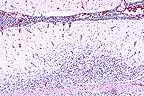
 Non suppurative meningoencephalitis
in an Appaloosa gelding infected with equine lentivirus (HE, 100X,
76K)
Non suppurative meningoencephalitis
in an Appaloosa gelding infected with equine lentivirus (HE, 100X,
76K)Signalment: Six-year-old Appaloosa gelding.
History: Horse was presented prior to necropsy with clinical signs of ataxia and uveitis. Several laboratory tests were performed (see below). The horse was euthanized.
Gross Pathology: All examined peripheral and visceral lymph nodes were 2-3 times normal size and hemorrhagic. The liver was enlarged and the spleen was normal size. There were approximately 50 bots on the gastric cardia. The femoral bone marrow was hyperplastic. The horse was not icteric.
Laboratory Results: Serum chemistry panel revealed elevated liver enzymes, (alkaline phosphatase-4012; SGOT-671; GGT-2511). A liver biopsy was characterized by severe, non-suppurative, pericholangitis. A CSF tap had a 4+ Pandy with 182 mg/dl of protein and 130 WBC (pleocytosis). The animal was positive for equine infectious anemia using both the Coggins' test and western blot analyses.
Contributor's Diagnosis and Comments: Severe nonsuppurative meningoencephalitis and choroid-plexitis with microgliosis. Lentivirus of equine infections anemia (EIA).
This case represents a classic presentation of the neural form of EIA. In the contributors experience, less than 2% of Coggin's positive horses have evidence of CNS involvement. This horse had typical gross lesions of recrudescing EIA. The farm of origin has had problems in the past with EIA and this case caused a quarantine to be placed on the farm.
The CNS lesion typically has an "inside out" pattern, whereby the inflammation is severe around the ventricular system. In this horse, that was best seen in the spinal cord. This horse tested negative for sarcocystosis, neosporsosis, VEE and EEE.
AFIP Diagnosis: Cerebellum, brain stem, midbrain, and choroid plexus: Choriomeningoencephalitis, nonsuppurative, multifocal, moderate, Appaloosa, equine.
Conference Note: Equine infectious anemia affects horses, mules, and donkeys. It is caused by a lentivirus in the family Retroviridae. The virus is spread by arthropods, which serve as mechanical rather than biologic vectors. The disease can also be spread by contaminated needles, syringes, tattoo equipment, or experimentally, by the administration of infected blood or the virus. The virus primarily infects cells of the monocyte-macrophage system; however, in the acute viremic stage, viral DNA is also found in cells of the liver, lymph nodes, bone marrow, spleen, and kidney. Equine infectious anemia causes intracellular hemolysis followed by immunoproliferation and finally by hematopoietic, and to a lesser extent, immune exhaustion. It is believed that the EIA virus does not directly kill cells and that the cellular damage is the result of indirect effects of the immune response. The clinical signs of EIA develop at the time of the appearance of circulating antibody which is complement fixing. C3 adheres to erythrocyte membranes, stimulating macrophage recognition and erythrophagocytosis. This results in severe intravascular hemolysis and anemia.
Clinical EIA is characterized by cyclic pyrexia, anorexia, weight loss, and pitting edema. Petechial hemorrhages are often seen on the tongue, conjunctiva, and vaginal mucosa. Icterus develops and is accompanied by tissue pallor due to anemia. Death may occur at this stage or the disease may regress and then recur at irregular intervals. Clinical pathology reveals severe anemia with RBC counts as low as 1 x 1012/liter and hemoglobin of 25-50 gm/l. Initially, the anemia is characterized by anisocytosis with moderate poikilocytosis. As the disease progresses, the anemia becomes normochromic, normocytic, and nonresponsive. There is accompanying thrombocytopenia, leukopenia, neutropenia, lymphopenia, and monocytosis. Monocytes often contain phagocytized erythrocytes.
Gross lesions are variable and dependant upon the course of the disease. Animals that die in the acute stages exhibit anemia, icterus, and widespread hemorrhage. Those that survive longer are emaciated and have serous atrophy of fat, which is also bilirubin-stained. They develop splenomegaly, with surface hemorrhages and inapparent lymphoid follicles on cut surfaces. The liver is also enlarged with capsular hemorrhages. Petechial hemorrhages are often present on renal capsules and perirenal tissues. The fat in bone marrow is replaced by hemopoietic tissue, turning the marrow diffusely red.
Microscopic lesions in chronic cases are most prominent in the heart, lungs, liver, spleen, kidney, bone marrow, and lymph nodes. There is atrophy of cardiomyocytes and the interstitium is expanded by edema and small numbers of lymphocytes. Similar inflammatory cells cause thickening of the alveolar septa. There is hepatic congestion with mild periportal infiltrates of lymphocytes; Kupffer cells are hyperplastic and contain hemosiderin. Splenic lymphoid follicles are often enlarged, but hypocellular. In addition, there is splenic hemosiderosis and plasmacytosis. Lymph nodes are edematous and have medullary hemosiderosis and hypocellular lymphoid follicles. The bone marrow has a reduction in the amount of fat and hemopoietic cells are densely packed. There is an erythroid shift with synchronous maturation and a concomitant reduction in granulocytic cells. As the disease progresses, cellular density decreases and the marrow becomes relatively hypocellular.
Equine idiopathic immune hemolytic anemia may clinically resemble EIA; however, affected horses respond to corticosteroid therapy, ususally are not thrombocytopenic, and lack the inflammation and cyclic fever of EIA.
Contributor: Department of Veterinary Pathobiology, Texas A&M University, College Station, TX 77843-4467.
References:
1. Held JP, McGavin MD, Geiser D. Ataxia as the only clinical
sign of cerebrospinal meningitis in a horse with equine infections
anemia. JAVMA 183:324-325, 1983.
2. McClure J, Lindsay W, Taylor W, et al. Ataxia in four horses with equine infections anemia. JAVMA 180:279-283, 1982.
3. McIlwraith W, Kitchen ND, Neurological signs and neuropathology associated with a case of equine infections anemia. Cornell Vet 68:238-249, 1978.
4. Tajima M, Yamagiwa S. Histopathological studies on the central nervous system in equine infectious anemia. 1. Description of the pathological changes in representative cases. Jap J Vet Sci 15:47, 1953.
5. Valli VEO: The hematopoietic system in Pathology of Domestic Animals. Jubb KVF, Kennedy PC, and Palmer N, eds., Academic Press, Inc., San Diego, 4th edition, pp. 182-186, 1993.
Signalment: 8-year-old male Pomeranian dog.
History: This dog was examined because of coughing and difficult breathing which began 2-3 months earlier. On physical examination, ulcerative skin lesions were noted on the ears, periocular region, lips, footpads and elbows. Hyperpigmentation of the ventral abdomen was also present. Initial skin biopsy was diagnosed as ulcerative dermatitis. ANA/LE tests were negative. A collapsed trachea was diagnosed radiographically. The dog returned 2 weeks later with severe progression of the skin lesion and respiratory distress; it was euthanitized.
Gross Pathology: The skin surrounding the eyes was alopecic, hyperemic, hyperpigmented and moist. The periocular ulceration extended 2.5 cm from the margins of the eyelids and was well demarcated. The skin along the margins of both ears was ulcerated and had focal areas of crusting. Two, 6 X 4 cm, areas of alopecia, hyperpigmentation and crusting were present in the skin of the ventral abdomen. Most footpads were ulcerated with separation from the dermis. Focal ulcers were present on the pressure points of both elbows and hocks. There was pronounced dorsoventral flattening of the trachea.
Laboratory Results: None submitted.
Contributor's Diagnosis and Comments: Severe necrotizing tracheitis associated with toxic epidermal necrolysis.
Toxic epidermal necrolysis (TEN) is a life-threatening, ulcerative skin disease that occurs rarely in the dog and cat. The facial skin, mucocutaneous junctions and oral mucosa are commonly involved. Necrosis of the mucosa of the trachea, large bronchi, pharynx and esophagus can also occur (Gross et al, 1992). Drug hypersensitivity reactions, neoplasia and concurrent infections have been implicated in the pathogenesis, although many cases are idiopathic. In TEN, early full-thickness epidermal necrosis occurs with minimal inflammatory reaction. Leukocyte infiltration occurs following ulceration. In this case, the ulcerative skin disease was advanced, with marked secondary pyoderma in areas of ulceration. This dog had no known exposure to drugs commonly associated with TEN, nor was there evidence of underlying infectious disease on postmortem examination.
AFIP Diagnosis: Trachea: Tracheitis, ulcerative, subacute, focally-extensive, severe, with pseudomembrane and squamous metaplasia, Pomeranian, canine.
Conference Note: Toxic epidermal necrolysis has been associated with drug therapy, systemic neoplasia, host vs. graft reactions, and bacterial infection. The pathogenesis is unknown. It is believed that cell-mediated immune mechanisms destroy the epidermal basal cells. Direct immunofluorescence in two cases of drug-induced TEN revealed intercellular fixation of immunoglobulins and complement in the basal cell layer of the epidermis.
Histologically, TEN is characterized by coagulative necrosis of the epidermis with minimal dermal inflammatory infiltrates. The necrotic epidermis separates from the dermis to form variably sized vesicles, which rupture, forming ulcers. Ulceration often induces a secondary dermatitis with predominance of neutrophils. Ultrastructural studies show condensation of tonofilaments around pyknotic nuclei and the disapperance of all organelles within the affected epithelial cells.
TEN has a high mortality rate due to fluid and electrolyte loss through the damaged epithelium. Differential diagnosis for TEN includes thermal burns, erythema multiforme, pemphigus vulgaris, bullous pemphigoid, systemic lupus erythematosus, and cutaneous vasculitis.
Contributor: Department of Pathology, College of Veterinary Medicine, University of Tennessee, Knoxville, TAN 37901-1071.
References:
1. Gross, T, Ihrke PH, Walder EJ: Toxic epidermal necrolysis.
In: Veterinary Dermatopathology, Mosby-Year Book Inc, St Louis,
pp 44-46, 1992.
2. Rachofsky MA, Chester DK, Read Wk: Toxic epidermal necrolysis. Compend Cont Educ Pract Vet 11:840-845, 1989.
3. Scott et al: Toxic epidermal necrolysis in two dogs and a cat. J Am An Hos Assoc 15:271-279, 1979.
4. Scott DW et al: Immune-mediated dermatosis in domestic animals: ten years after - part II. Comp Cont Educ for Pract Vet 9:539-553, 1987.
International Veterinary Pathology Slide Bank: Laser disc frame #22860
Signalment: 4.5-year-old spayed female Maine Coon cat.
History: During the month prior to euthanasia, the cat developed persistent neurologic signs characterized by head tremors and seizure-like activity.
Gross Pathology: The brain was grossly unremarkable. The liver was slightly pale and tan.
Laboratory Results: None submitted.
Contributor's Diagnosis and Comments: Cytoplasmic polyglucosan inclusions (Lafora bodies) within neuronal perikarya and processes, most numerous in Purkinje cells.
In sections of brain, there are few to many basophilic to amphophilic inclusions that occur within the cytoplasm of nerve cell bodies, neuronal processes, and scattered in the neuropil. These inclusions are usually round, range in size from 2 to 10 um diameter, and occasionally have a pale center or a thin, striated halo. These Lafora-like bodies are most numerous in the cerebellar molecular layers and within Purkinje cells, but are observed in all examined sections of brain.
Lafora bodies consist of complex polymers of glycoprotein, sometimes designated "polyglucosan bodies", and can occur within neuronal cell bodies or processes of the brain, spinal cord, or retinal ganglia. The round to globular basophilic cytoplasmic inclusions stain positively with PAS, alcian blue, and methenamine silver. In domestic animals, Lafora bodies have been observed most commonly in aging (at least 8 years of age) dogs and cats without apparent neurologic disease, but have also been described as incidental changes in some young animals. Lafora bodies have also been associated with a severe form of progressive myoclonus epilepsy in humans (juveniles) and dogs (young adult to middle-age), reflecting widespread intra-neuronal storage of these polyglucosans ("Lafora disease" or "neuronal glycoproteinosis"). The absence of other lesions to account for the observed neurologic signs incriminates the Lafora bodies as playing a role in the reported neurologic signs in this cat. In dogs with associated neurologic disease, the deposits are most commonly observed in Purkinje cells, similar to the distribution noted in this cat. In children with Lafora disease, other sites of polyglucosan deposition include skin and liver, and biopsies of these tissues have been used to help establish the diagnosis. Lafora bodies were not found in tissues from other than the CNS in this cat.
AFIP Diagnosis: Cerebellum, molecular and Purkinje cell layers: Polyglucosan bodies (Lafora bodies), neuronal and extracellular, diffuse, numerous, Maine-Coon, feline.
Conference Note: This case was also reviewed by AFIP's Department of Neuropathology. Lafora bodies are complex glycoprotein neuronal inclusions that can occur in the perikaryon, dendrites, or axons. They are PAS-positive and stain with alcian blue and methenamine silver. They can be found anywhere in the neuraxis and in retinal ganglion cells. Polyglucosan bodies have been associated with neurologic disease in humans, cats, dogs (Beagles, Basset Hounds, and Poodles), and a cockatiel. Lafora's disease in humans is a rare familial neurologic disorder of children. The disease is progressive and manifested by myoclonus. The characteristic histologic feature in the human disease is intraneuronal polyglucosan bodies, which occur predominantly in the substantia nigra and dentate nucleus. In dogs with neurologic disease attributed to the accumulation of intraneuronal polyglucosan bodies, the bodies are found predominantly within Purkinje cells and in thalamic neurons. In animals, Lafora bodies are frequently seen in the absence of neurologic disease. In these cases, the Lafora bodies can be found in any area of the brain and in the spinal cord.
Ultrastructurally, Lafora bodies have electron-dense fibrillary and sometimes granular components. They are not membrane bound and are often associated with rough endoplasmic reticulum and golgi apparatus.
Contributor: University of Georgia, College of Veterinary Medicine, Department of Veterinary Pathology, Athens, Georgia 30602-7388.
References:
1. Degenerative diseases of the central nervous system. In: BA
Summer, JF Cummings, and A deLahunta, Veterinary Neuropathology,
pp. 208-350. St. Louis: Mosbey, 1995.
2. Kamiya S and Suzuki Y. Polyglucosan bodies in the brain of the cat. J Comp Pathol, 101:253-267, 1989.
3. Jubb KVF and Huxtable CR. The nervous system, In: KVF Jubb, PC Kennedy, and N Palmer (eds), Pathology of Domestic Animals, fourth edition, pp. 267-439, San Diego: Academic Press, 1993.
Signalment: Farmed Atlantic salmon (Salmo salar), post-smolts.
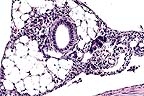 Diffuse severe pancreatic
degeneration and loss in a farmed Atlantic salmon. (HE, 40X, 50K)
Diffuse severe pancreatic
degeneration and loss in a farmed Atlantic salmon. (HE, 40X, 50K)
History: Inappetence, lethargy, and increased mortalities in farmed post-smolt Atlantic salmon during first year in sea water.
Gross Pathology: No food in gastrointestinal tract. Reduced body fat, especially in the region of pyloric cecae. Some fish also had yellow fecal casts.
Laboratory Results: No viruses or pathogenic bacteria were isolated.
Contributor's Diagnoses and Comments: Exocrine pancreas: Pancreatitis associated with severe necrosis of exocrine tissue and mild focal fibroplasia.
Heart: Myocardial necrosis, severe.
Red skeletal muscle: Hyaline necrosis, moderate.
Etiology: Pancreas disease: Atlantic salmon (Salmo salar).
There is marked loss of exocrine pancreatic tissue. A few foci of intact acinar tissue remain adjacent to islets of Langerhans and interlobular ducts. Loss of acinar tissue is associated with mild mononuclear cell infiltration and mild focal fibroplasia. Multifocal myocardial necrosis is also apparent. There is macrophage infiltration of necrotic fibers, vacuolation of the spongy trabecular ventricular myocardium and proliferation of endocardial cells. Moderate necrosis of the "red" skeletal muscle along the lateral line is apparent.
Pancreas disease (PD; polymyopathy syndrome; sudden death syndrome) causes major economic loss in the farmed Atlantic salmon industry in Europe. It can result in 10 to 50% mortalities and stunted growth in survivors. Until recently, the cause of PD was unknown. However, a toga-like virus has been recently isolated from affected fish (Nelson et al, 1995). Inoculation of this virus into healthy fish produces pancreatic, cardiac and skeletal muscle lesions similar to those seen in spontaneous outbreaks of PD. Studies of spontaneous outbreaks of the disease indicate that the initial lesion is acute pancreatic acinar cell necrosis and multifocal myocardial necrosis followed 2 to 3 weeks later by the development of skeletal muscle damage. It is speculated that the pancreatic and myocardial lesions may be the result of direct virus damage while the skeletal muscle lesions may have a viral and/or nutritional etiology.
AFIP Diagnoses: 1. Pancreas: Degeneration and loss, diffuse, severe, with subacute pancreatitis, steatitis, and serositis, Atlantic salmon (Salmo salar), piscine. 2. Heart, myocardium: Degeneration and necrosis, multifocal, moderate, with lymphohistiocytic myocarditis and endothelial cell hypertrophy. 3. Heart: Epicarditis, multifocal, lymphohistiocytic, moderate. 4. Skeletal muscle: Degeneration and necrosis, focally extensive, moderate, with fibrosis, lymphohistiocytic myositis and regenerative changes.
Conference Note: Pancreas disease is characterized by pancreatic atrophy with replacement by fibrosis and severe degeneration of cardiac and red skeletal muscle (aerobic skeletal muscle). The disease has been most reported in farm-raised salmon that have adapted to sea water (post-smolt). As the contributor notes, a togavirus has recently been identified as the cause of this disease.
Although the complete pathogenesis of pancreas disease has not been determined, there is evidence that vitamin E and selenium deficiency may play a role in development of the skeletal muscle lesions. Experimental vitamin E-selenium deficiency produces myofiber lesions quite similar to those that occur in trout and salmon with pancreas disease. It has also been found that salmon suffering from pancreas disease have lower serum and tissue levels of vitamin E and selenium when compared to healthy salmon. These findings suggest that virus-induced pancreatic atrophy may result in maldigestion/malabsorption and reduced absorption of vitamin E and/or selenium rendering skeletal muscle vulnerable to lipid peroxidation.
Contributor: Veterinary Sciences Division, Stoney Road, Stormont, Belfast, BT.-3SD, N. Ireland.
References:
1. McCoy, M.A., McLouglin, M.F., Rice, D.A., Kennedy, D. G. (1994)
Pancreas disease in Atlantic salmon (Salmo salar) and vitamin
E supplementation. Com. Biochem. Physiol. 109A:905-915.
2. McLoughlin, M.F., Nelson, R. T., McCormick, J.I., Rowley, H. (1995). Pathology of experimental pancreas disease in fresh water Atlantic salmon parr. J. Aquat. Hlth 7:104-110.
4. Nelson, R.T., McLoughlin, M.F., Rowley, H.M., Platten, M.A. McCormick, J.I. (1995) Isolation of a toga-like virus from farmed Atlantic salmon (Salmo salar) with pancreas disease. Dis. Aquat. Org 22:25-32.
* The American Veterinary Medical Association and the American College of Veterinary Pathologists are co-sponsors of the Registry of Veterinary Pathology. The C.L. Davis Foundation also provides substantial support for the Registry.