Lahunta A: Veterinary Neuropathology, Mosby, St. Louis, pp 102-110, 1995.
2. Appel MJG, Yates RA, foley GL, Berstein JJ, Santinelli S, Spelman LH, MIller LD, Arp LH, Anderson M, Barr M, Pearce-Kelling S, Summers BA: Canine distemper epizootic in lions, tigers, and leopards in North America. J Vet Diagn Invest 6:277-288, 1994.
3. Harder TC, Kenter M, Appel MJG, Roelke-Parker ME, Barrett T, Osterhaus ADME: Phylogenetic evidence of canine distemper virus in Serengeti's lions. Vaccine 13:521-523, 1995.
International Veterinary Pathology Slide Bank: Laser disc frame #225-7, 9680-1, 10155-6, 16937-8, 16985, 23076, and 23119.
* The American Veterinary Medical Association and the American College of Veterinary Pathologists are co-sponsors of the Registry of Veterinary Pathology. The C.L. Davis Foundation also provides substantial support for the Registry.