 Adenoviral inclusion in the
endothelium of a calf's kidney. (40X, HE, 103K)
Adenoviral inclusion in the
endothelium of a calf's kidney. (40X, HE, 103K) Adenoviral inclusion in the
endothelium of a calf's kidney. (40X, HE, 103K)
Adenoviral inclusion in the
endothelium of a calf's kidney. (40X, HE, 103K)
Signalment: 35-day-old male Holstein calf.
History: Eight calves from a pen of 50 died without apparent clinical signs.
Gross Pathology: Necropsy examination performed on one of the calves by the attending veterinarian revealed petechial hemorrhages on the serosal surfaces of the spleen, intestines, omentum and the medulla of the kidneys. Fresh and formalin fixed tissues including lung, liver, kidney, spleen and small intestine were submitted.
Laboratory Results: A heavy growth, in pure culture, of a mucoid E. coli was recovered from the small intestine. There was no bacterial growth from the other organs. Non-cytopathic BVD virus was recovered from the spleen.
Contributor's Diagnosis and Comments: Vasculitis with intranuclear inclusion bodies in endothelium, endothelial necrosis, thrombosis and hemorrhage, medulla, kidney.
The feedlot from which this calf was submitted represents a new concept in cattle production. One-day-old Holstein calves are assembled from various dairies in California, Oregon, Idaho and Arizona. They remain briefly in staging areas and then are placed on trucks and sent to a feedlot in central Arizona. For the first three weeks, the calves are in crates, fed milk replacer and started on a grain ration. At three weeks, they are weaned and put on a high concentrate ration in feedlot pens with 50-75 calves of the same age. Up to 10,000 head are present at any given time. Failure of passive transfer is the rule and together with the stress of transport, early weaning and mixing with calves from a wide variety of sources, there have been some interesting and explosive outbreaks of infectious disease observed in this operation. The owners confide that they can tolerate a 10% mortality and still maintain profitability; however, mortality has reached 25% for brief periods. Salmonellosis has been the main culprit but high morbidity and mortality have also occurred due to E. coli, IBR and the hemorrhagic syndrome caused by BVD virus. The adenovirus infection in this example was a relatively minor blip in the background death loss of this operation.
Bovine adenovirus infection is common in cattle but is usually clinically inapparent. Clinical disease is affected by various factors including the strain of virus, concurrent infection, stress, environmental conditions and management practices. The infection is usually manifested as an enteritis +/- respiratory disease. Typical adenovirus inclusions are found in the endothelium of vessels in the lamina propria and submucosa of the rumen, abomasum, and intestine with endothelial necrosis, vascular thrombosis and necrosis of the overlying mucosa. Inclusions may also be found in endothelial cells of the adrenal glands, liver, spleen, kidney, urinary bladder and mesenteric lymph nodes. Vessels with intranuclear inclusion bodies, thrombosis and hemorrhage were present in the serosa of intestinal sections of this calf but none were found in the lamina propria or submucosa. In addition, vasculitis, thrombosis and inclusions were present in the interstitium of the lung sections. Medium and large caliber vessels were predominantly affected. Alveolar walls were diffusely thickened by mononuclear cells.
AFIP Diagnosis: Kidney, medulla: Vasculitis, necrotizing, acute, diffuse, moderate, with hemorrhage, fibrin thrombi, subacute tubulointerstitial nephritis, tubular proteinosis, and endothelial basophilic intranuclear inclusion bodies, Holstein, bovine, etiology consistent with adenovirus.
Conference Note: Adenoviruses are DNA viruses that have been separated into two genera, the Mastadenovirus, or mammalian adenoviruses, and the Aviadenovirus, or avian adenoviruses. Within the genus Mastadenovirus, there are numerous species-specific serotypes, nine of which have been identified in cattle. After transmission, infection, and a viremic stage, adenovirus localizes in the endothelial cells of many organs (including the intestines, stomach and forestomachs, adrenal glands, lymph nodes, liver, spleen, urinary bladder, and kidneys) resulting in thrombosis and ischemia. Epitheliotrophic adenoviruses also have been isolated from ruminants.
Adenovirus is not considered to be an important pathogen in immunocompetent animals, in which it causes a mild respiratory or enteric disease. In immunosuppressed animals, however, adenovirus can cause severe pneumonia (adenovirus causes more deaths than any other pathogen in SCID foals) and ulcerative enteritis. Adenovirus causes more deaths than any other pathogen in severe combined immunodeficiency foals. The calf in this case probably was immunocompromised due to failure of passive transfer and stresses related to shipping. He was also infected with a noncytopathic strain of bovine viral diarrhea (BVD) virus. Recent research has suggested that infection with BVD virus can reduce numbers of B and T lymphocytes, decrease immunoglobulin secretion by B lymphocytes, impair monocyte chemotaxis and polymorphonuclear leukocyte function, and decrease lymphocyte responsiveness to mitogens. Multiple viral infections coupled with failure of passive transfer may explain the severity of lesions in this case.
Several other adenoviral diseases were discussed during the conference including canine adenovirus types I and II and the avian adenoviruses. Within the genus Aviadenovirus, there are three groups based on common group antigens. Group I viruses have been isolated from chickens, turkeys, geese, and other species. Group II viruses include those that cause turkey hemorrhagic enteritis, marble spleen disease, and chicken splenomegaly. Group III viruses are associated with egg drop syndrome in chickens and ducks.
Contributor: University of Arizona Veterinary Diagnostic Laboratory, 2831 N. Freeway, Tucson, AZ 85705.
References:
 Granulomatous panopthalmitis
in a Domestic Shorthair cat. (23K)
Granulomatous panopthalmitis
in a Domestic Shorthair cat. (23K)
 Branching filamentous N.
asteroides in the eye of a cat. (40X, GMS, 103K)
Branching filamentous N.
asteroides in the eye of a cat. (40X, GMS, 103K)
Signalment: 5-year-old neutered female Domestic Shorthair cat.
History: The cat had multiple subcutaneous abscesses which involved the skin of the ventral neck. Nocardia asteroides was cultured. Serologic tests for feline leukemia virus (FeLV) and feline immunodeficiency virus (FIV) were negative. Treatment with antibiotics for one month failed to resolve the lesions and the cat was euthanized.
Gross Pathology: The cat was in poor body condition. The skin of the ventral neck contained a 2.0 cm diameter, open, draining wound containing yellow, purulent exudate. The subcutaneous tissues from the mandible to the thoracic inlet were replaced by pink/tan granular tissue containing pockets of yellow exudate. The regional lymph nodes were swollen and contained foci of similar yellow exudate. The lungs contained multifocal to coalescing, 0.5-1.0 cm diameter, firm tan foci which affected approximately 50% of the lung. Both kidneys contained scattered, 0.25 cm diameter tan foci in the cortex. The retina of the right eye was elevated by soft tan material. A small amount of yellow/tan exudate was present in the anterior chamber.
Laboratory Results: Nocardia asteroides was cultured from the subcutaneous lesions, lymph nodes, kidney, liver, lung and spleen. Impression smears of the bone marrow and spleen were fluorescent antibody positive for FeLV.
Contributor's Diagnosis and Comments: Endophthalmitis,
pyogranulomatous, diffuse, severe, with branching, filamentous
bacteria, right globe. (Etiology: Nocardia asteroides
).
Pyogranulomatous inflammation with branching, filamentous, gram-positive bacteria was also identified microscopically in the lung, kidney, lymph node and skin lesions.
Nocardia is a soil saprophyte and an opportunistic pathogen. Infections may be local or systemic. Local infections are generally acquired via injury. Systemic infections occur subsequent to vascular invasion from focal lesions in the skin, gastrointestinal tract or lungs. Nocardiosis in humans is usually secondary to a pre-existing disorder such as immune- deficiency or immune-suppressive therapy or chronic debilitating disease. Cell mediated immunity is important in resistance to infection whereas humoral immunity is less so. There is some suggestion that depressed immune function may be a predisposing factor in some canine cases where it has occurred secondary to canine distemper virus infection. The concomitant FeLV infection may have been a predisposing factor in this example. Feline nocardiosis is a less common disease than that of dogs. Most cases have involved the thoracic organs with subsequent dissemination.
Bacterial endophthalmitis results from either local penetrating wounds or hematogenous spread of organisms. The ciliary body or choroid is usually affected first when the hematogenous route is involved. Any organism which can initiate a bacteremia may be involved, but specific examples of hematogenously acquired, bacterial endophthalmitis in cats are rare. Mycobacterium tuberculosis is mentioned but is largely of historical interest.
AFIP Diagnosis: Eye: Chorioretinitis, pyogranulomatous, fibrinohemorrhagic, severe, with retinal detachment, mild subacute anterior uveitis, and branching filamentous bacteria, Domestic Shorthair, feline.
Conference Note: Nocardia spp. belong to the family Nocardiaceae of the order Actinomycetales; this order also includes the families Mycobacteriaceae, Actinomycetaceae, Dermatophiaceae, and Streptomycetaceae. All Actinomycetales, except for mycobacteria, are referred to as actinomycetes. Nocardia spp. are branching, filamentous, gram-positive, partially acid-fast, GMS-positive, aerobic bacteria. Three species of Nocardia have been associated with disease in animals: N. asteroides, N. caviae, and N. brasiliensis. The differential diagnosis for filamentous bacterial infections also include the Actinomycetaceae; these bacteria tend to branch less, produce granules, are not acid-fast and are anaerobic or microaerophilic. All actinomycetes have complex cell walls which contain glucosamine, muramic acid, glutamic acid and alanine. In addition, the cell walls of Nocardia contain arabinose and galactose; the presence of these compounds defines their cell walls as type IV, a trait shared by the mycobacteria. The glycolipid make-up of the cell wall is responsible for the acid-fast staining of Nocardia, and plays a role in escaping phagolysosome formation and the effects of the oxidative burst.
Nocardial infections cause nonhealing wounds with involvement of deeper tissues and draining lymph nodes, empyema and disseminated disease. The localization of the eye lesion at the entrance of the central retinal artery suggests hematogenous spread in this case. Draining tracts are a regular feature; granule formation, however, is not commonly seen in N. asteroides infections. The inflammatory response often produces a serosanguinous exudate.
The lack of serum antibody is not uncommon in the terminal stages of retroviral disease, as seen in this case, and is caused by lymphocyte depletion and a paucity of immunoglobulin production. This case illustrates the importance of testing for the virus when retroviral infection is strongly suspected and serological testing is negative. This pattern is often reversed in the early stages of retroviral infection when there is a large amount of serum antibody present, but the virus is present in small numbers and can be difficult to detect. Immunosuppression resulting from FeLV infection probably predisposed this cat to the disseminated nocardial infection.
Contributor: University of Arizona, Department of Veterinary Sciences, 2831 N. Freeway, Tucson, AZ 85705.
References:
International Veterinary Pathology Slide Bank:
Laser disc frame #2460-61, 3191-93, 8768-70, 10140, 10643, 13105,
14410, 14612- 15.
 Multiple hepatic granulomas
in a Nubian-cross goat. (24K)
Multiple hepatic granulomas
in a Nubian-cross goat. (24K)
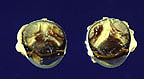
Signalment: 1-year-old Nubian-cross, buck, caprine
History: The Nubian-cross buck was presented to the Michigan State University. Veterinary Teaching Hospital large animal clinic for lameness and intermittent carrying of the right foreleg. Physical examination revealed laxity of the right glenohumeral joint and marked muscle atrophy in this region. Radiographs of the proximal right forelimb and shoulder revealed two radiolucent regions within the medullary cavity of the humerus consistent with either cystic or early lytic lesions. Euthanasia was elected, due to the progressive nature of this condition.
Gross Pathology: At necropsy the liver contained numerous disseminated, raised, oval, variably-sized (0.5 to 2.0 cm) nodules. On cut surface, these nodules contained off- white to tan caseous material, arranged in concentric rings and bounded by a thin tan connective tissue capsule. Additional similar appearing nodules were evident in multiple locations, including several abdominal lymph nodes (hepatic, renal, and mesenteric), the lung, right humerus and right 5th rib.
Laboratory Results: Bacteriologic culture of samples of abdominal lymph node, lung, and liver resulted in isolation of Rhodococcus equi at >1000, >1000, and <50 colony forming units, respectively. Culture results for Mycobacterium species were negative. Serum samples were negative on agar gel immunodiffusion testing for caprine arthritis encephalitis virus. Formalin-fixed tissues were submitted for histologic examination.
Contributor's Diagnosis and Comments: Extensive disseminated caseogranulomatous hepatitis with intracytoplasmic bacterial organisms. Etiology: Rhodococcus equi
Sections of liver contained multiple variably sized abscesses, characterized by central zones of caseous necrosis and cellular debris limited by intact neutrophils, lymphocytes, plasma cells, epithelioid macrophages, and multinucleated giant cells. The cytoplasm of macrophages and giant cells was composed of abundant lightly eosinophilic foamy material and occasional large, clear vacuoles containing fine basophilic granular material. This reaction was surrounded by a wide zone of fibroblast proliferation and a fibrous connective tissue capsule. There were multifocal central irregularly shaped basophilic granular zones of mineralization in many of the well developed abscesses. The surrounding hepatic parenchyma was compressed (atrophy) and displaced by the larger abscesses. Portal triads contained prominent lymphoid nodules, many of which had the characteristic appearance of reactive lymphoid nodules, with centrally located histiocytes and immature lymphocytes (lymphoblasts) surrounded by marginating layers of mature lymphocytes and plasma cells. Also noted were smaller lymphocytic aggregates throughout the parenchyma. There were occasional early formative abscesses with central neutrophils limited by lymphocytes, plasma cells, and macrophages present throughout the hepatic parenchyma. Mild bile duct proliferation was evident in these sections. Similar appearing abscesses were evident in sections of lung, abdominal lymph nodes (hepatic, mesenteric, and renal), and the medullary cavity of the right humerus and fifth rib. Gram stains identified large numbers of Gram positive coccobacilli located in clear vacuoles within the cytoplasm of macrophages and multinucleated giant cells.
Corynebacterium ovis was originally suspected as the etiology of the disseminated caseogranulomas in numerous tissues in this goat. Initially a neoplastic process involving the right humerus was suspected by the radiologists and clinicians involved in this case. The widespread distribution of the abscesses in an apparently healthy buck was equally surprising. There are a few reports of isolation of Rhodococcus equi from pulmonary or liver abscesses in goats. The presence of bone abscesses in the humerus and rib due to R. equi were considered quite unusual and to our knowledge has not been previously reported. This case suggests that R. equi should be considered as a possible cause of abscessation in goats along with the other more commonly recognized bacterial agents. AFIP Diagnosis: Liver: Granulomas, multiple, with caseous necrosis, mineralization, encapsulation, lymphoplasmacytic portal hepatitis, neutrophilic cholangitis, and intrahistiocytic coccobacilli, Nubian-cross, caprine.
Conference Note: Rhodococcus equi is a gram-positive bacterium that belongs to the order Actinomycetales. All actinomycete members have a complex cell wall which contains glucosamine, muramic acid, glutamic acid and alanine. This complex cell wall, like that of Nocardia and Mycobacterium, is thought to prevent phagosome-lysosome fusion, resist the oxidative burst and cause a nonspecific degranulation of lysosomes, thereby allowing intrahistiocytic survival and chronic infection.
Rhodococcus equi more commonly causes pyogranulomatous pneumonia and/or ulcerative enterocolitis in foals 1-3 months of age. Disseminated disease occurs infrequently in foals resulting in suppurative arthritis, osteomyelitis, hepatitis and splenitis. Immune- mediated (type III hypersensitivity) polysynovitis is a common sequela to foal rhodococcal pneumonia due to synovial IgG-antigen complex deposition. In pigs and cattle, R. equi causes lymphadenitis with granulomas similar to those in this case. Rhodococcus equi has also been recovered from respiratory lesions in human AIDS patients.
Most conference attendees included Corynebacterium ovis and Mycobacterium sp. in their differential diagnosis. Definitive diagnosis is usually dependent upon bacterial culture.
Contributor: Animal Health Diagnostic Laboratory, P.O. Box 30076, Lansing, MI 48909-7576.
References:
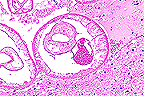 Cross-section of larval Parelaphestrongylus
in the brain of a goat. (40X, HE, 64K)
Cross-section of larval Parelaphestrongylus
in the brain of a goat. (40X, HE, 64K)
Signalment: 3-year-old female Toggenburg goat.
History: This goat had been displaying progressive signs of CNS disturbance for about one month. The disease started with ataxia and developed into a syndrome characterized by circling, compulsive walking, hypermetria, sensory deficits and eventually recumbency.
Gross Pathology: The goat was judged to be in fair body condition. No significant gross morphological alterations were observed with the exception of considerable amounts of clear, water-like fluid flowing from the subdural space upon incision.
Laboratory Results: None submitted.
Contributor's Diagnosis and Comments: Brain (frontal lobe): Meningoencephalitis, necrotizing, multifocal with sections of coiled larval nematode morphologically consistent with Elaphostrongylus/Parelaphostrongylus sp.
Lesions were present throughout the brain but predominated in the white matter of the frontal and temporal lobes, the basal ganglia and thalamus. Other brain regions, as well as the spinal cord, were found to be far less affected. Sections of a coiled larval nematode were found only in the white matter of the frontal lobe, although multiple malacic foci suggestive of larval migration were observed. Lymphocytes and macrophages were the predominant infiltrating cells; low numbers of eosinophils were present and glial reaction was enhanced in regions with necrotic foci.
AFIP Diagnosis: Cerebrum: Meningoencephalitis, lymphoplasmacytic and eosinophilic, multifocal, mild, with necrosis and larval metastrongyles, Toggenburg, caprine, etiology consistent with Parelaphostrongylus tenuis.
Conference Note: This case was reviewed by Dr. C. H. Gardiner, the parasitology consultant for the Department of Veterinary Pathology. Present in the section is a female larva of Parelaphostrongylus
. The parasite has a cuticle which expands laterally to form lateral internal ridges. The musculature is coelomyarian. There are accessory hypodermal chords, a feature limited to metastrongyles. The intestine is large, contains pigment and is composed of a few multinucleate cells.
P. tenuis naturally infects the American white-tailed deer (Odocoileus virgianus). In the white-tail, it causes little or no clinical disease, but in aberrant hosts it can cause severe neurologic lesions. Infections have been diagnosed in moose, caribou, wapiti, red deer, sheep, goats, and numerous exotic ruminants. Cattle are refractory to infection. The parasite range extends throughout the eastern U.S. and Canada.
Infective P. tenuis larvae (L3) are ingested by the host in a snail or slug (the obligate intermediate host). It is hypothesized that larvae penetrate the abomasal wall, traverse the peritoneal cavity, and reach the spinal cord within 10 days. Migration to the spinal cord may be along the spinal nerves. In white-tailed deer, the larvae develop and migrate within the dorsal horns of the gray matter for 20-30 days, then migrate into the subdural space. There they mature and deposit eggs in the meninges. The larvae hatch, enter the circulation and are carried to the lungs where they penetrate into the alveoli, are coughed up, swallowed and passed in the feces. The larvae then enter the intermediate host and develop into infective L3 larvae. In aberrant hosts the larvae fail to migrate into the meninges, but continue to develop and grow within the spinal cord and brain.
Angiostrongylus vasorum and A. cantonensis were also discussed. These parasites utilizes mollusks as intermediate hosts. When ingested, the larvae undergo an obligatory migration to the brain. About 4 weeks after ingestion, they enter the bloodstream, penetrate into alveolar spaces, and are coughed up, swallowed, and passed in the feces. A. vasorum causes pulmonary arteritis and cor pulmonale. A. cantonensis is a cause of eosinophilic meningoencephalitis in humans and other primates; they contract the disease by ingesting raw or undercooked intermediate or paratenic hosts.
Contributor: University of Zurich, Institute of Veterinary Pathology, Winterthurerstrasse 268, CH 8057 Zurich, Switzerland.
References:
Dana P. Scott
Captain, VC, USA
Registry of Veterinary Pathology*
Department of Veterinary Pathology
Armed Forces Institute of Pathology
(202)782-2615; DSN: 662-2615
Internet: Scott@email.afip.osd.mil
* The American Veterinary Medical Association and the American College of Veterinary Pathologists are co-sponsors of the Registry of Veterinary Pathology. The C.L. Davis Foundation also provides substantial support for the Registry.