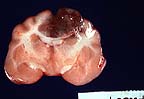
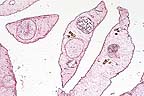
 Focal necrohemorrhagic encephalitis
in a meerkat with phaeohyphomycosis (13K)
Focal necrohemorrhagic encephalitis
in a meerkat with phaeohyphomycosis (13K)Signalment: Adult male slender-tailed meerkat (Suricata suricatta).
 Focal necrohemorrhagic encephalitis
in a meerkat with phaeohyphomycosis (13K)
Focal necrohemorrhagic encephalitis
in a meerkat with phaeohyphomycosis (13K)
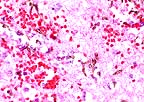 Pigmented fungal hyphae of
Cladosporium sp. in the cerebrum of a meerkat (Suricata
suricatta). (HE, 400X, 59K)
Pigmented fungal hyphae of
Cladosporium sp. in the cerebrum of a meerkat (Suricata
suricatta). (HE, 400X, 59K)
History: This was a wild born meerkat that had been in captivity for more than 9 years. During this time, no significant clinical signs were noted until 4 days before the animal's death when it was noted to be lethargic and continuously vocalizing. Attempts to catch it at that time were unsuccessful. The next day, circling and ataxia were present and the animal was brought to the veterinary hospital at the National Zoo for examination. A blood sample collected at this time revealed mild anemia (PCV = 35.5%; normal 39 - 49%) but all other hematologic and serum biochemical results were within normal ranges. Whole body radiographs were also unremarkable. The meerkat was administered chloramphenicol subcutaneously but by the next morning the ataxia had worsened and the animal was depressed. Thiamin, lactated Ringer's solution, and fluconazole were added to the therapy regimen. The animal's condition continued to deteriorate over the next 36 hours and it was found dead on the morning of the fifth day.
Gross Pathology: Gross lesions were restricted to the central nervous system. The dorsomedial aspect of the left cerebral hemisphere contained a firm, well-vascularized brown and white mottled nodular mass measuring 1.5 cm in greatest diameter. This lesion involved the corpus callosum but did not extend into the right cerebral hemisphere.
Laboratory Results: Clinical pathology results were unremarkable except for the mild anemia noted above.
Impression cytology of the brain lesion revealed high numbers of RBC's, neutrophils, and macrophages with occasional lymphocytes. Numerous brown septate branching fungal hyphae were also present.
Bacterial cultures of the brain were negative. Fungal culture grew an organism that was identified as a species of Cladosporium by the mycology laboratory at Bethesda Naval Hospital.
Frozen sections of brain were negative for rabies by IFA.
Contributor's Diagnosis and Comments:
Brain: meningoencephalitis, suppurative and hemorrhagic, subacute, focal, severe.
Brain: vasculitis, necrotizing, acute, multifocal, moderate.
Dematiaceous (naturally pigmented) fungi include a diverse group of ubiquitous saprophytic organisms most commonly associated with soil, wood, and decaying vegetation. Opportunistic infections by these organisms occur in humans and a wide variety of animals -- through wound contamination and perhaps by ingestion or inhalation of spores. The terms chromoblastomycosis, chromomyocosis, and phaeohyphomycosis have all been used to described infections by dematiaceous fungi resulting in confusion over proper terminology. Most authors now agree that "phaeohyphomycosis" should be used to describe subcutaneous or systemic infections by pigmented fungi which form hyphal structures in tissue whereas in "chromoblastomycosis", thick-walled septate yeast-like forms known as "sclerotic bodies" predominate although hyphae may rarely be found.
In humans and other mammals, chromoblastomycosis is usually confined to the subcutis and surrounding soft tissues. However, widely disseminated infections commonly occur in amphibians. The agents of phaeohyphomycosis usually cause cutaneous or subcutaneous infections but are more likely to be associated with systemic infections than those of chromoblastomycosis. Several of these (e.g. Dactylaria gallopava) appear to have a predilection for the CNS and infections by these fungi are usually limited to the brain, without any other identified sources of dissemination (as in this meerkat). Successful therapy of either chromoblastomycosis or phaeohyphomycosis relies on wide surgical excision of affected areas. Standard antifungal drugs appear to have little effect on these organism.
The source of infection for this meerkat was not determined; its cagemates have remained asymptomatic for over 18 months.
AFIP Diagnosis: Cerebrum: Encephalitis, necrohemorrhagic, acute, focally extensive, severe, with necrotizing vasculitis and pigmented fungal hyphae, meerkat (Suricata suricatta), viverrid.
Conference Note: Phaeohyphomycosis usually causes subcutaneous mycosis in mammals. In tissue, the fungal hyphae are pigmented, septate, and have walls that are irregular in width and length and demonstrate non-dichotomous branching. Systemic infections seldom arise from subcutaneous lesions; they are usually acquired via the respiratory tract. Animals that develop systemic phaeohyphomycosis are often immunocompromised or debilitated.
Cutaneous lesions are composed of single or multiple nodules which occasionally ulcerate or form fistulous tracts. Lesions are most common on the face or distal extremities in the cat; in horses, infection is more common on the body. Histologically, cutaneous lesions consist of fungal hyphae, sometimes rimmed by Splendore-Hoeppli material, within microabscesses. The microabscesses are bounded by granulation tissue and granulomatous inflammation.
Contributor: Department of Pathology, National Zoological Park, Smithsonian Institution, Washington, D.C. 20008.
References:
1. Chandler FW and JC Watts: Pathologic Diagnosis of Fungal Infections,
ASCP Press, Chicago, 1987.
2. McGinnis MR: Laboratory Handbook of Medical Mycology, Academic Press, New York, 1980.
3. Miller EA, RJ Montali, EC Ramsay, and BA Ridout: Disseminated chromoblastomycosis in a colony of ornate-horned frogs (Ceratophyrys ornata). J Zoo Wildl Med 23:433-438, 1992.
4. Rippon JW: Medical Mycology: The Pathogenic Fungi and the Pathogenic Actinomycetes Third Edition, WB Saunders, Philadelphia, 1988.
5. Yager JA, and Scott DW: The skin and appendages in Pathology of Domestic Animals. Jubb KVF, Kennedy PC, and Palmer N, eds., 4th ed, Vol 1, pp. 668, 1993.
International Veterinary Pathology Slide Bank: Laser disc frame #2099.
Signalment: Acanthodraco vitticeps (inland bearded dragon).
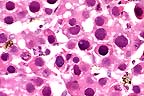 Necrotic hepatocytes in the
liver of a bearded dragon. Almost every is karyomegalic as the
result of a large smudgy basophilic adenoviral inclusion. (HE,
400X, 48K)
Necrotic hepatocytes in the
liver of a bearded dragon. Almost every is karyomegalic as the
result of a large smudgy basophilic adenoviral inclusion. (HE,
400X, 48K)
History: The sections are drawn from three representative cases in an outbreak of sudden deaths of 80% of a batch of 4 to 8-week-old captive-bred bearded dragon hatchlings that occurred over several weeks. No signs of ill-health had been observed in the parent animals; Salmonella spp. had been isolated sporadically from fecal culture of both parents and hatchlings. The animals were housed in several separate batches, all of which were affected. No premonitory signs of illness were observed; affected animals were found dead.
Gross Pathology: Twenty-five animals were examined. In all cases the animals either had a pale, mottled liver or were grossly normal.
Laboratory Results: Salmonella spp. (Group A to I positive) were isolated in mixed culture from some but not all cases.
Contributor's Diagnosis and Comments:
33735, unlabelled Liver, necrosis, diffuse, acute, severe with hepatomegaly and amphophilic intranuclear inclusion bodies. Stomach, intranuclear inclusion bodies, superficial epithelium, rare. Duodenum, intranuclear inclusion bodies, epithelial cells. Pancreas, intranuclear inclusion bodies, acinar cells, rare.
33733 Liver, necrosis, diffuse, acute, severe with hepatomegaly and amphophilic intranuclear inclusion bodies. Duodenum, intranuclear inclusion bodies, epithelial cells. Pancreas, intranuclear inclusion bodies, acinar cells, rare. Lung, normal.
33709 Liver, necrosis, diffuse, acute, severe with hepatomegaly and amphophilic intranuclear inclusion bodies. Intestine, intranuclear inclusion bodies, epithelial cells. Pancreas, intranuclear inclusion bodies, acinar cells, rare. Stomach, normal.
Of twenty-five animals euthanized or submitted dead for necropsy, twenty showed liver lesions; most had inclusions at other epithelial sites similar to the cases presented here. The degree of hepatic inflammation associated with the presence of intranuclear inclusions varied widely. Additional sites affected in other cases included gall bladder epithelium, oral epithelium, kidney tubular epithelium, esophageal epithelium, lung and salivary gland. Viral culture produced only contaminants. Adenovirus-like particles were observed in electron micrographs of liver tissue from several affected animals. A very similar outbreak among juvenile Rankin's dragon lizards (Pogona henrylawsoni) has been reported (Frye et al., 1994), characterized by hepatic necrosis with large, basophilic intranuclear inclusion bodies. Electron microscopy showed numerous intranuclear paracrystalline arrays of adenovirus-like particles. A similar adenovirus infection has been reported in a bearded dragon in New Zealand (Julian and Durham, 1990).
AFIP Diagnosis:
1. Liver: Necrosis, disseminated, with numerous basophilic intranuclear
inclusion bodies, Inland Bearded Dragon (Acanthodraco vitticeps),
reptile.
2. Stomach, duodenum, and pancreas, epithelial cells: Basophilic
intranuclear inclusion bodies, small numbers.
Conference Note: Adenovirus, a double-stranded DNA virus, has been reported in numerous reptiles including Nile crocodiles, boa constrictors, rosy boas, rat snakes, Gaboon vipers, Savannah monitor lizards, Jackson's chameleon, and Rankin's dragon. All of these reptiles had intranuclear viral inclusions in the gastrointestinal tract and/or liver; most also had hepatic necrosis.
Adenovirus is established in some breeding groups of bearded dragons in the United States. Transmission studies have not been done; however, lizards hatched from eggs incubated apart from parents and having no contact with adult lizards after hatching have developed the disease. This suggests that vertical transmission occurs.
In a recent case report of adenovirus and dependovirus-like infection of bearded dragons, the ultrastructural characteristics of the reptilian adenovirus included the formation of paracrystalline arrays in the nucleus of infected cells. The viral particles were 60-66 nm in diameter, non-enveloped, and had electron-dense or electron-lucent cores.
Contributor: Division of Comparative Medicine, Department of Pathology, Johns Hopkins University, Baltimore, MD 21205.
References:
1. Frye, F.L, Munn, K.J., Gardner, M., Barten, S.L. and L. B.
Hadfy (1994) Adenovirus-like hepatitis in a group of related Rankin's
dragon lizards (Pogona henrylawsoni) Journal of Zoo and Wildlife
Medicine 25 (1) 167-171.
2. Julian, A.F. and P.J.K. Durham. (1990) Adenoviral hepatitis in a female bearded dragon (Amphilbolurus barbatus). New Zealand Veterinary Journal 30: 59-60.
3. Jacobson ER, Kopit W, Kenedy FA, and Funk RS: Coinfection of a bearded dragon, Pogona vitticeps, with adenovirus- and dependovirus-like viruses. Vet Pathol 33:343-346, 1996.
Signalment: 12-week-old female racing pigeon.
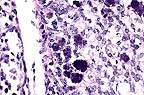 Numerous intrahistiocytic
cytoplasmic inclusions in the bursa of a racing pigeon with avian
cercoviral infection. (HE, 400X, 6K)
Numerous intrahistiocytic
cytoplasmic inclusions in the bursa of a racing pigeon with avian
cercoviral infection. (HE, 400X, 6K)
History: Multiple deaths in young birds. Severe watery diarrhea (E. coli cultured).
Gross Pathology: Splenomegaly; yellow, mottled liver. Gaseous material in bursa of Fabricius.
Laboratory Results: Escherichia coli cultured from feces.
Contributor's Diagnosis and Comments: Moderate diffuse lymphoid necrosis and depletion, bursa of Fabricius. Acute bursal abscessation.
Lymphoid depletion was also evident in the spleen. The cause of death was considered secondary bacterial infection associated with an immunocompromised state. Although no additional diagnostic tests were performed, the presence of characteristic intracytoplasmic inclusions bodies was considered to be consistent with circovirus infection.
AFIP Diagnosis: Bursa of Fabricius: Lymphoid depletion, diffuse, severe, with lymphocyte necrosis and intrahistiocytic basophilic intracytoplasmic inclusion bodies, pigeon, avian.
Conference Note: Circoviruses are a newly proposed group of circular, single-stranded DNA viruses. The circoviruses of pigeons, psittacine beak and feather disease virus, chicken anemia agent, and nonpathogenic porcine circovirus share similar ultrastructural characteristics, protein composition, and antigenic features.
Pigeons with circoviral infections develop lesions in the spleen, bursa of Fabricius, gut-associated lymphoid tissue, and bronchus-associated lymphoid tissue. Changes in the spleen can range from lymphofollicular hyperplasia with mild lymphocyte necrosis to lymphoid depletion and histiocytosis. Bursal changes can also range from mild lymphocyte necrosis to severe cystic bursal atrophy. Basophilic intracytoplasmic inclusion bodies can be seen in macrophages in splenic, bursal, gut-associated, and bronchus-associated lymphoid tissue and in bursal epithelial cells. Ultrastructurally, the virus appears in paracrystalline arrays of tightly packed, nonenveloped icosahedral virions 14-17 nm in diameter. Many circovirus infections in pigeons are complicated by secondary bacterial or viral disease, probably as a result of some degree of immunosuppression secondary to viral infection of the monocyte-macrophage system.
Psittacine beak and feather disease induces symmetrical feather dystrophy and loss due to necrosis and hyperplasia of epithelial cells in the epidermal collar and epidermal, basal and intermediate zones of the developing rachis of the feather shaft. Epidermal cells of the beak are similarly affected. Lymphoid depletion and lymphocyte necrosis occur in the spleen and bursa of Fabricius. Basophilic intracytoplasmic inclusions can be found in cells of the feather, beak, thymus, and bursa.
Chicken anemia agent is responsible for bone marrow hypoplasia with thymic and bursal atrophy in young chicks.
Contributor: Consolidated Veterinary Diagnostics, Inc. 2825 KOVR Drive, West Sacramento, CA 95691.
References:
1. Woods, L.W., et al. Circovirus Infections in Pigeons. Proceedings
of the Association of Avian Veterinarians. P. 156-157, 1993.
2. Woods LW, Latimer KS, et al: A retrospective study of circovirus infection in pigeons: nine cases (1986-1993). J Vet Diagn Invest 6:156-164, 1994.
3. Shivaprasad HL, Chin RP, Jeffrey JS, Latimer KS, Nordhausen RW, Niagro FD, and Campagnoli RP: Particles resembling circovirus in the bursa of Fabricius of pigeons. Avian diseases 38:635-641, 1994.
4. Gerlach H: Viruses in Avian Medicine: Principles and Applications. Ritchie BW, Harrison GJ, and Harrison LR eds. Wingers Publishing, Inc., Lake Worth, Florida, pp. 894-903, 1994.
Signalment: 18-month-old castrated male Holstein bovine.
 Granulomatous pancreatitis
with marked fibrous connective tissue deposition and abundant
black fluke pigment in a bovine with eurytremiasis. (29K)
Granulomatous pancreatitis
with marked fibrous connective tissue deposition and abundant
black fluke pigment in a bovine with eurytremiasis. (29K)
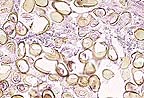
 Cross section of several Eurytrema
sp. flukes from the pancreatic ducts of an 18-month-old Holstein..
Note the prominent suckers, spongy body cavity and vitellarian
glands which identifies them as trematodes on microscopic examination.
(HE, 200X,
Cross section of several Eurytrema
sp. flukes from the pancreatic ducts of an 18-month-old Holstein..
Note the prominent suckers, spongy body cavity and vitellarian
glands which identifies them as trematodes on microscopic examination.
(HE, 200X,
 Operculated, oval trematode
eggs with a thick brown shell are characteristic findings in Eurytrema
infections. (HE, 400X, 61K)
Operculated, oval trematode
eggs with a thick brown shell are characteristic findings in Eurytrema
infections. (HE, 400X, 61K)
History: This animal had progressive loss of weight and moderate anemia during the 6 months prior to death. It was herded along with other several cattle and had stayed during the previous winter on a small river island. It was euthanatizied due to prolonged recumbency. Two other bovines from this herd died with similar clinical histories.
Gross Pathology: A formalin-fixed bovine pancreas was sent to our laboratory for histopathologic examination. No necropsy description was provided. The pancreas was markedly firm, had a thickened white capsule and an irregular capsular surface. At the cut surface (Kodachrome), there were multiple cystic (dilated) ducts with thickened fibrous walls which were packed with leaf-shaped trematodes. Multiple round brown to black areas were also seen. Normal parenchyma was obscured by these changes.
Laboratory Results: The trematodes were identified as Eurytrema sp.
Contributor's Diagnosis and Comments: Chronic granulomatous interstitial pancreatitis and fibrous atrophy of the pancreas with intralesional trematodes and eggs, diffuse, severe, Etiologic diagnosis: parasitic pancreatitis. Etiology: Eurytrema sp.
Eurytrema pancreaticum (Class: Trematoda) and several other species of the genus Eurytrema have been described in the pancreatic ducts and, rarely, bile ducts and duodenum of cattle, sheep, goats, buffaloes, camels, pigs and other species including man. Infections of ruminants are reported from Asia, Brazil and other South American countries. In Brazil, E. coelamaticum is described as the most common species in the pancreatic ducts of cattle and sheep but it is not clear whether this and E. pancreaticum are actually distinct species. The adult fluke measures 8-16 x 5-8.5 mm. The prepatent period in cattle and sheep is 80-100 days.
Eggs are passed in the feces and are ingested by the first intermediate host which, in Brazil, was determined to be the land snail Bradybaena similaris. Within the snail, two generations of sporocysts occur. About 5 months after infection, the second generation produces cercariae which are liberated to the pasture just a few hours before daybreak. Cercariae are eaten by the second intermediate host, grasshoppers from the genus Concephalus, as is the case in Brazil, or by tree crickets (Oecanthus longicuandus), as is the case in Malaysia and the former USSR. Three weeks after infecting the grasshopper, infective metacercaria will appear. Reportedly, a microsporidial protozoan can infect cercariae and prevent their encystment, thereby influencing the trematode burden of the final hosts. Final hosts are infected by accidentally ingesting infected grasshoppers. Metacercaria are encysted in the duodenum, migrate via the accessory pancreatic ducts, and are distributed throughout tributary pancreatic ducts.
Pancreatic eurytrematosis is commonly found in Brazil, being reported from central, southeastern and southern regions. In one report, it is mentioned that 406,768 out of 55,004,855 pancreases had been rejected by the Federal Meat Inspection department during a 10 year period (1970-79) due to lesions caused by Eurytrema sp. Another paper reports that in one county, in the state of S o Paulo, 70% of the cattle slaughtered in the county slaughterhouse had pancreatic infection by Eurytrema sp.
Pancreatic lesions caused by Eurytrema sp. in domestic ruminants vary from almost absent to severe fibrosis with extensive pancreatic tissue loss, as in this case. Adult flukes are always in the lumina of ducts but eggs may penetrate the walls causing granulomatous inflammatory reactions with giant multinucleate cells confined to the walls of pancreatic ducts. The lost pancreatic tissue is replaced, sometimes almost completely, by fibrous or adipose tissue; whatever pancreatic tissue remains is essentially normal. These histopathological aspects are present in this case. The size of adult flukes are reportedly inversely proportional to the intensity of pancreatic lesions probably reflecting lack of nutrition to the worm due to tissue destruction; in those pancreases with minimal tissue destruction, adult flukes are large, active and restricted to larger ducts. In severely damaged pancreases, flukes are stunted or shrunken and found in the smallest ductules. Lesions caused by E. pancreaticum appear to occur more frequently in the left lobe of the pancreas.
Clinical signs associated with pancreatic eurytrematosis in domestic ruminants may not be remarkable. Poor body condition and cachexia are frequently reported but subclinical cases are common. Changes resembling diabetes mellitus have, on occasion, been described in sheep and cattle; however, other reports state that the endocrine pancreas is usually spared. Centrilobular hepatic fatty change, and nephrosis with marked glucosuria have been described in sheep, and glucosuria has been reported in cattle.
A progressive loss of weight was detected in this bovine and in two others in the same herd. It is a fair assumption that such severe pancreatic atrophy, as seen here, would contribute to this animal's poor health. Intervening factors such as infectious diseases, cold winters, or a dearth of forage could contribute to the demise of an animal already weakened due to the pancreatic lesion.
The region of southern Brazil where this steer originated is endemic for pancreatic eurytrematosis. After receiving this case, we carried out a small survey in the abattoir where cattle from this region are usually slaughtered; we found that 30 pancreases had lesions similar in type to the ones of this case; notwithstanding, all bovines slaughtered there in the period surveyed were in excellent body condition and not one was reported to present any clinical signs.
AFIP Diagnosis: Pancreas: Parenchymal loss, diffuse, severe, with fibrosis, chronic pancreatitis, and numerous eggs and intraductal adult trematodes, Holstein, bovine.
Conference Note: This case was reviewed by Dr. C.H. Gardiner, parasitology consultant for the Department of Veterinary Pathology, AFIP. In North America, Eurytrema procyonis can be found within the pancreatic duct of small carnivores, including raccoons, cats, and foxes. Clinical signs and histologic lesions are similar to those seen in ruminants infected with E. pancreaticum.
Contributor: Departamento de Patologia, Universidade Federal de Santa Maria, 97119-900 Santa Maria, RS, Brazil.
References:
1. Basch PF: Patterns of transmission of the trematode Eurytrema
pancreaticum in Malaysia. Am J. Vet. Res. 27(11):234-240, 1966.
2. Graydon RJ, Carmichael IH, Sanchez MD, Weisosari E., Widjayanti S: Mortalities and wasting in Indonsesian sheep associated with the trematode Eurytrema pancreaticum. Vet. Rec. 7:433, 1992.
3. Jubb KVF: The pancreas, In Jubb KVF, Kennedy PC, Palmer N (eds.): Pathology domestic animals, 4th ed., Academic Press, NY, vol, 2, chap. 3, P. 417, 1993.
4-7. Soulsby EJL Helminths, arthropods and protozoa of domesticated animals, 67th ed., Ball‚re, Tindall, London, pp. 15-17, 1968.
* The American Veterinary Medical Association and the American College of Veterinary Pathologists are co-sponsors of the Registry of Veterinary Pathology. The C.L. Davis Foundation also provides substantial support for the Registry.