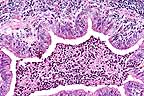
 Suppurative bronchopneumonia
in a rabbit with a combination of Bordetella bronchiseptica and
CAR bacillus. (HE, 200X, 71K)
Suppurative bronchopneumonia
in a rabbit with a combination of Bordetella bronchiseptica and
CAR bacillus. (HE, 200X, 71K)Signalment: 4-month-old male New Zealand white rabbit.
History: The rabbit was one of several in a shipment from a commercial vendor, in which abnormal respiratory noises were detected. Moist rales were detected by auscultation. The animal was euthanzied and a necropsy was performed.
Gross Pathology: The hilar aspects of the left cranial, left middle, and right cranial lung lobes contained multiple areas of consolidation.
Laboratory Results: Bordetella bronchiseptica was isolated from cultures of pulmonary lesions, trachea and nasal passages. This particular rabbit was seronegative to a commercial rabbit CAR bacillus ELISA test.
Contributor's Diagnosis and Comments: Purulent bronchopneumonia with lymphoid hyperplasia - cilia-associated respiratory bacillus - Bordetella bronchoseptica.
Bordetella bronchiseptica has been isolated from normal rabbits and young rabbits with respiratory disease. The organism localizes along the cilia of the respiratory epithelium in rabbits, and has been shown to cause ciliostasis in the canine trachea. In chronic infection, it is associated with peribronchial lymphoid hyperplasia, as well as suppurative bronchopneumonia.
Cilia-associated respiratory (CAR) bacillus localizes within the cilia and is associated with peribronchial lymphoid hyperplasia. Natural infections of CAR bacillus in rabbits are reportedly not associated with clinical signs, gross or histologic lesions. Based upon analysis of the 16s mRNA gene sequence, rabbit-origin CAR bacillus is most closely related to members of the genus Helicobacter, white rat-origin CAR bacillus is closely related to the genus Flavobacterium. Serologic tests based upon rat CAR bacillus antigen may be inadequate to detect rabbit CAR bacillus infections.
AFIP Diagnosis: Lung: Bronchopneumonia, suppurative, focally-extensive to diffuse, moderate to severe, New Zealand white rabbit, lagomorph.
Conference Note: Bordetella bronchiseptica is a gram-negative coccobacillus. It is an important cause of respiratory disease in dogs, pigs (atrophic rhinitis), rabbits, and laboratory rodents. Bordetella avium is the agent of turkey coryza and is also pathogenic for quail. Bordetella sp. are most likely transmitted by aerosols, as the organisms are short-lived in the environment. Once introduced to the respiratory tract, the bacteria attach to epithelial cells by fimbriae, filamentous hemagglutinin, and peractin (an adhesin). Once the epithelium is colonized, the bacteria produce several toxins including adenylate cyclase toxin, dermonecrotic toxin, and tracheal cytotoxin. Adenylate cyclase toxin, a member of the RTX toxin family, causes toxic levels of cAMP within macrophages and neutrophils, which prevents the respiratory burst and phagocytic activity. Dermonecrotic toxin is an intracellular product that is released upon lysis of the bacterium and inhibits sodium-potassium ATPase, resulting in vasoconstriction. Tracheal cytotoxin decreases ciliary activity and induces structural changes of ciliated epithelial cells. The loss of ciliated cells causes accumulation of mucus, bacteria, and foreign particles, increasing the likelihood of secondary bacterial infections.
Cilia-associated respiratory (CAR) bacillus causes chronic tracheobronchitis and bronchopneumonia in Iaboratory rodents. In rabbits, the CAR bacillus has been shown to be of minimal clinical importance, causing only mild goblet cell hyperplasia with no inflammatory infiltrate and mild parabronchiolar lymphoplasmacytic hyperplasia with concurrent epithelial cell and/or goblet cell hyperplasia. Rat and rabbit strains of CAR bacillus appear to have significant differences, the rat strain being more closely related to Flavobacterium and the rabbit strain more closely related to Helicobacter. It is not known if the differences in lesions between rodents and rabbits is due to variations in virulence factors, host susceptibility, or both.
In this case, there are myriad silver-staining bacilli among the cilia of the bronchial epithelium. Simultaneous infection of CAR bacillus and Bordetella bronchiseptica have been reported and it has been theroized that this combination increases the severity of the respiratory disease.
Contributor: The Milton S. Hershey Medical Center, The Pennsylvania State University, Department of Comparative, Medicine, P.O. Box 850 Hershey, PA 17033.
References:
1. Cundiff DD, Besch-Williford CL, Hook RR, et al.: Characterization
of Cilia-Associated Respiratory Bacillus in Rabbits and Analysis
fo the 16S mRNA Gene Sequence. Lab Animal Science 45:22-26, 1995.
2. Cundiff DD, Besch-Williford CD, Hook RR, et. al.: Characterization of Cilia-Associated Respiratory Bacillus Isolated from Rats and Rabbits. Lab Animal Science 44:305-312, 1994.
3. Kurisu K. Kyo S. Shiomoto Y, et.al.: Cilia-Associated Respiratory Bacillus Infection in Rabbits. Lab Animal Science 40:413-415, 1990.
4. Percy DG, Barthold SW: Pathology of Laboratory Rodents and Rabbits. Iowa State Univ. Press, Ames, pp 191, 1993.
5. Bemis DA, Burns EH: Bordetella in Pathogenesis of bacterial infections in animals, Byles Cl and Thoen CO, eds., Iowa State Univ. Press, pp. 201-215, 1993.
International Veterinary Pathology Slide Bank: Laser disc frame #2242, 7513-14, 12716, 12939, 13993, and 15557-9.
Signalment: Two male Long Evans rats (89-462 & 463) and one male Sprague Dawley rat (89-481).
History: These rats were sacrificed for health assessment. Blood was sumitted for serology; nasal, rectal and pharngeal swabs were submitted for bacteriologic culture; and feces were examined for intestinal parasites.
Gross Pathology: No gross lesions were noted on the necropsy report.
Laboratory Results: Fecal - negative, microbiology - negative. Serology - the two Long Evans rats were serologically positive for Sendai virus (89-462 and 463); the Sprague Dawley rat was postive for sialodacryoadenitis virus (SDA) / rat coronavirus (RCV).
Contributor's Diagnosis and Comments: 1. Salivary glands, submandibular and parotid: Sialoadenitis, necrotizing, subacute, diffuse, moderate, with associated periglandular inflammation and squamous metaplasia of interlobular ducts.
2. Lymph nodes, submandibular: Hyperplasia, lymphoid, cortical, diffuse, moderate, with multifocal sinus hemorrhage.
The changes seen in these cases are consistent with infection by SDA/RCV. The lesions vary somewhat between animals and between sections, but they are generally characterized as diagnosed above. There is also diffuse acinar atrophy in the submandibular salivary glands in some sections. Although two of the rats were seropositive for Sendai virus, no lesions attributable to that agent were seen in any of the sections of lung which were examined.
Viral sialodacryoadenitis is a naturally occurring and fairly common disease of the laboratory rat. Lesions associated with the disease include inflammation of the salivary and lacrimal glands. Severe involvement of the lacrimal glands can lead to compromised function and permanent ocular damage. It has recently been shown that lesions tend to persist in some lacrimal glands, making them useful for microscopic examination for retrospective, presumptive diagnoses of SDAV outbreaks. Other lesions associated with SDAV are transient rhinitis, tracheobronchitis, and alveolitis. In addition, experimental intranasal inoculation of suckling rats with SDAV causes acute necrotizing encephalitis. SDA virus also exacerbates respiratory mycoplasmosis under experimental conditions and causes depletion of salivary gland epidermal growth factor.
AFIP Diagnosis: 1. Salivary glands, submandibular and parotid:
Sialoadenitis, necrotizing, subacute, diffuse, moderate, with
marked edema, and squamous metaplasia of intralobular ducts, rat,
rodent.
2. Lymph node, mandibular: Lymphoid hyperplasia, follicular and
paracortical, moderate, with plasmacytosis.
Conference Note: Sialodacryoadentitis in rats is caused by the sialodacryoadentitis virus (SDAV), a corona virus that is antigenically similar to mouse hepatitis virus and Parker's rat coronavirus. Rats infected with SDAV display sniffling, photophobia, chromodacryorrhea, intermandibular swelling, and reduced fertility. Histologically, there is necrotizing sialoadenitis of the submandibular and parotid salivary glands, and necrotizing dacryoadentitis of the Harderian glands and exorbital lacrimal glands. The sublinguial salivary gland, a mucous gland, is spared. A transient rhinitis, tracheobronchitis, and alveolitis are also associated with SDAV infection. Lymphoid hyperplasia of mandibular lymph nodes is a common finding in active SDAV infections.
As the contributor notes, infection with SDAV decreases levels of salivary gland epidermal growth factor (EGF). Epidermal growth factor is secreted primarily by the granular convoluted tubule cells of the submandibular glands of mice and rats. It is produced in small quanities in renal tubules, Brunner's glands, and serous glands of the nasal cavity. Epidermal growth factor may be secreted into the circulation; however, current evidence suggests that it is swallowed and reabsorbed in the gastrointestinal system by receptor-mediated endocytosis. The physiologic role of EGF in rodents is not fully understood; however, it has been shown to influence carcinogenesis. For example, sialoadenectomized mice have a much lower incidence of spontaneous mammary tumors as compared to intact mice. In addition, sialoadenectomy in mammary tumor-bearing mice led to cessation of tumor growth. Reproductive parameters are also influenced by EGF. In sialoadenectomized male mice, sperm counts were reduced 50%, and in sialoadenectomized females there was reduced milk production and mammary gland development during lactation. These studies suggest that SDAV infection could have significant effects on experimental studies on EGF-dependant functions.
Contributor: Uniformed Services University of the Health Sciences, 4301 Jones Bridge Rd. Bethesda, MD 20814.
References:
1. Jacoby, RO, Bhatt, PN, Jonas, AM: Pathologenesis of sialodacryoadentiis
virus in gnotobiotic rats. Vet Pathol 12:196-209, 1975.
2. Percy, DH, Hayes, MA, Kocal, TE, Wojcinski, ZW: Depletion of salivary gland epidermal growth factor by sialodacryoadenitis virus infection in the Wistar rat. Vet Pathol 25:183-192, 1988.
3. Percy, DG, Lymch, JA, Descoteaux, J-P: Central nervous system lesion suckling mice and rats inoculated intranasally with sialodacryoadenitis virus. Vet Pathol 23:42-49, 1986.
4. Percy, DG, Wojcinski, ZW, Schunk, MK: Sequential changes in the Harderian and exorbital glands in Wistar rats infected with sialodacryoadenitis virus. Vet Pathol 26:238-245, 1989.
Signalment: 5-year-old female New Zealand white rabbit.
 Mammary neoplasm from a New
Zealand rabbit (18K).
Mammary neoplasm from a New
Zealand rabbit (18K).
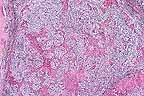 Infiltrative mammary adenocarcinoma
in a New Zealand White rabbit. (HE, 40X, 76K)
Infiltrative mammary adenocarcinoma
in a New Zealand White rabbit. (HE, 40X, 76K)
History: This rabbit was used for experimental antibody production. It was noted to have multiple ventral abdominal masses. Chest radiographs indicated an enlarged heart and possible pulmonary metastases. It was euthanized and submitted for a necropsy examination.
Gross Pathology: There were multiple masses in many mammary glands. All left-sided glands and the last (inguinal) right sided gland had masses. The masses were multi-lobulated, tan to cream colored, and surrounded by fine fibrous capsules. They were located in the subcutis, unattached to underlying muscle and overlying skin, and ranged from 0.5 cm diameter to 6 x 5 x 2 cm. The masses contained very small (less than 1 mm) to a few larger (half cm) pockets of creamy tan fluid. There were no skin ulcers. Some nipples were enlarged and firm. There were numerous fluid filled cysts (1-3 mm) in the uterine endometrium. The heart was normal, and there was no gross evidence of metastasis. Body fat was abundant, and other organ systems were grossly unremarkable.
Laboratory Results: None submitted.
Contributor's Diagnosis and Comments: Mammary Gland: Adenocarcinoma, intraductal and papillary, multiple, New Zealand White rabbit.
Mammary gland adenocarcinomas of rabbits are relatively uncommon. The literature from 1911 to 1962 includes 36 cases of primary mammary gland adenocarcinomas of rabbits. This tumor occurs most often in multiparous females older than three years. In theory, preexisting endocrinological disturbances sensitize the glandular epithelium to the carcinogenic effects of estrogen. There may be a genetic component as well, since the majority of reported cases have occurred in Belgian and English breeds.
The invasive (intraductal) form is often, but not always, preceded by slowly progressive cystic and papillomatous changes, and is the eventual outcome of the papillomatous stage. Mammary adenocarcinomas usually develop independently of the more common uterine adenocarcinoma of rabbits, although cystic mammary changes occur in association with about 25% of uterine adenocarcinomas. This rabbit had diffuse, cystic endometrial hyperplasia, considered by some to be an antecedent to uterine carcinoma. Metastasis to the lungs, abdominal organs, lymph nodes and bone marrow have been reported in approximately 20% of cases. In this case there were no gross or microscopic metastases. Testicular adenocarcinoma also has been reported in rabbits.
AFIP Diagnosis: Mammary gland: Adenocarcinoma, mammary, New Zealand White rabbit, lagomorph.
Conference Note: Other changes commonly seen in association with mammary adenocarcinoma in rabbits include vacuolar change of adrenal cortical cells, pituitary gland hyperplasia, and cystic hyperplasia of uterine endometrial glands. These three changes are consistent with hyperestrinism; however, neither uterine or mammary adenocarcinoma have been experimentally induced by the administration of estrogen.
As in rabbits, familial risk factors and estrogen imbalances have also been implicated in human breast carcinoma. Genetic predisposition has been associated with a small locus in chromosome 17q21, named BRCA1 (breast carcinoma 1). This gene has also been linked to ovarian cancer. The role of estrogen in development of human breast carcinoma is not completely understood; however, excess estrogens have been associated with an increased breast cancer risk. In humans, normal breast epithelium possesses estrogen and progesterone receptors, as do some types of human breast cancer. Human breast cancer cells have also been shown to produce growth factors such as transforming growth factor-à, epithelial growth factor, fibroblast growth factor, platelet derived growth factor, and transforming growth factor- . These growth factors are believed to be involved in an autocrine mechanism of tumor progression. Production of these growth factors is dependant on estrogen and the interactions of estrogen receptors on cancer cells. Circulating hormone and the autocrine growth factors are also presumed to be involved in tumor progression.
Contributor: National Institute of Health, NCRR/VRP, 28 Library Dr MSC 5230, Bethesda, MD 20892-5230.
References:
1. Weisbroth SH, Flatt RE, and Kraus AL, eds (1974). "The
Biology of the Laboratory Rabbit." Chp. 14, pp. 335-347.
Academic Press, New York.
2. Cotran RS, Kumar V, and Robbins SL: Pathologic Basis of Disease. W.B. Saunders Co., Philadelphia, 5th ed., pp. 1099-1109, 1994.
International Veterinary Pathology Slide Bank: Laser disc frame #642, 2624-25, 8431-32, 8535-37, 9609, 10839, 10877-78, 11563, 11610, 11623, 11674, 20980, 22876, and 22945-46.
Signalment: 4 to 6-week-old Sprague-Dawley (Crl:CD (SD)BR VAF Plus ) rat.
History: Rats were dosed intraperitoneally with acrylamide at 50 mg/kg every other day for a total of 10 doses. The animals developed marked hindlimb paresis. Animals were necropsied and tissues fixed by vascular perfusion. Peripheral nerves were embedded in epoxy.
Gross Pathology: No gross changes were present.
Laboratory Results: None submitted.
Contributor's Diagnosis and Comments: Morphologic Diagnosis: Axonal degeneration, slight, multifocal, tibial nerve.
Histologic findings. Peripheral axonopathy is evident with myelin fragments, ellipsoids and axonal fragments with more severely affected axons having macrophages with phagocytized lipid.
Acrylamide is a neurotoxic industrial chemical. It is used in the production of polyacrylamide gels and polymerization of the monomer produces flocculators useful in mining and purification of water supplies. It is also an important grouting agent used as a soil stabilizer agent being pumped into the soil with a catalyst in the construction of dams, tunnels and roadways. Toxic effects of the monomer were noted soon after it was first manufactured, with reversible tingling, numbness of the fingers and ataxia in the legs of workers. Acrylamide produces a central-peripheral distal axonopathy (dying back). The lesions are most predominant in peripheral motor and sensory fibers, with larger and longer axons being more severely affected. Changes in the central nervous system, axonopathy in the spinal cord, Purkinje cell necrosis in the cerebellum, and dorsal root ganglia changes can occur but are less marked. Acrylamide causes the accumulation of neurofilaments within the affected axons. It is suggested that the axonal degeneration is related to alteration of axonal transport resulting from redistribution of cytoskeletal proteins. Acrylamide treatment increases the phosphorylation of neurofilaments. This increase may be secondary to impairment of axonal transport. Acrylamide is metabolized by mixed function oxidase and/or conjugated to glutathione. The metabolites are less neurotoxic or non-neurotoxic. The vinyl and amide groups are important for the neurotoxicity of acrylamide. The well-described and reproducible effects of acrylamide make it a common choice in neurotoxicity studies requiring a positive-control.
AFIP Diagnosis: Tibial nerve (per contributor): Degeneration, axonal, diffuse, moderate, with mild myelin degeneration, Sprague-Dawley rat, rodent.
Conference Note: Distal axonopathy refers to disorders that produce degeneration of the terminal and preterminal axon. These axonal degenerative changes had previously been identified as "dying back" of the axon. It is now understood that the entire cell body is affected but, because distal axonal projections are dependent on perikaryon synthetic and transport energy, distal portions appear to be more severely affected. Under homeostatic conditions, distal axonal membranes and function are maintained. When there is an energy deficit, deprivation of antioxidants, or physical obstruction of axon transport, the distal portions of the axon cannot be maintained. Failure to deliver required elements and/or failure to remove metabolic by-products leads to axonal degeneration and secondary demyelination follows. Conditions causing this type of distal neuropathy include organophosphate intoxication, thiamine deficiency, vitamin E deficiency, and isoniazid intoxication. In primary sensory neurons, distal axonopathies are characterized by degeneration of both the central axonal projection in the spinal dorsal columns and of the peripheral axon. This type of sensory neuron degeneration is known as central-distal axonopathy. Acrylamide intoxication, as mentioned by the contributor, produces central-peripheral axonopathy by interfering with both anterograde and reterograde fast axonal transport. Accumulation of neurofilaments in the distal portions of the largest and longest axons (the distal ends of spinal cord fibers, and the distal basket fibers of Purkinje cells), signals interference of axonal transport by acrylamide intoxication. In addition to toxins, there are several inherited metabolic defects that result in distal or central-distal axonopathies. Canine giant axonal neuropathy in German Shepherd dogs, for instance, is an inherited central-peripheral axonopathy that causes swelling and degeneration of long axons in the peripheral and central nervous system. Axonal changes in this disease are very similar to those induced by acrylamide. There is a similar human condition, giant axonal neuropathy of children.
Contributor: Corning Hazleton, Pathology Department, P.O. Box 7545, Madison, WI 53707-7545.
References:
1. Abou-Donia MB, and RP Gupta, Involvement of Cytoskeletal Proteins
in Chemically Induced Neuropathies. In: Principles of Neurotoxicolgy,
pp. 153, ed. LW Chang. Marcel Dekker, Inc. New York, NY, 1994.
2. Le Quesne PM. Acrylamide. In: Experimental and Clinical Neurotoxicology. Pp. 309, eds. PS Spencer and HH Schaumburg. Williams and Wilkins, Baltimore, MD. 1980.
3. O'Shaughnessy DJ, and GJ Losos. Compariason of Central and peripheral Nervous System Lesions Caused by High-Dose Short-Term and Low-Dose Subchronic Acrylamide Treatment in Rats. Toxical Path. 14(4):389-394, 1986.
4. Summers BA, Cummings JF, and de Lahunta A: Veterinary Neuropathology. Mosby, St Louis, pp. 437-453, 1995.
* The American Veterinary Medical Association and the American College of Veterinary Pathologists are co-sponsors of the Registry of Veterinary Pathology. The C.L. Davis Foundation also provides substantial support for the Registry.