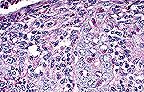
 Necrotizing hepatitis in a
SCID mouse with cytomegalovirus inclusions (40X, HE, 103K)
Necrotizing hepatitis in a
SCID mouse with cytomegalovirus inclusions (40X, HE, 103K)
 Necrotizing hepatitis in a
SCID mouse with cytomegalovirus inclusions (40X, HE, 103K)
Necrotizing hepatitis in a
SCID mouse with cytomegalovirus inclusions (40X, HE, 103K)
Signalment: 10-week-old male, pathogen free C.B-17-scid/scid mice
History: Twenty mice were given intraperitoneally 0.1 ml of inoculum containing 3 x 104 plaque forming units (PFU) of a viral agent that occurs naturally in wild Mus musculus. Three animals were to be sacrificed and all major organs collected for histopathologic evaluation on postinoculation (PI) days: 5, 8, 12, 15, 18 and 21. However, all mice that remained alive after day 15 died on the 16th and 17th days PI. Most mice had rough hair coats and hunched posture beginning on day 12 PI. The tissues submitted were from the three mice killed on day 15 PI.
Gross Pathology: Mice killed on days 12 and 15 PI had small pale to red foci in their livers and pale spleens. Tissues of all major organs were fixed in 70% alcoholic formalin, routinely sectioned at 5 mæ and stained with H&E.
Laboratory Results: Histopathologic changes in these mice followed a distinct pattern. The liver and spleen were the prime targets of the infection. On day 5, they had only mild focal necrosis; severity increased steadily thereafter. At day 15, the number and size of necrotic foci in liver had increased to a point of coalescence in some mice; the spleen was diffusely affected by the necrotizing process. At day 5, organs other than liver and spleen were unaffected. By day 8, mild focal necrosis was present in: salivary glands, stomach, small intestine, large intestine, lymph nodes and kidney. The lungs and small foci of alveolar macrophage proliferation; subsequently, these foci enlarged but never reached the severity of lesions in liver and spleen. Foci of necrosis appeared in pancreas and adrenal beginning at day 12 and in nasal passages and bone marrow at day 15. In some cases, focal necrosis and erosion of mucosal epithelium occurred in stomach and large intestine beginning at day 12 and increasing to ulcerations at 15, suggesting that these lesions and sepsis from the gut flora may have contributed to deaths of animals beyond 15 days PI. Cytomegaly and intranuclear inclusions typical of cytomegalovirus infections were present in all lesions.
Contributor's Diagnosis and Comments:
Etiology: Experimental mouse cytomegalovirus infection.
Natural infections of murine cytomegalovirus (MCMV) occur primarily in wild mice. Laboratory mice experimentally infected with this virus are frequently used as models of human cytomegalovirus infection. The purpose of the study from which these cases were taken was to characterize the histologic disease following administration of a single dose (3X104PFU) of a standard laboratory strain of MCMV (Smith) to C.B-17-scid/scid mice, for use as a model for testing the efficacy of new antiviral compounds. Mice homozygous for the autosomal recessive gene, severe combined immunodeficiency (scid) on chromosome 16, lack B and T cell-mediated immune responses while retaining myeloid, natural killer and antigen-presenting cell activities. The molecular basis is defective VDJ recombinase activity.
Another genetic factor, Cmv-1, also may have been involved. Cmv-1, located in the natural killer complex on chromosome 6, is the autosomal dominant non-H-2 gene that controls MCMV replication in the spleen. The Cmv-11 allele (low splenic replication of MCMV) in mouse strains of C57BL background confers resistance to lethal infection and the Cmv-1h allele (high splenic replication of MCMV) in BALB/c, DBA/2J and A/J mice confers susceptibility to lethal disease. (BALB/c is the major parental contributor to C.B-17-scid/scid). Nevertheless, the uniform high susceptibility of C.B-17-scid/scid mice to MCMV makes them ideal for in vivo testing of antiviral compounds.
That the liver and spleen were found in our small study to be the initial targets of infection also is in keeping with the pattern of pathogenesis recently reported by Collins, et al. (1994). They found that following MCMV infection of mice there is (i) a primary viremia (virus in circulating leukocytes) that peaks on days 5 to 7 PI and results in dissemination of virus mainly to liver and spleen (reticuloendothelial organs), and (ii) after viral amplification in liver and spleen, a subsequent, more intense secondary viremia occurs with dissemination to other organs.
AFIP Diagnosis:
Conference Note: Cytomegalovirus is a host-specific beta herpesvirus that has been isolated from humans, various primates, pigs, mice, rats, guinea pigs, hamsters, moles, horses, and cows. The virus is spread by respiratory droplets, urine and reproductive fluids; perinatal transmission at parturition and intrauterine transmission also occur. In humans, transmission by blood transfusion and transplation of virus infected grafts has also been documented. Murine cytomegalovirus (MCMV) infections are subclinical and limited to the salivary glands in immunocompetent mice . In man, cytomegalovirus has become a common and lethal consequence of HIV infection. This has created interest in immunosuppressed mice with MCMV as a model for the study of the human AIDS associated disease and for testing new antiviral compounds.
As previously noted, natural infection with cytomegalovirus is uncommon in laboratory mice. The presence of necrosis and syncytial cell formation in various organs is more frequently seen in laboratory mice infected with mouse hepatitis virus (MHV). This coronavirus is very common and has a high mortality rate in neonatal mice. There are two antigenically indistinguishable biotypes. One is a respiratory form that first affects the nasal epithelium then disseminates to the liver, lymphoid tissues, and other organs. Within target organs there is necrosis and the formation of synctial cells. There is also an enteric form that most often localizes in the epithelium of the colon with minimal dissemination.
The spleen in this case demonstrates the extramedullary hematopoiesis that is commonly seen in many rodents; this is considered to be a normal finding. Also of note is the lymphoid depletion in the splenic white pulp and lymph nodes, an expected histologic finding in scid mice.
Contributor: Department of Comparative Medicine, University of Alabama at Birmingham, Birmingham, AL 35294-0019.
References:
Signalment: Adult female, Sprague-Dawley rat
History: This rat was sacrificed moribund on day 624 of a carcinogenicity study. The abdominal cavity contained a moderate amount of dark red fluid and multiple yellow gelatinous tissue masses floating free. The ovaries were mottled, had a gelatinous appearance and measured 5x5x4 cm and 2x1x1 cm. Multiple serosal surfaces in the abdomen were seeded by mottled, gelatinous nodules. Additional gross findings at necropsy were those that are commonly seen in aged rats and included tissue masses in the pituitary, mammary gland, and liver.
Gross Pathology: none submitted
Laboratory Results: none submitted
Contributor's Diagnosis and Comments: Ovary: Yolk sac carcinoma, bilateral and malignant thecal-cell tumor, bilateral.
Slides were submitted from two blocks that were prepared from the left ovary. All slides contain large areas of the thecal component characterized by whorls of closely apposed spindle cells. On the edge of the section, finely vacuolated cuboidal cells are arranged in nests or ribbons in a homogenous eosinophilic intercellular matrix and represent the yolk sac carcinoma.
The unusual feature of this case was the presence of two uncommon ovarian tumors in both ovaries of one rat. The yolk sac component widely metastasized by seeding on serosal surfaces in the abdomen; yolk sac carcinoma was present in sections of the mesentery, mesenteric lymph nodes, stomach, small intestine, spleen, kidney, liver, uterus, urinary bladder and free in the abdomen.
AFIP Diagnosis:
Conference Note: Primary ovarian tumors are derived from either epithelial cells, sex cord-stromal cells, germ cells, or soft tissues. Epithelial tumors are derived from the surface epithelium of the ovary; in the rat, they include cystadenoma/cystadenocarcinoma, tubulostromal adenoma/carcinoma, and mesothelioma. The sex cord-stromal tumors are the granulosa cell tumors, luteomas, thecomas, sertoliform tubular adenoma and the undifferentiated sex cord-stromal tumor. Dysgerminoma, teratoma, choriocarcinoma, and yolk sac carcinoma comprise the germ cell origin tumors of the ovary. The soft tissue neoplasms are the fibroma/fibrosarcoma and hemangioma/hemangiosarcoma. The incidence of ovarian tumors differs among strains of mice. Epithelial tumors are more common than granulosa/theca cell tumors.
Yolk sac carcinomas are characterized by an eosinophilic, PAS-positive matrix which surrounds uniform cells with well defined cell borders. Thecomas consist of spindle or stellate cells, often arranged in interlacing fascicles. The tumor was considered benign because of the lack of atypia, low mitotic rate, and expansive growth. Thecomas may produce estrogen, especially in cattle.
Contributor: Ciba-Geigy, 556 Morris Avenue, Summit, NJ 07901.
References:
Signalment: 22-week-old, male, Fischer 344 rat
History: Tissue are from two that were euthanized following a 90 day continuous inhalation exposure to 1000mg/m3 shale-derived JP4 jet fuel vapor.
Gross Pathology: none submitted
Laboratory Results: none submitted
Contributor's Diagnosis and Comments:
The submitted sections of kidney demonstrate the triad of histologic changes described in male rats with hydrocarbon-induced nephropathy. The lesion is characterized by intracytoplasmic accumulation of large hyaline droplets in proximal convoluted tubules, formation of eosinophilic casts distending tubules in the outer medulla, and acceleration of the spontaneously occurring chronic nephropathy of rats. Features of chronic nephropathy in these sections include basophilic alteration and regeneration of tubular epithelium, thickening of tubular and glomerular basement membranes, tubular atrophy, interstitial fibrosis and lymphocyte infiltration, and occasional hyaline casts.
The primary site of injury with hydrocarbon nephropathy is thought to be the P2 segment of the proximal convoluted tubule. Binding of hydrocarbons to the protein à2æ- globulin in the cytoplasm of these epithelial cells inhibits lysosomal catabolism of the protein, forming large globular to angular, eosinophilic droplets within secondary lysosomes. The droplets stain positive with Mallory's Heidenhain stain, but do not stain with periodic acid-Schiff (PAS) procedure. Accumulation of these droplets leads to lysosomal protein overload and ultimately cell death. Sloughed cells and necrotic debris become bottlenecked downstream forming large granular casts at the junction of the P3 segment and the thin loop of Henle in the medulla. Although the above lesions are reversible post-exposure, rats in the chronic recovery period often develop characteristic kidney lesions including linear basophilic mineral deposits in the renal papilla, hyperplasia of pelvic urothelium, and continued exacerbation of chronic progressive nephrosis.
Hyaline droplet nephropathy is a sex and species specific condition unique to male rats. The list of chemicals that induce à2æ-globulin accumulation (CIGA) is extensive and includes certain fuels, solvents, pesticides, drugs, and even food additives such as d-limonene. Some CIGAs have also been shown to increase the incidence of renal tumors in male rats. Since many of these compounds are not directly mutagenic, their influence on tumorigenesis is thought to be via promotion. A proposed mechanism for CIGA-associated renal tumors in male rats states that the hyaline droplet nephropathy and subsequent cytotoxicity cause a sustained and increased proliferation of regenerating tubular epithelium. This proliferative response allows for a promotion effect on spontaneously initiated cells and/or an increased frequency of mutation in these cells. If this hypothesis is correct, then the CIGA- associated renal tumors found exclusively in male rats may be irrelevant to human carcinogenic risk assessment of nongenotoxic chemicals.
AFIP Diagnosis:
Conference Note: As the contributor notes, there are many chemicals which can cause the accumulation of hyaline droplets in the renal proximal tubular epithelium of male rats. Hyaline droplet accumulation has also been noted in both male and female rats and mice with histiocytic sarcoma. The protein in these hyaline droplets was determined to be lysozyme, a major secretory product of macrophages and monocytes. Hyaline droplet accumulation secondary to neoplasia must be differentiated from the chemically induced hyaline droplet nephropathy in male rats.
Chronic nephropathy is the most common renal disease of old laboratory rats; by 2 years of age, 75% of rats are affected. Development of chronic nephropathy is dependent on genetic background (there is a high incidence in albino strains, Fischer- 344, Wistar and Marshall rats), sex (males are more susceptible than females; there is a decreased incidence in castrated males), and diet (incidence of disease increases with high protein diet). Early changes consist of thickening of the mesangium and basement membranes of the glomerulus. These changes progress with age resulting in periglomerular fibrosis, crescent formation, synechia, and glomerular obsolescence. Tubular changes include thickening of the basement membranes, atrophy, and dilatation and hyaline cast formation. The most prominent clinico-pathologic change is albuminuria. Advanced stages of chronic nephropathy may interfere with differentiation of age related changes and drug induced changes in chronic toxicity studies.
Contributor: Triservice Toxicology, Armstrong Laboratory (AL/OET), Path, 2856 G Street, Bldg. 79, Wright-Patterson AFB, OH 45433-7400.
References:
 Botryomycosis due to Staphylococcus
infection in a nude mouse. (40X, HE, 97K)
Botryomycosis due to Staphylococcus
infection in a nude mouse. (40X, HE, 97K)
Signalment: Female athymic nude mouse
History: Mouse was losing weight after being in the facility for 6 months.
Gross Pathology: Multiple, small white nodules were present subcutaneously in the parotid and submandibular area.
Laboratory Results: Staphylococcus aureus cultured from lesion.
Contributor's Diagnosis and Comments: Multiple periglandular and intraglandular abscesses of the parotid and submandibular salivary gland.
Staphylococci are common microflora of the skin which cause a facial dermatitis in several strains of mice, both normal and immune deficient. Susceptible strains include C56BL, C3H, DBA and BALB/c. Athymic nudes often develop multiple abscesses, ulcerations or deep granulomas. Lesions are usually initiated by a skin wound, and their spread is aided by such conditions as high environmental temperature or humidity, crowding, dietary change, rapid growth or low dietary protein. The term botryomycosis is often used to refer to chronic staphylococcal infections characterized by granule formation and histologically by Splendore-Hoeppli material coating bacterial colonies, as seen in this case. Staphylococcal infections of the genital mucosa in males commonly produce preputial gland abscesses.
AFIP Diagnosis: Salivary gland and associated soft tissue: Inflammation, chronic- suppurative, multifocal, severe, with Splendore-Hoeppli material, and numerous gram- positive cocci, athymic nude mouse, rodent.
Conference Note: Staphylococci cause many types of infections including pneumonia, endocarditis, toxemia, and dermatitis. These bacteria possess many virulence factors that potentiate their ability to invade host tissues and survive. Staphylococcus aureus has surface receptors for fibrinogen, fibronectin and vitronectin, which allow them to bind to endothelial cells. S. aureus also has a laminin receptor which binds to extracellular matrix proteins and allows for tissue invasion. Many staphylococci have capsules which resist phagocytosis, and they possess a lipase which degrades protective lipids on the skin surface. S. aureus also produces a wide variety of hemolytic toxins and enterotoxins.
Botryomycosis in mice is often caused by coagulase-positive Staphylococcus. As the contributor notes, the bacteria often gain entrance after traumatic disruption of the epithelium. Many abscesses occur on the head, as in this case, and these mice often have periodontal granulomas and microabscesses. This suggests that the bacteria are introduced via dental alveoli and then spread through the subcutis and facial soft tissues. Although most cases of botryomycosis involve individual animals, there have been outbreaks of human phage types of Staphylococcus aureus in specific pathogen free colonies of mice. C57BL/6Bd(SPF) strains, swiss albinos, C57BL/6J strains, and C3H/HeJ strains are reported to be the most susceptible. It is not possible to eliminate staphylococci from barrier maintained mice, and periodic monitoring of the environment, bedding, food, and personnel may be necessary to minimize the spread of pathogenic strains.
A number of other infectious agents that cause suppurative or granulomatous disease induce formation of granules of Splendore-Hoeppli material. Included are fungi such as Pseudallescheria boydii, Madurella spp., Fusarium spp., Aspergillus nidulans, the dermatophytes, and many others opportunistic fungi and bacteria such as Actinomyces, Streptomyces, Nocardia, Streptococcus, Escherichia, Pseudomonas, Proteus, and probably many others.
Contributor: Center for Biologics Evaluation & Research, Food & Drug Administration, 8800 Rockville Pike, Bethesda, MD 20892.
References:
Dana P. Scott
Captain, VC, USA
Registry of Veterinary Pathology*
Department of Veterinary Pathology
Armed Forces Institute of Pathology
(202)782-2615; DSN: 662-2615
Internet: Scott@email.afip.osd.mil
* The American Veterinary Medical Association and the American College of Veterinary Pathologists are co-sponsors of the Registry of Veterinary Pathology. The C.L. Davis Foundation also provides substantial support for the Registry.