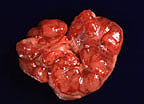
 Hemorrhagic lymph node in a
pig with hog cholera (23K)
Hemorrhagic lymph node in a
pig with hog cholera (23K) Hemorrhagic lymph node in a
pig with hog cholera (23K)
Hemorrhagic lymph node in a
pig with hog cholera (23K)
 Splenic infarction in a pig
injected with hog cholera virus (40X, HE, 91K)
Splenic infarction in a pig
injected with hog cholera virus (40X, HE, 91K)
Signalment: 100 pound mixed breed pig.
History: The pig was inoculated intramuscularly with hog cholera virus (Ames strain). The pig became febrile and depressed two days postinoculation. Bloody conjunctivitis and tonsillar necrosis were noted clinically. The animal was euthanized when moribund at seven days postinoculation.
Gross Pathology: Postmortem findings included multifocal necrosis of tonsillar epithelium, peripheral hemorrhages in some lymph nodes, cerebral edema, and peripheral infarcts in the spleen.
Laboratory Results: Not applicable.
Contributor's Diagnosis and Comments: Infarction, multifocal, subacute, spleen.
Etiology: Hog cholera virus (Genus Pestivirus, Family Flaviviridae).
Hog cholera, also known as classical swine fever, is a severe systemic viral disease caused by a member of the Pestivirus genus. This genus has recently been moved from the Family Togaviridae to the Family Flaviviridae. Thought to have arisen in the Ohio River valley in the early part of the nineteenth century, the disease subsequently spread to many parts of the world. Hog cholera was officially eradicated from the U.S. in 1978 and now North America is one of the few places in the world where it does not occur.
Traditionally, hog cholera is an acute disease of high morbidity and mortality, although evolution of the virus has led to a higher incidence of subacute and chronic forms.
Transmission of the disease occurs when susceptible pigs contact urine, feces, or oronasal secretions of infected pigs. With virulent strains, animals become febrile, anorexic, and severely depressed. Diarrhea and nervous signs are frequently observed. A more insidious form of hog cholera occurs when pregnant sows are infected with strains of lesser virulence. Clinical illness may be inapparent or missed, and the virus passes the placental barrier to the fetus. Depending on the stage of gestation, the piglets may die in utero, survive to term with congenital defects, or be born in a state of immunologic tolerance and shed large quantities of virus.
The most reliable postmortem features of acute hog cholera are tonsillar necrosis, peripheral hemorrhages in the lymph nodes and multifocal petechial hemorrhages. Splenic infarcts are seen uncommonly but when they occur, raise a very high index of suspicion of hog cholera.
The virus replicates principally in lymphocytes and vascular endothelium. Although the most unifying histologic feature - swelling and degeneration of small blood vessels - can be seen in many organs, it is perhaps easiest to visualize in brain. This vascular change is the underlying basis of the necrosis in many tissues.
AFIP Diagnosis: Spleen: Necrosis, angiocentric, multifocal, with hemorrhage (hemorrhagic infarcts), mixed breed, porcine.
Conference Note: The genus Pestivirus includes hog cholera virus, bovine viral diarrhea virus (BVDV) and border disease virus. Hog cholera virus is antigenically similar to BVDV and natural BVDV infection in pigs produces clinical signs and lesions that are indistinguishable from hog cholera. In addition, BVDV, hog cholera virus, and border disease virus each cause transplacental infection and resulting in abortion, fetal abnormalities, or persistent infection.
There are many antigenic strains of hog cholera virus and strains vary in virulence. Virulent strains induce an acute disease that is characterized by fever, anorexia, depression, and leukopenia and a mortality rate that approaches 90%. In acute hog cholera, the virus is transmitted by direct contact and initially replicates in the epithelium of the pharyngeal tonsil, then spreads to the cervical lymph nodes via lymphatic vessels. In 3-4 days, there is a generalized viremia with the virus replicating in epithelial cells, endothelial cells, and cells of the mononuclear phagocyte system. Degeneration and necrosis of endothelial cells leads to vascular compromise, ischemia, and the induction of disseminated intravascular coagulation. These vascular changes are responsible for the lesions associated with classic acute hog cholera including petechial hemorrhage of the kidneys, urinary bladder, and gastric mucosa, splenic infarction and lymph node hemorrhage. CNS disease occurs occasionally and is characterized by vasculitis and perivascular cuffing by mononuclear cells.
Chronic hog cholera infection has been defined as a lethal disease with a duration of 30 days or more. There is less hemorrhage associated with chronic hog cholera. Epithelial damage is often prominent in the gastrointestinal tract (producing a necrotizing colitis/enteritis and button ulcers), and lung. Damage to the lymphoreticular system frequently results in lymphopenia, immunosuppression and secondary bacterial infection.
In utero infection with hog cholera virus can result in abortion, fetal anomalies, or persistently infected piglets. Persistently infected piglets act as a viral reservoir and can develop a late-onset form of hog cholera which is similar to mucosal disease in cattle.
Differential diagnosis for hog cholera considered at the conference included African swine fever and salmonellosis.
Contributor: Foreign Animal Disease Diagnostic Laboratory, USDA, P.O. Box 848, Greenport, NY 11944.
References:
International Veterinary Pathology Slide Bank:
Laser disc frame #12402-417, 20454-457, 22006-7.
 Squamous cell carcinoma in
the lung of a SD rat. (40X, HE, 88K)
Squamous cell carcinoma in
the lung of a SD rat. (40X, HE, 88K)
Signalment: 2-year-old female Sprague-Dawley rat [Crl:CD(R)(SD)BR VAF].
History: This rat was a subject in a 2-year carcinogenicity study. The animals were fed a standard certified laboratory rodent diet containing not less than 23.0% crude protein ad libitum. This rat was humanely euthanized at the termination of the study.
Gross Pathology: The left kidney contained a red and white mottled cystic mass measuring 4.0 cm in diameter. The right kidney contained a white cystic nodule in the cortex.
A 2.5 cm white cystic mass was present in the diaphragmatic lobe of the right lung.
Laboratory Results: None.
Contributor's Diagnosis and Comments: Lung: Squamous cell carcinoma. Kidney(s): Squamous cell carcinoma.
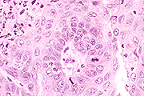
The mass in the left kidney was composed primarily of spindle shaped cells which replaced the renal parenchyma. The mass in the right kidney (submitted) was confined to the cortex, and contained islands and trabeculae of squamous cells as well as sheets of spindle-shaped cells similar to those in the left kidney. In addition, multinucleated cells with eosinophilic cytoplasm were noted within the spindle cell population. The lung contained a single irregular mass composed of islands and trabeculae of well-differentiated squamous cells and keratin pearls. Necrosis was a feature at all three sites. (All features may not be present on every slide.) The lung was considered to be the primary site of this squamous cell carcinoma. Although the sheets of spindle cells present in both renal metastases could be a morphologic form of the primary tumor, these areas were finally interpreted to be stromal proliferation induced by the malignant epithelial cells. Squamous cell tumors with extensive scirrhous response have been reported to arise from the epithelium of the renal pelvis in rats as well as other species, and pulmonary metastasis can occur. However, the squamous cell carcinomas of renal pelvic origin are not known to occur bilaterally. The presence of bilateral renal tumors and a single pulmonary mass made primary squamous cell carcinoma of the lung the most logical diagnosis in this case.
AFIP Diagnosis: 1. Lung: Squamous cell carcinoma, Sprague-Dawley
rat, rodent.
2. Kidney: Squamous cell carcinoma, metastatic.
Conference Note: The lung mass is composed of polygonal cells arranged in trabeculae, nests, and papillary projections that replace the normal pulmonary architecture. The neoplastic cells have distinct cell borders and moderate amounts of eosinophilic, granular cytoplasm. The nuclei are irregularly round, vesiculate, and have 1-4 magenta nucleoli. The mitotic rate is 1 per high power field. There is extensive multifocal necrosis, neutrophilic inflammation, and keratin accumulations. Squamous cell carcinoma of the lung is believed to arise from bronchial, bronchiolar, or alveolar epithelium, or from benign keratinizing squamous cysts. Some of the conference attendees diagnosed this pulmonary tumor as bronchiolo-alveolar carcinoma. Bronchiolo-alveolar carcinomas grow on the preexisting pulmonary stroma in papillary, glandular and/or alveolar patterns and occasionally contain areas of squamous differentiation.
The mass in the kidney is composed of cords of neoplastic epithelial cells which are surrounded by an extensive zone of spindle-shaped cells. In some areas, "intercellular bridges" are evident between the epithelial cells indicating squamous differentiation. The spindle cells do not exhibit clear atypia. Thus, the contributor's opinion that the renal mass is metastatic squamous cell carcinoma accompanied by a marked stromal reaction is favored. Immunohistochemical stains for keratin and vimentin did not stain positive controls and thus could not be interpreted.
Contributor: The Procter & Gamble Company, Miami Valley Laboratories, P.O. Box 398707, Cincinnati, OH 45239-8707.
References:
International Veterinary Pathology Slide Bank:
Laser disc frame #8430, 8519-20, 8550-1.
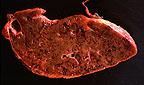 Liver of a sheep with fascioliasis.
(46K)
Liver of a sheep with fascioliasis.
(46K)
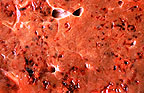 Hepatic necrosis and fibrosis
with multifocal black fluke pigment (33K)
Hepatic necrosis and fibrosis
with multifocal black fluke pigment (33K)
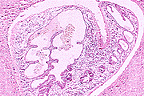 Cross section of Fasciola hepatica
in the liver of a sheep. (20X, HE, 86K)
Cross section of Fasciola hepatica
in the liver of a sheep. (20X, HE, 86K)
Signalment: 4-year-old female Corriedale ovine.
History: This animal belonged to a herd of 154 sheep that have been grazing in a pasture which had been previously used for irrigated rice crops for 3 consecutive years. From August 1 to September 3 (late winter) 18 sheep (11.7%) died after showing weakness, pale discoloration of the mucosae, distended abdominal cavities, and loss of wool. After the animals were noticed ill, death supervened within about 24 hours. The deaths occurred during the lambing season. This sheep was euthanized while moribund.
Gross Pathology: There was about 10 liters of serosanguinous fluid in the abdominal cavity and about 100ml in the pericardial sac. The capsule of the liver was covered by a thick multilayered fibrinous exudate. The liver was somewhat enlarged, with altered shape (more rounded) and firmer. The capsular surface was irregular and the cut surface had multiple linear hemorrhagic tracts on cross section.
Laboratory Results: In a parasitologic examination of the liver, immature forms of Fasciola hepatica were identified. The total number of F. hepatica specimens in the liver was estimated to be 600, all of which were immature forms, 72% around 2-4 weeks of age and 28% of about 5-7 weeks of age.
Contributor's Diagnosis and Comments: Hepatitis, necrohemorrhagic, chronic-active, moderate, with intralesional immature trematodes. Fibrinous perihepatitis, chronic-active, severe.
Etiologic diagnosis: Parasitic hepatitis.
Etiology: Fasciola hepatica.
F. hepatica is the most common and important fluke of the liver of domestic animals and has a cosmopolitan distribution. The adults of F. hepatica (Limneus 1758) are leaf shaped, 2.5cm or longer trematodes that live mainly in the bile ducts or gallbladder of cattle, sheep, goats, and other species including man, although they are economically important only in sheep and cattle. They are hermaphroditic; thus, only one fluke can establish a patent infection. Each adult lives as long as 11 years and may produce 20,000 eggs/day. Due to the longevity of individual mature fluke in bile ducts, infected adult sheep and cattle may remain as carriers. The eggs are passed with the bile from the bile ducts to the duodenum and leave the host in the feces. At optimal temperature (26oC) and moisture, eggs will hatch in 10-12 days producing the first larval stage, the miracidium, which will only survive in a wet environment. The miracidium actively penetrates into a snail of the genus Limnea. Within the snail, sporocysts develop from miracidium. Each sporocyst gives rise to 5-8 rediae. Rediae give rise to daughter rediae and cercariae. The latter leave the snail 4-7 weeks after penetration and settle on blades of grass or other plants just below water level. They then encyst becoming metacercariae and either stay on the plant or sink to the bottom of the water. Under favorable conditions these events (egg to encysted cercariae) take 1-2 months. The final host ingests the metacercariae on the plants. Cattle and other animals that walk into the water to drink may stir up the metacercariae lying at the bottom and swallow them. Ingested metacercariae excyst in the duodenum and penetrate the intestinal wall. Twenty four hours post infection (PI), the majority of young flukes are in the abdominal cavity and by 4-6 days PI, the majority have penetrated the Glisson's capsule of the liver and are migrating through the hepatic parenchyma. Although some immature flukes may reach the liver hematogenously, the usual route is through the peritoneal cavity. Migration within the liver parenchyma lasts 5-6 weeks. About 7 weeks PI, the immature flukes start to reach the bile ducts where they reach sexual maturity within 2-3 months PI. Then, eggs are found in the bile and subsequently in the feces. Some young flukes may accidentally enter hepatic veins and systemic circulation to lodge in unusual sites, particularly the lungs of cattle.
The region where this case occurred is a lowland and moist area, enzootic for F. hepatica infection in cattle and sheep and also for bacillary hemoglobinuria in cattle; as is well known, bacillary hemoglobinuria occurs worldwide only in areas where F. hepatica abounds. It is thought that the fluke induces the initial lesion necessary for clostridial development. Enzootic areas for fascioliasis are the same areas enzootic for the snail (Limnea spp.) which serves as intermediate host. Two main forms of fascioliasis occur depending on the stage of development of the causative agent. Acute fascioliasis occurs 5-6 months after a large number of cercariae suddenly invade the liver. The acute form is mainly due to the traumatic effects of immature wandering flukes in the hepatic parenchyma and is occasionally associated with mortality in sheep as it was in this case. Also, the immature wandering of F. hepatica in the liver of sheep provide a suitable environment for germination of Clostridium novyi, the organism which causes black disease (infectious necrotic hepatitis). Chronic fascioliasis is the slow developing form and results from the action of adult flukes in the bile ducts. Overlap of the two forms may occur.
Frequently irrigated soil, as in the present case, is also highly suitable for the development of F. hepatica. The snails burrow into the soil between irrigations and large numbers of metacercariae are produced when water is present.
The histological liver lesions in this case are typical of acute fascioliasis of sheep and include multiple linear tracts or their cross sections. Immature flukes are seen in some of these tracts. Most of the tracts are lined by hepatocytes in coagulative necrosis and contain degenerated hepatocytes, inflammatory cells, excreta of the flukes and other undetermined debris. They are surrounded by fibrous tissue which occasionally communicates with portal triads. Changes in bile ducts are minimal or absent. In most sections there is a thick layer of fibrin covering the hepatic capsule which is thickened by fibrous connective tissue. Although the histological lesions seen here should be staged as chronic due to the well developed fibrosis, the clinical presentation and pathogenesis of this case are those of the acute clinical form of fascioliasis.
AFIP Diagnosis: Liver: Necrosis and hemorrhage, multifocal to coalescing, with bridging portal fibrosis, chronic inflammation, hemosiderosis and trematode larvae, Corriedale, ovine, etiology consistent with Fasciola hepatica.
Conference Note: Common causes of distomiasis in domestic animals includes Fascioloides magna, Fasciola gigantica, Fasciola hepatica, and Dicrocoelium dendriticum. Fasciola gigantica is found in Africa, Asia, and Hawaii; its morphology and life cycle are similar to F. hepatica.
Fascioloides magna can reach 3 inches in length and 1 inch in width. Its natural hosts are deer and American elk. In definitive hosts, F. magna encysts in a thick fibrous capsule within hepatic parenchyma. The cyst communicates with a bile duct allowing trematode eggs and excretory products to pass from the cyst. In cattle and pigs, F. magna encysts but does not communicate with the bile duct; the cysts fill with eggs and a large amount of fluke-excreted iron-porphyrin pigment. In sheep, F. magna does not encyst and migrates continuously through the parenchyma inducing extensive hepatic necrosis and often causing death.
Dicrocoelium dendriticum requires two intermediate hosts. The first is a land snail in which cercariae develop and are eventually excreted in "slime balls". These cercariae-containing structures are ingested by ants, the second intermediate host. The metacercariae that develop in the ant are infective to the definitive host. One or two metacercariae migrate to the sub-esophageal ganglion of the ant; the ant's behavior is altered, resulting in the ant clinging to the tops of grasses and other plants, facilitating ingestion by grazing herbivores. Once the metacercariae are ingested by the definitive host, they excyst in the duodenum and migrate up the bile duct into the liver.
Some sections contain trematode eggs indicating that adults as well as larvae were present in the liver. The presence of adults explains the varying stages of infection noted histologically, which range from acute necrosis caused by larval migration to chronic inflammation and fibrosis.
Contributor: Universidade Federal de Santa Maria, Departamento de Patologia, Universidade Federal de Santa Maria, 97119-900 Santa Maria RS, Brazil.
References:
International Veterinary Pathology Slide Bank:
Laser disc frame #88, 2902, 3034, 5692, 11737-38, 11740, 12785,
16186-87, 20448, 20449, and 23622-23.
Case IV - DM (AFIP 2508093)
Signalment: Old adult castrated male German Shepherd crossbred dog.
History: The dog was referred to a veterinary teaching hospital for weight loss and dyschezia. Masses around the anus were palpable by digital rectal examination.
Gross Pathology: The left anal sac was replaced by a lobulated, firm, off- white to yellow, spherical mass approximately 2 cm in diameter that was nodular and solid on cut surface. The right anal sac was dilated with secretion and the cranial half of the sac was replaced by a lobulated, white, solid mass measuring 5x10x10 mm. Five retroperitoneal masses similar to those seen at the anal sacs were present in sublumbar and dorsal pelvis areas. These masses measured 1-5 cm in diameter, and filled much of the pelvic canal.
Laboratory Results: Serum calcium concentration was 14.6 mg/dl. Other findings were unremarkable.
Contributor's Diagnosis and Comments: Adenocarcinoma of the apocrine glands of the anal sac. The neoplasm was assumed to be the cause of hypercalcemia in this dog.
AF
IP Diagnosis: 1. Anal sac and adjacent tissues: Adenocarcinoma
of the apocrine glands of the anal sac, German Shepherd Dog, canine.
2. Perianal gland: Adenitis, eosinophilic and granulomatous, focal,
moderate.
Conference Note: There is a lobulated, unencapsulated, expansile and infiltrative neoplasm adjacent to the anal sac. The neoplasm is composed of polygonal to cuboidal cells that form solid lobules and microacini that are supported by a fine to moderate fibrovascular stoma. The neoplastic cells have indistinct cell borders with moderate amounts of lightly eosinophilic, granular cytoplasm. The nuclei are irregularly round with finely stippled chromatin and indistinct nucleoli. The mitotic rate is 1 per 5 high power field. Multifocally there are aggregates of small numbers of lymphocytes and fewer macrophages within the mass.
Adenocarcinoma of the apocrine glands of the anal sac is the most common malignant neoplasm of the perineum in aged female dogs; it is seen less frequently in male dogs. Metastasis is common, frequently to sublumbar lymph nodes, as occurred in this case. Often, the primary tumor is small and not detected clinically, but abdominal lymph nodes are greatly enlarged by metastatic adenocarcinoma. Thus, it is important for the pathologist to recognize this neoplasm when present in biopsy samples of abdominal lymph nodes and other tissues even when there is no history of a mass in the anal sac area. Widespread metastasis may also occur. The majority of dogs with adenocarcinoma of the apocrine glands of the anal sac develop humoral hypercalcemia of malignancy (HHM). Criteria for the diagnosis of HHM includes persistent hypercalcemia and hypophosphatemia, absence of tumor metastasis in bone, atrophy of the parathyroid glands and thyroid C-cell hyperplasia. There is remission of hypercalcemia when the tumor is excised and recurrence with regrowth. The pathogenesis of HHM is not fully understood; however, adenocarcinomas of the glands of the anal sac are associated with elevations in serum levels of parathyroid hormone related protein (PTHrP). When anti-PTHrP antibody is given, serum calcium levels decrease, implying that PTHrP plays a significant role in producing HHM.
Mink have a high incidence of adenocarcinoma of the apocrine glands of the anal sac. In mink, the neoplasm often has a myoepithelial component and is not associated with hypercalcemia. As in dogs, metastasis often occurs when the primary tumor is still quite small.
Contributor: Abbott Laboratories, Department of Pathology, D-469, AP13A, 100 Abbott Park Road, Abbott Park, Illinois 60064-3500.
References:
International Veterinary Pathology Slide Bank:
Laser disc frame #3815, 8346-48.
Dana P. Scott
Captain, VC, USA
Registry of Veterinary Pathology*
Department of Veterinary Pathology
Armed Forces Institute of Pathology
(202)782-2615; DSN: 662-2615
Internet: Scott@email.afip.osd.mil
* The American Veterinary Medical Association and the American College of Veterinary Pathologists are co-sponsors of the Registry of Veterinary Pathology. The C.L. Davis Foundation also provides substantial support for the Registry.